Distinct mechanisms of inhibition of Kv2 potassium channels by tetraethylammonium and RY785
Curation statements for this article:-
Curated by eLife
eLife Assessment
This study represents an important advance in our understanding of how certain inhibitors affect the behavior of voltage gated potassium channels. Robust molecular dynamics simulation and analysis methods lead to a new proposed inhibition mechanism with strength of support being mostly convincing, though computational evidence is limited for some conformations discussed. This study has considerable significance for the fields of ion channel physiology and pharmacology and could aid in development of selective inhibitors for protein targets.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
- Reading List (BiophysicsColab)
Abstract
Voltage-gated K+ channels play central roles in human physiology, both in health and disease. A repertoire of inhibitors that are both potent and specific would therefore be of great value, not only as pharmacological agents but also as research tools. The small molecule RY785 has been described as particularly promising in this regard, as it selectively inhibits channels in the Kv2 subfamily with high potency. Kv2 channels are expressed in multiple cell types in humans, and are of particular importance for neuronal function. The mechanism of action of RY785 has not yet been determined at the molecular level, but functional studies indicate it differs from that of less specific inhibitors, such as quaternary-ammonium compounds or aminopyridines; RY785 is distinct also in that it is electroneutral. To examine this mechanism at the single-molecule level, we have carried out a series of all-atom molecular dynamics simulations based on the experimental structure of the Kv2.1 channel in the activated, open state. First, we report a 25-microsecond trajectory calculated in the absence of any inhibitor, under an applied voltage of 100 mV, which demonstrates outward K+ flow under simulation conditions at rates comparable to experimental measurements. Additional simulations in which either RY785 or tetraethylammonium (TEA) is introduced in solution show both inhibitors spontaneously enter the channel through the cytoplasmic gate, with distinct effects. In agreement with prior structural studies, we observe that TEA binds to a site adjacent to the selectivity filter, on the pore axis, thereby blocking the flow of K+ ions. RY785, by contrast, binds to the channel walls, off-axis, and allows K+ flow while the cytoplasmic gate remains open. The observed mode of RY785 binding, however, indicates that its mechanism of action is to stabilize and occlude a semi-open state of the gate, by bridging hydrophobic protein-protein interactions therein; this hypothesis would explain the puzzling experimental observation that RY785 recognition influences the gating currents generated by the voltage sensors, 3 nm away.
Article activity feed
-

eLife Assessment
This study represents an important advance in our understanding of how certain inhibitors affect the behavior of voltage gated potassium channels. Robust molecular dynamics simulation and analysis methods lead to a new proposed inhibition mechanism with strength of support being mostly convincing, though computational evidence is limited for some conformations discussed. This study has considerable significance for the fields of ion channel physiology and pharmacology and could aid in development of selective inhibitors for protein targets.
-

Reviewer #1 (Public review):
Summary:
The authors were seeking to identify a molecular mechanism whereby the small molecule RY785 selectively inhibits Kv2.1 channels. Specifically, the authors sought to explain some of the functional differences that RY785 exhibits in experimental electrophysiology experiments as compared to other Kv inhibitors, namely the charged and non-specific inhibitor tetraethylammonium (TEA). The authors used a recently published cryo-EM Kv2.1 channel structure in the open activated state and performed a series of multi-microsecond-long all-atom molecular dynamics simulations to study Kv2.1 channel conduction under the applied membrane voltage with and without RY785 or TEA present. They observed that while TEA directly blocks K+ permeation by occluding ion permeation pathway, RY785 binds to multiple non-polar …
Reviewer #1 (Public review):
Summary:
The authors were seeking to identify a molecular mechanism whereby the small molecule RY785 selectively inhibits Kv2.1 channels. Specifically, the authors sought to explain some of the functional differences that RY785 exhibits in experimental electrophysiology experiments as compared to other Kv inhibitors, namely the charged and non-specific inhibitor tetraethylammonium (TEA). The authors used a recently published cryo-EM Kv2.1 channel structure in the open activated state and performed a series of multi-microsecond-long all-atom molecular dynamics simulations to study Kv2.1 channel conduction under the applied membrane voltage with and without RY785 or TEA present. They observed that while TEA directly blocks K+ permeation by occluding ion permeation pathway, RY785 binds to multiple non-polar residues near the hydrophobic gate of the channel driving it to a semi-closed non-conductive state. They confirmed this mechanism using an additional set of simulations and used it to explain experimental electrophysiology data,
Strengths:
The total length of simulation time is impressive, totaling many tens of microseconds. The authors develop their own forcefield parameters for the RY785 molecule based on extensive QM based parameterization. The computed permeation rate of K+ ions through the channel observed under applied voltage conditions is in reasonable agreement with experimental estimates of the single channel conductance. The authors have performed extensive simulations with the apo channel as well as both TEA and RY785. The simulations with TEA reasonably demonstrate that TEA directly blocks K+ permeation by binding in the center of the Kv2.1 channel cavity, preventing K+ ions from reaching the SCav site. The authors conclude that RY785 likely stabilizes a partially closed conformation of the Kv2.1 channel and thereby inhibits K+ current. This conclusion is plausible given that RY785 makes stable contacts with multiple hydrophobic residues in the S6 helix, which they can also validate using a recently published closed-state Kv2.1 channel cryo-EM structure. This further provides a possible mechanism for the experimental observations that RY785 speeds up the deactivation kinetics of Kv2 channels from a previous experimental electrophysiology study.
Weaknesses:
The authors, however, did not directly observe this semi-closed channel conformation and in fact acknowledge that more direct simulation evidence would require extensive enhanced-sampling simulations beyond the scope of this study. They have not estimated the effect of RY785 binding on the protein-based hydrophobic pore constriction, which may further substantiate their proposed mechanism. And while the authors quantified K+ permeation, they have not made any estimates of the ligand binding affinities or rates, which could have been potentially compared to experiment and used to validate their models.
However, despite those relatively minor weaknesses, the conclusions of the study are convincing, and overall this is a solid study helping us to understand two distinct molecular mechanisms of the voltage-gated potassium channel Kv2.1 inhibition by TEA and RY785, respectively.
-

Reviewer #2 (Public review):
Summary
In this manuscript, Zhang et al. investigate the conduction and inhibition mechanisms of the Kv2.1 channel, with a particular focus on the distinct effects of TEA and RY785 on Kv2 potassium channels. Using microsecond-scale molecular dynamics simulations, the authors characterize K⁺ ion permeation and RY785-mediated inhibition within the central pore. Their results reveal an inhibition mechanism that differs from those described for other Kv channel inhibitors.
Strengths
The study identifies a distinctive inhibitory mode for RY785, which binds along the channel walls in the open-state structure while still permitting a reduced level of K⁺ conduction. In addition, the authors propose a long-range allosteric coupling between RY785 binding in the central pore and changes in the structural dynamics of …
Reviewer #2 (Public review):
Summary
In this manuscript, Zhang et al. investigate the conduction and inhibition mechanisms of the Kv2.1 channel, with a particular focus on the distinct effects of TEA and RY785 on Kv2 potassium channels. Using microsecond-scale molecular dynamics simulations, the authors characterize K⁺ ion permeation and RY785-mediated inhibition within the central pore. Their results reveal an inhibition mechanism that differs from those described for other Kv channel inhibitors.
Strengths
The study identifies a distinctive inhibitory mode for RY785, which binds along the channel walls in the open-state structure while still permitting a reduced level of K⁺ conduction. In addition, the authors propose a long-range allosteric coupling between RY785 binding in the central pore and changes in the structural dynamics of Kv2.1. Overall, this is a well-organized and carefully executed study, employing robust simulation and analysis methodologies. The work provides novel mechanistic insights into voltage-gated potassium channel inhibition and may offer useful guidance for future structure-based drug design efforts.
Weaknesses:
The study needs to consider the possibility of multiple binding sites for PY785, particularly given its impact on voltage sensors and gating currents. Specifically, the potential for allosteric binding sites in the voltage-sensing domain (VSD) should be assessed, as some allosteric modulators with thiazole moieties are known to bind VSD domains in multiple voltage-gated sodium channels (Ahuja et al., 2015; Li et al., 2022; McCormack et al., 2013; Mulcahy et al., 2019). Increasing structural and functional evidence supports the existence of multiple ligand-binding modes in voltage-gated ion channels. For example, polyunsaturated fatty acids have been shown to bind to KCNQ1 at both the voltage sensor domain and the pore domain (https://doi.org/10.1085/jgp.202012850). Similarly, cannabidiol has been structurally resolved in Nav1.7 at two distinct sites, one in a fenestration and another near the IFM-binding pocket (https://doi.org/10.1038/s41467-023-39307-6). These advances illustrate that ligand effects cannot always be interpreted based solely on a single binding site identified previously.
-

Author response:
The following is the authors’ response to the original reviews.
eLife Assessment
This study represents an important advance in our understanding of how certain inhibitors affect the behavior of voltage gated potassium channels. Robust molecular dynamics simulation and analysis methods lead to a new proposed inhibition mechanism with strength of support being mostly convincing, and incomplete in some aspects. This study has considerable significance for the fields of ion channel physiology and pharmacology and could aid in development of selective inhibitors for protein targets
We are encouraged by this favorable assessment and thank editors and reviewers for their constructive feedback and recommendations. We trust that the revisions made to the manuscript will clarify the aspects that had been perceived to be …
Author response:
The following is the authors’ response to the original reviews.
eLife Assessment
This study represents an important advance in our understanding of how certain inhibitors affect the behavior of voltage gated potassium channels. Robust molecular dynamics simulation and analysis methods lead to a new proposed inhibition mechanism with strength of support being mostly convincing, and incomplete in some aspects. This study has considerable significance for the fields of ion channel physiology and pharmacology and could aid in development of selective inhibitors for protein targets
We are encouraged by this favorable assessment and thank editors and reviewers for their constructive feedback and recommendations. We trust that the revisions made to the manuscript will clarify the aspects that had been perceived to be incomplete.
Reviewer #1 (Public review):
Summary:
This study seeks to identify a molecular mechanism whereby the small molecule RY785 selectively inhibits Kv2.1 channels. Specifically, it sought to explain some of the functional differences that RY785 exhibits in experimental electrophysiology experiments as compared to other Kv inhibitors, namely the charged and non-specific inhibitor tetraethylammonium (TEA). This study used a recently published cryo-EM Kv2.1 channel structure in the open activated state and performed a series of multi-microsecond-long all-atom molecular dynamics simulations to study Kv2.1 channel conduction under the applied membrane voltage with and without RY785 or TEA present. While TEA directly blocks K+ permeation by occluding ion permeation pathway, RY785 binds to multiple nonpolar residues near the hydrophobic gate of the channel driving it to a semi-closed non-conductive state. This mechanism was confirmed using an additional set of simulations and used to explain experimental electrophysiology data.
Strengths:
The total length of simulation time is impressive, totaling many tens of microseconds. The study develops forcefield parameters for the RY785 molecule based on extensive QM-based parameterization. The computed permeation rate of K+ ions through the channel observed under applied voltage conditions is in reasonable agreement with experimental estimates of the singlechannel conductance. The study performed extensive simulations with the apo channel as well as both TEA and RY785. The simulations with TEA reasonably demonstrate that TEA directly blocks K+ permeation by binding in the center of the Kv2.1 channel cavity, preventing K+ ions from reaching the SCav site. The conclusion is that RY785 likely stabilizes a partially closed conformation of the Kv2.1 channel and thereby inhibits the K+ current. This conclusion is plausible given that RY785 makes stable contact with multiple hydrophobic residues in the S6 helix. This further provides a possible mechanism for the experimental observations that RY785 speeds up the deactivation kinetics of Kv2 channels from a previous experimental electrophysiology study.
Weaknesses:
The study, however, did not produce this semi-closed channel conformation and acknowledges that more direct simulation evidence would require extensive enhanced-sampling simulations. The study has not estimated the effect of RY785 binding on the protein-based hydrophobic pore constriction, which may further substantiate their proposed mechanism. And while the study quantified K+ permeation, it does not make any estimates of the ligand binding affinities or rates, which could have been potentially compared to the experiment and used to validate the models.
As stated in the original manuscript, we concur that the mechanism we propose remains hypothetical until further studies of the complete conformational cycle of the channel are conducted. The recently determined structure of a Kv2.1 channel in the closed state (Mandala and MacKinnon, PNAS 2025) presents an excellent opportunity to do so. Indeed, a cursory analysis of that structure shows that a Pro-Ile-Pro motif in helix S6 marks the position of the intracellular gate, where the pore domain constricts maximally (aside from the selectivity filter). As illustrated in Fig. 5, this motif is precisely where the benzimidazole and thiazole moieties of RY785 bind in our simulations. The mechanism we outline in Fig. 7 thus seems very plausible, in our view; that is RY785 occludes the K+ permeation pathway before the pore domain reaches the closed conformation, explaining the observed electrophysiological effects (see Discussion). The Discussion has been revised to note the recent discovery of the aforementioned structure, its implications for the mechanism we propose, and the opportunities for further research that are now open.
Reviewer #3 (Public review):
Summary:
In this manuscript, Zhang et al. investigate the conductivity and inhibition mechanisms of the Kv2.1 channel, focusing on the distinct effects of TEA and RY785 on Kv2 potassium channels. The study employs microsecond-scale molecular dynamics simulations to characterize K+ ion permeation and compound binding inhibition in the central pore.
Strengths:
The findings reveal a unique inhibition mechanism for RY785, which binds to the channel walls in the open structure while allowing reduced K+ flow. The study also proposes a long-range allosteric coupling between RY785 binding in the central pore and its effects on voltage-sensing domain dynamics. Overall, this well-organized paper presents a high-quality study with robust simulation and analysis methods, offering novel insights into voltage-gated ion channel inhibition that could prove valuable for future drug design efforts.
Weaknesses:
(1) The study neglects to consider the possibility of multiple binding sites for RY785, particularly given its impact on voltage sensors and gating currents. Specifically, there is potential for allosteric binding sites in the voltage-sensing domain (VSD), as some allosteric modulators with thiazole moieties are known to bind VSD domains in multiple voltage-gated sodium channels (Ahuja et al., 2015; Li et al., 2022; McCormack et al., 2013; Mulcahy et al., 2019).
As noted in the manuscript, we designed our simulations to explore the possibility that RY785 binds within the pore domain, because TEA and RY785 are competitive and TEA is known to bind within the pore. That RY785 did in fact spontaneously and reproducibly bind within the pore was however not a predetermined outcome; if the site of interaction for the inhibitor was elsewhere in the channel, the simulation would not have shown a stable associated state, which would have prompted us to examine other possible sites, including the voltage sensors. It was also not predetermined or foreseeable a priori that the mode of interaction we observed in simulation provides a straightforward rationale for the electrophysiological effects of RY785. Based on our results, therefore, we believe that RY785 binds within the pore of Kv2. As stated by the reviewer, other allosteric modulators are known to bind instead to the sensors; to our knowledge, however, there is no precedent of a small-molecule inhibitor that simultaneously acts on the sensors and the pore domain. We therefore believe that future studies should focus on corroborating or refuting the mechanism we propose, through additional experimental and computational work; if, contrary to our claim, RY785 is found not to bind to the pore domain, it would be logical to explore other possible sites of interaction, as the reviewer suggests. The Discussion has been modified to address this point.
(2) The study describes RY785 as a selective inhibitor of Kv2 channels and characterizes its binding residues through MD simulations. However, it is not clear whether the identified RY785-binding residues are indeed unique to Kv2 channels.
To clarify this question, we have included a multiple sequence alignment as Supplementary Figure 1; the revised manuscript refers to this figure in the Discussion section. The alignment reveals that the cluster of residues forming contacts with RY785 (Val409, Pro406, Ile405, Ile401, and Val398) is indeed specific to Kv2.1. Among Kv channels, Kv3.1 and Kv4.1 exhibit the greatest similarity to Kv2.1 at these positions, but they differ in a crucial substitution: Ile405 in Kv2.1 is replaced by Val. This replacement shortens the sidechain, undoubtedly reducing the magnitude of the hydrophobic interaction between inhibitor and channel (Val is approximately 6 kcal/mol, i.e. 1,000 times, more hydrophilic than Ile). Kv5.1 differs from Kv2.1 at two positions: Pro406 is replaced by His, and Val409 by Ile. The introduction of His abolishes the hydrophobic interaction at that position, and the need for hydration likely perturbs all adjacent contacts with RY785. Lastly, Kv6-Kv10 and Cav channels feature entirely different residues at these positions. Consistent with these findings, a recent study by the Sack lab (https://elifesciences.org/articles/99410) has demonstrated that Kv5, Kv6, Kv8, and Kv9 pore subunits confer resistance to RY785, while a high-throughput electrophysiological study carried out by Merck (Herrington et al., 2011) reported that RY785 shows no significant activity against Cav channels. The sequence alignment offers a simple interpretation for these experimental observations, namely that RY785 is recognized by Kv2 channels through the abovementioned hydrophobic cluster within the pore domain.
(3) The study does not clarify the details, rationale, and ramifications of a biasing potential to dihedral angles.
We refer the reviewer to published work, for example Stix et al, 2023 and Tan et al, 2022. We provide additional comments below.
(4) The observation that the Kv2.1 central pore remains partially permeable to K+ ions when RY785 is bound is intriguing, yet it was not revealed whether polar groups of RY785 always interact with K+ ions.
We detected no persistent specific interactions between RY785 and the permeant K+ ions.
Recommendations for the authors:
Reviewer #2 (Recommendations for the authors):
The manuscript describes atomistic molecular dynamics (MD) simulations of a voltage-gated potassium channel Kv2.1 using its cryo-EM structure in the open activated state and its inhibition by a classical non-specific cationic blocker tetraethylammonium (TEA) as well as a novel selective inhibitor RY785. Using multi-microsecond-long all-atom MD runs under the applied membrane voltage of 100 mV the authors were able to confirm that the channel structure represents an open conducting state with the computed single-channel conductance lower than experimental values, but still in the same order of magnitude range. They also determined that both TEA and RY785 bind in the channel pore between the cytoplasmic hydrophobic gate and narrow selectivity filter (SF) region near the extracellular side. However, while TEA directly blocks a knock-on K+ conduction by physically obstructing ion access to the SF, the mechanism of action of RY785 is different. It does not directly prevent K+ access to the SF but rather binds to multiple residues in the hydrophobic gate region, which effectively narrows a pore and drives the channel toward a semi-closed nonconductive conformation, which might be distinct from one with the deactivated voltage sensors and closed pore observed at hyperpolarized membrane potentials. However, additional studies beyond the scope of this work might be needed to fully establish this mechanism as suggested by the authors.
The manuscript is written very well and represents a significant advance in the field of ion channel research. I do not have any major issues, which need to be addressed. However, I have several suggestions.
For the apo-channel K+ conduction MD simulation under the applied voltage, the authors seem to observe mostly a direct or Coulomb knock-on mechanism across the SF with almost no water copermeation. This is in line with computational electrophysiology studies with dual membrane setup by B. de Groot and others but in disagreement with multiple previous studies by B. Roux and others also using applied electric field and CHARMM force fields as in the present study. I wonder why the outcomes are so different. Is it related to the Kv2.1 channel itself, a relatively small applied electric field used (corresponding to a membrane potential of 100 mV vs. 500-750 mV used in many previous simulations), ion force field (e.g., LJ parameters), or some other factors? Could weak dihedral restraints on the protein backbone and side chains contribute to this mechanism? I also wonder if the authors might have considered different initial SF ion configurations. Related to that, I wonder if the authors observed any SF distortions in their simulations including frequently observed backbone carbonyl flipping and/or dilation/contraction.
We are aware of these discrepancies between published simulation studies, but cannot offer a satisfactory explanation, beyond speculation. The reviewer is correct that the mechanism of ion permeation we observe is comparable to that reported by de Groot, as we noted in Tan et al, 2022 and Stix et al, 2023. Neither in this nor in those previous studies did we observe any persistent distortions of the selectivity filter – but that outcome was expected by construction. The weak biasing potentials acting on the mainchain dihedral angles allow for local fluctuations but not a persistent deformation, relative to the conductive form determined experimentally.
For MD simulations with the ligand present, I wonder if the authors can comment on the effect of the ligand especially RY785 on the pore size or more importantly size of the hydrophobic gate. The presence of the ligand itself would definitely result in a narrower pore, but I also wonder if this would also lead to a rearrangement of pore sidechain and/or backbone residues, which would lead to a narrower pore from a protein itself thus confirming the proposed mechanism of driving the channel towards a semi-closed state. It is easy to compute but I wonder if the presence of weak dihedral restraints may preclude this analysis.
Yes, while the simulation design used in this study allows for local fluctuations in the mainchain structure and nearly unrestricted sidechain dynamics, changes in either the secondary or tertiary structure of the channel are strongly disfavored. This approach is thus sufficient to examine ligand binding or ion flow in the microsecond timescale but not channel gating. In the revised version of the Discussion, we outline a roadmap for future computational studies of that gating process, on the basis of the open-channel structure we used and the recently determined structure of the closed state.
The authors state that RY785 does not block K+ ion, but it does significantly slow the rate of K+ ion access to the pore Scav site. Is this not a part of the mechanism for inhibition of the channel? The authors seem to focus on the primary mechanism of inhibition as the RY785 promoting channel closing, but would it not also reduce K+ current in the open state by slowing the rate of K+ entry into the cavity and selectivity filter? The authors should address this point in the text. I am also somewhat confused that in the MD simulations performed by the authors, there is still some K+ conduction with RY785 in the pore, which is not in 100% agreement with electrophysiology experiments. Does it mean that the channel in the simulations has not yet reached that semiclosed state or a reduced K+ conduction is not observed experimentally?
The salient experimental observation is RY785 abrogates K+ currents through Kv2 channels (Herrington et al, 2011; Marquis et al, 2022). In our view, that observation can be explained in one of two ways: either RY785 completely blocks the flow of K+ ions across the channel while the pore domain remains in the conductive, open state – like TEA does – or RY785 induces or facilitates the closing of the channel, thereby abrogating K+ flow. The fact that we observe K+ flow while RY785 is bound to the channel is therefore not in disagreement with the electrophysiological measurements, but it does rule out the first of those two possible interpretations of the existing experiments. As it happens, the second possible explanation, i.e. that RY785 facilitates the closing of the pore domain, also provides a rationale for another puzzling experimental observation, namely that RY785 shifts the voltage dependence of the currents produced by the voltage sensors as they reconfigure to open or close the intracellular gate.
Also, I wonder if the authors considered that since there are 4 potential equivalent sites in the pore (although, overlapping) more than one RY785 might be needed to prevent K+ conduction, even though the experimental Hill coefficient of ~1 does not indicate cooperativity.
Admittedly, our simulation design was based on the premise that only one RY785 molecule might be recognized within the pore. Based on the outcome of the simulations, we are confident that this assumption was valid, as the binding pose that we identified rules out multiple occupancy – which would be indeed consistent with a Hill coefficient of ~1.
I also wonder if the authors considered estimating ligand binding affinities and/or "on" rates from their simulations to have a more direct comparison with experiments and test the accuracy of their models. There are multiple enhanced sampling techniques allowing to do that, although it can be a study on its own.
We thank the reviewer for this suggestion, which we will consider for future studies.
The authors also discussed that they could not study Kv2.1 deactivation in a reasonable simulation time. Indeed it is very challenging but they should cite previous studies e.g. 2012 Jensen et al paper (PMID: 22499946) on this subject. There are structures of Kv channels with the deactivated voltagesensing domains (VSDs) available, e..g of EAG1 channel (PDB 8EP1), although they do not have a domain-swapped architecture. There are structural modeling approaches including AlphaFold, which can be potentially used to get a Kv2.1 structure with deactivated VSDs, and targeted MD, string method etc. can be used to study transition between different states with and without bound ligands.
As noted, a structure of a Kv2 channel with a closed pore has now been determined experimentally. In the revised Discussion, we comment on what this structure tells us about the mechanism of inhibition we propose, and how it could be leveraged in future studies.
The authors should be commended for doing a thorough QM-based force field parameterization of RY785. However, a validation of the developed force field parameters is lacking. In terms of QM validation, a gas-phase dipole moment can be compared in terms of direction and magnitude (it's normal to be overestimated to implicitly reflect solvent-induced polarization). If there are any experimental data available for this compound, they can be tested as well.
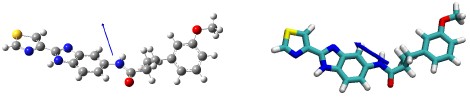
We agree with the reviewer that forcefield validation is important, but to our knowledge no experimental data exists for RY785 to compare with, such as hydration free energies. We did however compare the gas-phase dipole moment computed with QM and with the MM forcefield we developed based on atomic charges optimized to reproduce QM interactions with water. The MM model yields a gas-phase dipole moment of 3.94 D, which is 20% greater than the QM dipole moment, or 3.23 D. That deviation is within the typical range for electroneutral molecules (Vanommeslaeghe et al, 2010), and as the reviewer notes, reflects the solvent-induced polarization implicit in the derivation of atomic charges. As shown in Author response image 1, the orientation of the dipole moment calculated with MM (right, blue arrow) is also in good agreement with that predicted with QM (left)
Author response image 1.
(1) p. 3 "the last two helices in each subunit" -> "the last two transmembrane helices in each subunit".
Thanks. Corrected.
(2) p. 5 "and therefore do not cause large density variations e.g. 100-fold or greater.". I would be more specific here and indicate what are the actual variations in density or free energy encountered and how they are compared e.g. with thermal fluctuations (~kT).
Thanks. The exact variations in K+ density had been included in the original manuscript, in Fig. 2C, but we failed to refer to this figure at this point in the description of the results. The ion density is plotted in a log scale to facilitate conversion to free-energy units. Corrected.
(3) p. 6 Figure 1 caption "and along the perpendicular to the membrane" -> "perpendicular to the membrane normal"?. "The channel is an assembly of four distinct subunits (in colors);" -> "The channel is an assembly of four identical subunits (distinct by colors);". I would use the same protein coloring method in panels B and C as was used in panel A.
Thanks. Corrected as needed.
(4) p. 6 Figure 2 In panel B I would appreciate a representative complete ion permeation event trace. In panel C caption I would indicate corresponding sites "S0-S4, Scav" for each residue mentioned. I also would not use gray color for site names in the figure.
We appreciate the suggestion, but believe the figure is clear as is. Panel B is meant to focused on the mechanism of knock-on. Panel A includes numerous complete permeation events.
(5) p. 7 Figure 3 caption. Please indicate which atoms of residues T373 and P406 were used to define SF and gate positions. Chemical structures of both TEA and RY785 would be useful. In panels C and F channel interacting residues (if any) would be helpful to show.
The revised caption clarifies that the positions of T373 and P406 are represented by their carbonalpha atoms. A close-up view of the structures of TEA and RY785 is included in the Supplementary Information section.
(6) p. 8. Figure 4 caption. Please indicate if N atoms ere used for density maps in panels B and C, and which value of the density was used to show meshes. In panel A please indicate what are the units of the density shown by color maps.
The caption has been revised to clarify these questions.
(7) p. 9 "inside the protein" -> "inside the channel pore".
Thanks. Corrected.
(8) p. 10 "which lines the cavity" -> "which lines the water-filled cavity"
We appreciate the suggestion but believe the wording is clear as is.
(9) p.10 Fig. 5. It would be helpful to distinguish residues from different chains e.g. by different colors rather than using different colors for different residues. The S atom in RY785 is hard to recognize due to the yellow color used for C atoms. Figure 5B is very confusing. It is not clear what this plot represents. For instance, what does it mean that Pro405 has ~10 contacts in 20% of simulation snapshots? Does it mean 10 C..C/S interactions within 4.5 A? I am not sure what the value of this is. I think a bar or radar chart plot showing % of contacts with one, two, or more residues of each type would be more helpful.
Thanks. The revised caption ought to clarify how to interpret the plot.
(10) p. 12 "Due to its 2-fold molecular symmetry". TEA has a tetrahedral point group or Td symmetry. It has several two-fold rotational axes though.
Thanks. Corrected.
(11) p. 12 "it prevents K+ ions in the cytoplasmic space from destabilizing the K+ ions that reside in the selectivity filter" I am not sure if this statement is entirely accurate as there might be destabilization of a multi-ion SF configuration not ions per see.
We believe this statement is clear as is.
(12) p. 13 Fig. 7 caption "includes non-conductive or transiently inactivated states" - I am not sure what "transiently inactivated state" is as inactivation is a specific term used in ion channel research and it does not seem to be explicitly considered in this study.
A reference has been included in the caption for readers interested in the process of inactivation.
(13) p. 14 "the net charge of these constructs is thus zero". This would depend on the number of basic and acidic residues in the protein.
Yes, it does – and as a result the construct we model has a net zero charge.
(14) p. 14 I wonder if the protein was constrained or heavily restrained during MARTINI membrane building and equilibration procedure. Otherwise, C-alpha mapping would be problematic and clashes with lipid membrane atoms might take place as well.
It was indeed. When a protein is simulated using the MARTINI coarse-grained forcefield, its fold must be preserved through a network of strong ‘virtual’ bonds between adjacent carbon-alpha atoms. This is standard practice so we do not believe it requires further explanation.
(15) p. 15 PME - please spell out and provide reference.
Corrected.
(16) p. 15 "with a smooth switching function" - is it a special or standard switching function? Also, was it used for energy or forces?
The switching function brings both forces and energies to a value of zero at the cut-off value, smoothly. We refer the reviewer to the NAMD manual for further details.
(17) p. 15 '𝑘 = 1 𝑘B𝑇.' Please confirm that there is a factor of "1" there, which can be actually skipped if this is the case.
The value of k = 1 KBT is correct.
(18) p. 15. Please cite PMID: 22001851 for the transmembrane electric field application technique.
Corrected.
(19) p. 15 "and CHARMM36m" -> "and CHARMM36m force field".
Corrected.
(20) p. 16 "the four proteins subunits" -> "the four protein subunits".
Corrected.
(21) p. 16. Please provide the reference for CGenFF. It's reference 49.
Corrected.
Supporting Information (SI): CGenFF is misspelled in multiple figure captions in the SI. All potential energy scans indicate "angle", but some are bond angles while others are dihedral angles. Using subscripts for atom numbers is confusing and does not match the numbering scheme used in Fig. S1. So, please use the same style of numbering throughout, e.g. C46-C42-N43 (without subscripts). Please label the X and Y axes in Figsures S2-S19 and S21. In Figure S22 please perform a linear regression analysis and/or compute Pearson correlation coefficients and indicate trend lines. Table S1. It would be good to compute RMS or mean unsigned errors to get an idea about accuracy. Also, please indicate if reference QM values were scaled by 1.16 for energies or offset for distances.
The Supplementary Information has been corrected. We thank the reviewer for their detailed feedback.
Reviewer #3 (Recommendations for the authors):
(1) The study needs to consider the possibility of multiple binding sites for RY785, particularly given its impact on voltage sensors and gating currents. Specifically, the potential for allosteric binding sites in the voltage-sensing domain (VSD) should be assessed, as some allosteric modulators with thiazole moieties are known to bind VSD domains in multiple voltage-gated sodium channels (Ahuja et al., 2015; Li et al., 2022; McCormack et al., 2013; Mulcahy et al., 2019). Molecular docking and/or MD simulations could quickly test this hypothesis. If this hypothesis is not true, a comprehensive search can exclude such a possibility, which can also confirm the long-range allosteric coupling between RY785 binding in the central pore and voltage-sensing domain dynamics.
Please see our response above.
(2) The authors describe RY785 as a selective inhibitor of Kv2 channels and characterize its binding residues through MD simulations. To support this claim, Figure 5 needs to include a multiple sequence alignment with other Kv channels. This would help demonstrate whether the identified RY785-binding residues are indeed unique to Kv2 channels.
Please see our response above.
(3) The study applies a biasing potential to 𝜙, 𝜓, and 𝜒1 dihedral angles. Please clarify:
(a) Is this potential solely to prevent selectivity filter collapse/degradation, as mentioned in a previous D. E. Shaw Research publication (Jensen et al., 2012)?
Yes, that is correct.
(b) If it applies to all amino acids, can this potential prevent other changes, such as in the voltagesensing domain?
Yes, that is correct.
(c) What specific "large-scale structural changes" does this potential preclude?
For example, it would preclude the spontaneous degradation of the secondary or tertiary structure of the protein. We have revised the Methods section to make these points clearer.
(d) Given that such biasing potentials on backbone dihedral angles can decrease conformational flexibility, and considering that Kv channel permeability/conductivity could be highly sensitive to filter flexibility, what insights can you provide about the impact of the force constant k on channel conductivity?
In previous studies based on an identical methodology (Stix et al, 2023; Tan et al, 2022), we have observed good agreement between calculated and experimental conductance values – at least as good as can be hoped for, when all approximations are considered. Based on the data presented in those studies, we have no reason to believe our methodology inhibits the permeability of the channel, which is logical as the local structural fluctuations required for K+ flow across the selectivity filter are not impaired, by definition. To the contrary, the fact that these weak biasing potentials make the conductive form of the filter the most favorable state in simulation enable a clear-cut analysis of conductance under plausible simulation conditions, both in terms applied voltage and K+ concentration. We refer the reviewer to the abovementioned studies for further details and a discussion of this subject.
(4) The observation that the Kv2.1 central pore remains partially permeable to K+ ions when RY785 is bound is intriguing. Given the compact nature of the central cavity when RY785 is bound, it would be valuable to investigate whether polar groups of RY785 (e.g., nitrogens from the amide, benzimidazole, and thiazole moieties) always interact with K+ ions. Characterizing these interactions could inform the design of similar compounds with differential modulation effects.
We examined this possibility and detected no convincing interaction patterns between RY785 and K+ ions – logically, inhibitor and ions are in close proximity while residing concurrently within the pore, but we detected no evidence of specific interactions.
Minor points:
It is strongly recommended that the refined force field parameters for RY785 be shared as a separate supplementary file in CHARMM force field format. This addition would be valuable for the scientific community, allowing other researchers to use or compare these parameters in future studies.
We agree entirely. Upon publication of the VOR for this article the forcefield parameters for RY785 will be made freely available for download at https://github.com/Faraldo-Gomez-Lab-atNIH/Download.
The study uses a KCl concentration of 300 mM, which exceeds typical intracellular K+ levels. While this may be intentional to enhance K+ permeation probability, a brief justification for this choice should be included in the Methods section.
Yes, what motivated this choice in this and in our previous studies of K+ channels was the expectation of a greater number of permeation events, for a given simulation length, and therefore greater confidence (i.e. statistical significance) in the observed ion conductance, or in the degree to which it might be inhibited by a blocker. It worth noting that 300 mM KCl, while atypical in the intracellular environment, is often used in electrophysiological studies. The Methods section has been amended to clarify this point.
-

-

-

eLife Assessment
This study represents an important advance in our understanding of how certain inhibitors affect the behavior of voltage gated potassium channels. Robust molecular dynamics simulation and analysis methods lead to a new proposed inhibition mechanism with strength of support being mostly convincing, and incomplete in some aspects. This study has considerable significance for the fields of ion channel physiology and pharmacology and could aid in development of selective inhibitors for protein targets.
-

Reviewer #1 (Public review):
Summary:
This study seeks to identify a molecular mechanism whereby the small molecule RY785 selectively inhibits Kv2.1 channels. Specifically, it sought to explain some of the functional differences that RY785 exhibits in experimental electrophysiology experiments as compared to other Kv inhibitors, namely the charged and non-specific inhibitor tetraethylammonium (TEA). This study used a recently published cryo-EM Kv2.1 channel structure in the open activated state and performed a series of multi-microsecond-long all-atom molecular dynamics simulations to study Kv2.1 channel conduction under the applied membrane voltage with and without RY785 or TEA present. While TEA directly blocks K+ permeation by occluding ion permeation pathway, RY785 binds to multiple non-polar residues near the hydrophobic gate of …
Reviewer #1 (Public review):
Summary:
This study seeks to identify a molecular mechanism whereby the small molecule RY785 selectively inhibits Kv2.1 channels. Specifically, it sought to explain some of the functional differences that RY785 exhibits in experimental electrophysiology experiments as compared to other Kv inhibitors, namely the charged and non-specific inhibitor tetraethylammonium (TEA). This study used a recently published cryo-EM Kv2.1 channel structure in the open activated state and performed a series of multi-microsecond-long all-atom molecular dynamics simulations to study Kv2.1 channel conduction under the applied membrane voltage with and without RY785 or TEA present. While TEA directly blocks K+ permeation by occluding ion permeation pathway, RY785 binds to multiple non-polar residues near the hydrophobic gate of the channel driving it to a semi-closed non-conductive state. This mechanism was confirmed using an additional set of simulations and used to explain experimental electrophysiology data,
Strengths:
The total length of simulation time is impressive, totaling many tens of microseconds. The study develops forcefield parameters for the RY785 molecule based on extensive QM-based parameterization. The computed permeation rate of K+ ions through the channel observed under applied voltage conditions is in reasonable agreement with experimental estimates of the single-channel conductance. The study performed extensive simulations with the apo channel as well as both TEA and RY785. The simulations with TEA reasonably demonstrate that TEA directly blocks K+ permeation by binding in the center of the Kv2.1 channel cavity, preventing K+ ions from reaching the SCav site. The conclusion is that RY785 likely stabilizes a partially closed conformation of the Kv2.1 channel and thereby inhibits the K+ current. This conclusion is plausible given that RY785 makes stable contact with multiple hydrophobic residues in the S6 helix. This further provides a possible mechanism for the experimental observations that RY785 speeds up the deactivation kinetics of Kv2 channels from a previous experimental electrophysiology study.
Weaknesses:
The study, however, did not produce this semi-closed channel conformation and acknowledges that more direct simulation evidence would require extensive enhanced-sampling simulations. The study has not estimated the effect of RY785 binding on the protein-based hydrophobic pore constriction, which may further substantiate their proposed mechanism. And while the study quantified K+ permeation, it does not make any estimates of the ligand binding affinities or rates, which could have been potentially compared to the experiment and used to validate the models.
-

Reviewer #3 (Public review):
Summary:
In this manuscript, Zhang et al. investigate the conductivity and inhibition mechanisms of the Kv2.1 channel, focusing on the distinct effects of TEA and RY785 on Kv2 potassium channels. The study employs microsecond-scale molecular dynamics simulations to characterize K+ ion permeation and compound binding inhibition in the central pore.
Strengths:
The findings reveal a unique inhibition mechanism for RY785, which binds to the channel walls in the open structure while allowing reduced K+ flow. The study also proposes a long-range allosteric coupling between RY785 binding in the central pore and its effects on voltage-sensing domain dynamics. Overall, this well-organized paper presents a high-quality study with robust simulation and analysis methods, offering novel insights into voltage-gated ion …
Reviewer #3 (Public review):
Summary:
In this manuscript, Zhang et al. investigate the conductivity and inhibition mechanisms of the Kv2.1 channel, focusing on the distinct effects of TEA and RY785 on Kv2 potassium channels. The study employs microsecond-scale molecular dynamics simulations to characterize K+ ion permeation and compound binding inhibition in the central pore.
Strengths:
The findings reveal a unique inhibition mechanism for RY785, which binds to the channel walls in the open structure while allowing reduced K+ flow. The study also proposes a long-range allosteric coupling between RY785 binding in the central pore and its effects on voltage-sensing domain dynamics. Overall, this well-organized paper presents a high-quality study with robust simulation and analysis methods, offering novel insights into voltage-gated ion channel inhibition that could prove valuable for future drug design efforts.
Weaknesses:
(1) The study neglects to consider the possibility of multiple binding sites for RY785, particularly given its impact on voltage sensors and gating currents. Specifically, there is potential for allosteric binding sites in the voltage-sensing domain (VSD), as some allosteric modulators with thiazole moieties are known to bind VSD domains in multiple voltage-gated sodium channels (Ahuja et al., 2015; Li et al., 2022; McCormack et al., 2013; Mulcahy et al., 2019).
(2) The study describes RY785 as a selective inhibitor of Kv2 channels and characterizes its binding residues through MD simulations. However, it is not clear whether the identified RY785-binding residues are indeed unique to Kv2 channels.
(3) The study does not clarify the details, rationale, and ramifications of a biasing potential to dihedral angles.
(4) The observation that the Kv2.1 central pore remains partially permeable to K+ ions when RY785 is bound is intriguing, yet it was not revealed whether polar groups of RY785 always interact with K+ ions.
-

-