Structural and dynamic impacts of single-atom disruptions to guide RNA interactions within the recognition lobe of Geobacillus stearothermophilus Cas9
Curation statements for this article:-
Curated by eLife
eLife Assessment
This study offers valuable insights into the conformational dynamics of the nucleic acid recognition lobe of GeoCas9, a thermophilic Cas9 from Geobacillus stearothermophilus. The authors investigate the influence of local dynamics and allosteric regulation on guide RNA binding affinity and DNA cleavage specificity through molecular dynamics simulations, advanced NMR techniques, RNA binding studies, and mutagenesis. While the mutations studied do not lead to significant changes in GeoCas9 cleavage activity, the study provides convincing evidence for the role of allosteric mechanisms and interdomain communication in Cas9 enzymes, and will be of great interest to biochemists and biophysicists exploring these complex systems.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
The intuitive manipulation of specific amino acids to alter the activity or specificity of CRISPR-Cas9 has been a topic of great interest. As a large multi-domain RNA-guided endonuclease, the intricate molecular crosstalk within the Cas9 protein hinges on its conformational dynamics, but a comprehensive understanding of the extent and timescale of the motions that drive its allosteric function and association with nucleic acids remains elusive. Here, we investigated the structure and multi-timescale molecular motions of the recognition (Rec) lobe of Geo Cas9, a thermophilic Cas9 from Geobacillus stearothermophilus . Our results provide new atomic details about the Geo Rec subdomains ( Geo Rec1, Geo Rec2) and the full-length domain in solution. Two rationally designed mutants, K267E and R332A, enhanced and redistributed micro-millisecond flexibility throughout Geo Rec, and NMR studies of the interaction between Geo Rec and its guide RNA showed that mutations reduced this affinity and the stability of the ribonucleoprotein complex. Despite measured biophysical differences due to the mutations, DNA cleavage assays reveal no functional differences in on-target activity, and similar specificity. These data suggest that guide RNA interactions can be tuned at the biophysical level in the absence of major functional losses but also raise questions about the underlying mechanism of Geo Cas9, since analogous single-point mutations have significantly impacted on- and off-target DNA editing in mesophilic Streptococcus pyogenes Cas9. A K267E/R332A double mutant did also did not enhance Geo Cas9 specificity, highlighting the robust tolerance of mutations to the Rec lobe of Geo Cas9 and species-dependent complexity of Rec across Cas9 paralogs. Ultimately, this work provides an avenue by which to modulate the structure, motion, and guide RNA interactions at the level of the Rec lobe of Geo Cas9, setting the stage for future studies of Geo Cas9 variants and their effect on its allosteric mechanism.
Article activity feed
-

-

-

-

eLife Assessment
This study offers valuable insights into the conformational dynamics of the nucleic acid recognition lobe of GeoCas9, a thermophilic Cas9 from Geobacillus stearothermophilus. The authors investigate the influence of local dynamics and allosteric regulation on guide RNA binding affinity and DNA cleavage specificity through molecular dynamics simulations, advanced NMR techniques, RNA binding studies, and mutagenesis. While the mutations studied do not lead to significant changes in GeoCas9 cleavage activity, the study provides convincing evidence for the role of allosteric mechanisms and interdomain communication in Cas9 enzymes, and will be of great interest to biochemists and biophysicists exploring these complex systems.
-

Reviewer #1 (Public review):
Summary:
In this study from Belato, Knight and co-workers, the authors investigated the Rec domain of a thermophilic Cas9 from Geobacillus stearothermophilus (GeoCas9). The authors investigated three constructs, two individual subdomains of Rec (Rec1 and Rec2) and the full Rec domain. This domain is involved in binding to the guide RNA of Cas9, as well as the RNA-DNA duplex that is formed upon target binding. The authors performed RNA binding and relaxation experiments using NMR for the wild-type domain as well as two-point mutants. They observed differences in RNA binding activities as well as the flexibility of the domain. The authors also performed molecular dynamics and functional experiments on full-length GeoCas9 to determine whether these biophysical differences affect the RNA binding or cleavage …
Reviewer #1 (Public review):
Summary:
In this study from Belato, Knight and co-workers, the authors investigated the Rec domain of a thermophilic Cas9 from Geobacillus stearothermophilus (GeoCas9). The authors investigated three constructs, two individual subdomains of Rec (Rec1 and Rec2) and the full Rec domain. This domain is involved in binding to the guide RNA of Cas9, as well as the RNA-DNA duplex that is formed upon target binding. The authors performed RNA binding and relaxation experiments using NMR for the wild-type domain as well as two-point mutants. They observed differences in RNA binding activities as well as the flexibility of the domain. The authors also performed molecular dynamics and functional experiments on full-length GeoCas9 to determine whether these biophysical differences affect the RNA binding or cleavage activity. Although the authors observed some changes in the thermal stability of the mutant GeoCas9-gRNA complex, they did not observe substantial differences in the guide RNA binding or cleavage activities of the mutant GeoCas9 variants.
Overall, this manuscript provides a detailed biophysical analysis of the GeoCas9 Rec domain. The NMR assignments for this construct should prove very useful, and can serve as the basis for future similar studies of GeoCas9 Rec domain mutants. While the two mutants tested in the study did not produce significant differences from wild-type GeoCas9, the study rules out the possibility that analogous mutations can be translated between type II-A and II-C Cas9 orthologs. Together, these findings may provide the grounds for future engineering of higher fidelity variants of GeoCas9
-

Reviewer #2 (Public review):
The manuscript from Belato et al., used advanced NMR approaches and a mutagenesis campaign probe the conformational dynamics of the recognition lobe (Rec) of the CRISPR Cas9 enzyme from G. stearothermophilus (GeoCas9). Using truncated and full-length constructs they assess the impacts of two different point mutations have on the redistribution and timescale of these motions and assess gRNA recognition and specificity. Single point mutations in the Rec domain in a Cas9 from a related species had profound impacts on- and off-target DNA editing, therefore the authors reasoned analogous mutations in GeoCas9 would have similar effects. However, despite a redistribution of local motions and changes in global stability, their chosen mutations had little impact on DNA editing in the context of the full-length enzyme.
Reviewer #2 (Public review):
The manuscript from Belato et al., used advanced NMR approaches and a mutagenesis campaign probe the conformational dynamics of the recognition lobe (Rec) of the CRISPR Cas9 enzyme from G. stearothermophilus (GeoCas9). Using truncated and full-length constructs they assess the impacts of two different point mutations have on the redistribution and timescale of these motions and assess gRNA recognition and specificity. Single point mutations in the Rec domain in a Cas9 from a related species had profound impacts on- and off-target DNA editing, therefore the authors reasoned analogous mutations in GeoCas9 would have similar effects. However, despite a redistribution of local motions and changes in global stability, their chosen mutations had little impact on DNA editing in the context of the full-length enzyme.
In their revised manuscript, the authors were highly responsive to the reviewer's comments incorporating new experimental results including molecular dynamics simulations and RNA binding data using full-length GeoCas9, as well as reframing their discussion and conclusions in consideration of the new data. They were receptive to suggestions for clarification in both the text and methods section. With these changes, the manuscript has been significantly improved.
Their studies highlight the species-specific complexity of interdomain communication and allosteric mechanisms used by these multi-domain endonucleases. The noted strengths of the article remain, and despite the negative results, their approach will garner interest from investigators interested in understanding how the activity and specificity of these enzymes can be engineered to tune activity and limit off-target cleavage by these enzymes. Generally, the manuscript highlights the challenges of studying the effect of allosteric networks on protein function, particularly in multidomain proteins, and thus will be of broad interest to the community.
-

Reviewer #3 (Public review):
The authors explore the role of Rec domains in a thermophilic Cas9 enzyme. They report on the crystal structure of part of the recognition lobe, its dynamics from NMR spin relaxation and relaxation-dispersion data, its interaction mode with guide RNA, and the effect of two single-point mutations hypothesised to enhance specificity. They find that mutations have small effects on Rec domain structure and stability but lead to significant rearrangement of micro- to milli-second dynamics which does not translate into major changes in guide RNA affinity or DNA cleavage specificity, illustrating the inherent tolerance of GeoCas9. The work can be considered as a first step towards understanding motions in GeoCas9 recognition lobe, although no clear hotspots were discovered with potential for future rational design …
Reviewer #3 (Public review):
The authors explore the role of Rec domains in a thermophilic Cas9 enzyme. They report on the crystal structure of part of the recognition lobe, its dynamics from NMR spin relaxation and relaxation-dispersion data, its interaction mode with guide RNA, and the effect of two single-point mutations hypothesised to enhance specificity. They find that mutations have small effects on Rec domain structure and stability but lead to significant rearrangement of micro- to milli-second dynamics which does not translate into major changes in guide RNA affinity or DNA cleavage specificity, illustrating the inherent tolerance of GeoCas9. The work can be considered as a first step towards understanding motions in GeoCas9 recognition lobe, although no clear hotspots were discovered with potential for future rational design of enhanced Cas9 variants.
Strengths:
- Detailed biophysical and structural investigation, despite a few technical limitations inherent with working with complex targets, provides converging evidence that molecular dynamics embedded in the recognition lobes allow GeoCas9 to operate on a broad range of substrates.
- Since the authors and others have shown that substrate specificity is dictated by equivalent hotspot mutations in other Cas9 variants, we are one step closer to understanding this phenomenon.Weaknesses:
- Since the mutations investigated here do not significantly affect substrate binding or enzymatic activity, it is difficult to rationalize anything for enzyme engineering at this point.
- Further investigation of the determinants of the observed dynamic modes, and follow-up with rationally designed mutations would hopefully allow to create a real model of the mechanism, but I do understand that this goes beyond the scope of this study. -

Author response:
The following is the authors’ response to the previous reviews
Responses to final minor critiques following initial revision
Reviewer #1 (Recommendations for the authors):
The authors have generally done an excellent job of addressing my and the other reviewers' concerns. I have a few additional concerns that the authors could consider addressing through changes to the text:
We thank the Reviewer for this assessment and are glad to have addressed the major points.
- Regarding the gRNA used for NMR studies, I thank the authors for adding additional rationale for their design of the RNA used. However, I still believe that it is misleading to term this RNA as a "gRNA", given that it is mainly composed of a sequence that is arbitrary (the spacer) and the sections of the gRNA that are constant between all gRNAs are …
Author response:
The following is the authors’ response to the previous reviews
Responses to final minor critiques following initial revision
Reviewer #1 (Recommendations for the authors):
The authors have generally done an excellent job of addressing my and the other reviewers' concerns. I have a few additional concerns that the authors could consider addressing through changes to the text:
We thank the Reviewer for this assessment and are glad to have addressed the major points.
- Regarding the gRNA used for NMR studies, I thank the authors for adding additional rationale for their design of the RNA used. However, I still believe that it is misleading to term this RNA as a "gRNA", given that it is mainly composed of a sequence that is arbitrary (the spacer) and the sections of the gRNA that are constant between all gRNAs are truncated in a way that removes secondary structure that is likely essential for specific contacts with the Rec domains. I do not believe the authors need to make alterations to any of their experiments. However, I do think their description of the "gRNA" should be updated to properly reflect that this RNA lacks any of the secondary structure present in a typical gRNA, much of which is necessary to confer specificity of binding between GeoCas9 and the gRNA. As mentioned in my previous review, this may be best achieved by adding a cartoon of the secondary structure of the full-length gRNA and highlighting the region that was used in the truncated "gRNA".
We understand the Reviewer’s point. For any experiment in which the gRNA was truncated (i.e. NMR or some MST studies), we have clarified the text and no longer call it a “gRNA.” We state initially that it is a portion of the gRNA and then call it simply an “RNA.”
For experiments using the full-length constructs, we have kept the term “gRNA,” as it remains appropriate.
We have also added a final Supplementary figure (S12) showing the structures of the truncated and full-length RNAs used, based on the _Geo_Cas9 cryo-EM structure and predicted with RNAfold.
- Lines 256-257: "The ~3-fold decrease in Kd...". I believe the authors are discussing the Kd's of the mutants relative to WT, in which case the Kd increased. Also, the fold-change appears closer to 2fold than to 3-fold.
Yes, the Reviewer makes a good catch. We have corrected this.
- Lines 407-408: "The mutations also diminished the stability of the full-length GeoCas9 RNP complex." This statement seems at odds with the authors' conclusions in the Results section that the full-length GeoCas9 variants had comparable affinities for the gRNAs (lines 376-382)
We agree that this seems contradictory. In the absence of full-length structures for all variants, we can’t definitively state what causes this. It could be that the mutation has an interesting allosteric effect on structure that does not affect RNA binding but induces the Cas9 protein to simply fall apart at lower temperatures, rendering the binding interaction moot. We have added a statement to this section.
- The authors chose to keep "SpCas9" for consistency with their prior work and the work of many several others, including Doudna et al and Zhang et al. However, I will note that their publications on GeoCas9, the Doudna lab did use SpyCas9 to ensure consistent nomenclature within the publications.
We have made the change to “_Spy_Cas9”
Reviewer #3 (Recommendations for the authors):
The authors clearly answered most of my concerns. I still have some technical questions about the analysis of CPMG-RD data but the numbers provided now seem to make sense. While I still think that crystal structures of the point mutant would make the conclusions more "bullet proof", I do appreciate the work associated with this and consider that the manuscript can be published as is.
We agree that additional magnetic fields could allow for additional models of CPMG data fitting and that additional crystal structures of the mutants could add to the conclusions. We appreciate the Reviewer recognizing the balance of the current results and potential future studies in signing off on publication.
-

-

eLife Assessment
This study offers valuable insights into the conformational dynamics of the nucleic acid recognition lobe of GeoCas9, a thermophilic Cas9 from Geobacillus stearothermophilus. The authors investigate the influence of local dynamics and allosteric regulation on guide RNA binding affinity and DNA cleavage specificity through advanced NMR techniques and mutagenesis. The revised manuscript incorporates new experimental data, including molecular dynamics simulations and additional RNA binding studies, which provide convincing support for the findings. While the mutations studied do not lead to significant changes in GeoCas9 cleavage activity, the study contributes to a better understanding of the allosteric mechanisms and interdomain communication in Cas9 enzymes, and will be of great interest to biochemists and biophysicists …
eLife Assessment
This study offers valuable insights into the conformational dynamics of the nucleic acid recognition lobe of GeoCas9, a thermophilic Cas9 from Geobacillus stearothermophilus. The authors investigate the influence of local dynamics and allosteric regulation on guide RNA binding affinity and DNA cleavage specificity through advanced NMR techniques and mutagenesis. The revised manuscript incorporates new experimental data, including molecular dynamics simulations and additional RNA binding studies, which provide convincing support for the findings. While the mutations studied do not lead to significant changes in GeoCas9 cleavage activity, the study contributes to a better understanding of the allosteric mechanisms and interdomain communication in Cas9 enzymes, and will be of great interest to biochemists and biophysicists exploring these complex systems.
-

Reviewer #1 (Public review):
Summary:
In this study from Belato, Knight and co-workers, the authors investigated the Rec domain of a thermophilic Cas9 from Geobacillus stearothermophilus (GeoCas9). The authors investigated three constructs, two individual subdomains of Rec (Rec1 and Rec2) and the full Rec domain. This domain is involved in binding to the guide RNA of Cas9, as well as the RNA-DNA duplex that is formed upon target binding. The authors performed RNA binding and relaxation experiments using NMR for the wild-type domain as well as two-point mutants. They observed differences in RNA binding activities as well as the flexibility of the domain. The authors also performed molecular dynamics and functional experiments on full-length GeoCas9 to determine whether these biophysical differences affect the RNA binding or cleavage …
Reviewer #1 (Public review):
Summary:
In this study from Belato, Knight and co-workers, the authors investigated the Rec domain of a thermophilic Cas9 from Geobacillus stearothermophilus (GeoCas9). The authors investigated three constructs, two individual subdomains of Rec (Rec1 and Rec2) and the full Rec domain. This domain is involved in binding to the guide RNA of Cas9, as well as the RNA-DNA duplex that is formed upon target binding. The authors performed RNA binding and relaxation experiments using NMR for the wild-type domain as well as two-point mutants. They observed differences in RNA binding activities as well as the flexibility of the domain. The authors also performed molecular dynamics and functional experiments on full-length GeoCas9 to determine whether these biophysical differences affect the RNA binding or cleavage activity. Although the authors observed some changes in the thermal stability of the mutant GeoCas9-gRNA complex, they did not observe substantial differences in the guide RNA binding or cleavage activities of the mutant GeoCas9 variants.
Overall, this manuscript provides a detailed biophysical analysis of the GeoCas9 Rec domain. The NMR assignments for this construct should prove very useful, and can serve as the basis for future similar studies of GeoCas9 Rec domain mutants. While the two mutants tested in the study did not produce significant differences from wild-type GeoCas9, the study rules out the possibility that analogous mutations can be translated between type II-A and II-C Cas9 orthologs. Together, these findings may provide the grounds for future engineering of higher fidelity variants of GeoCas9
-

Reviewer #2 (Public review):
The manuscript from Belato et al., used advanced NMR approaches and a mutagenesis campaign probe the conformational dynamics of the recognition lobe (Rec) of the CRISPR Cas9 enzyme from G. stearothermophilus (GeoCas9). Using truncated and full-length constructs they assess the impacts of two different point mutations have on the redistribution and timescale of these motions and assess gRNA recognition and specificity. Single point mutations in the Rec domain in a Cas9 from a related species had profound impacts on- and off-target DNA editing, therefore the authors reasoned analogous mutations in GeoCas9 would have similar effects. However, despite a redistribution of local motions and changes in global stability, their chosen mutations had little impact on DNA editing in the context of the full-length enzyme.
Reviewer #2 (Public review):
The manuscript from Belato et al., used advanced NMR approaches and a mutagenesis campaign probe the conformational dynamics of the recognition lobe (Rec) of the CRISPR Cas9 enzyme from G. stearothermophilus (GeoCas9). Using truncated and full-length constructs they assess the impacts of two different point mutations have on the redistribution and timescale of these motions and assess gRNA recognition and specificity. Single point mutations in the Rec domain in a Cas9 from a related species had profound impacts on- and off-target DNA editing, therefore the authors reasoned analogous mutations in GeoCas9 would have similar effects. However, despite a redistribution of local motions and changes in global stability, their chosen mutations had little impact on DNA editing in the context of the full-length enzyme.
In their revised manuscript, the authors were highly responsive to the reviewer's comments incorporating new experimental results including molecular dynamics simulations and RNA binding data using full-length GeoCas9, as well as reframing their discussion and conclusions in consideration of the new data. They were receptive to suggestions for clarification in both the text and methods section. With these changes, the manuscript has been significantly improved.
Their studies highlight the species-specific complexity of interdomain communication and allosteric mechanisms used by these multi-domain endonucleases. The noted strengths of the article remain, and despite the negative results, their approach will garner interest from investigators interested in understanding how the activity and specificity of these enzymes can be engineered to tune activity and limit off-target cleavage by these enzymes. Generally, the manuscript highlights the challenges of studying the effect of allosteric networks on protein function, particularly in multidomain proteins, and thus will be of broad interest to the community.
-

Reviewer #3 (Public review):
The authors explore the role of Rec domains in a thermophilic Cas9 enzyme. They report on the crystal structure of part of the recognition lobe, its dynamics from NMR spin relaxation and relaxation-dispersion data, its interaction mode with guide RNA, and the effect of two single-point mutations hypothesised to enhance specificity. They find that mutations have small effects on Rec domain structure and stability but lead to significant rearrangement of micro- to milli-second dynamics which does not translate into major changes in guide RNA affinity or DNA cleavage specificity, illustrating the inherent tolerance of GeoCas9. The work can be considered as a first step towards understanding motions in GeoCas9 recognition lobe, although no clear hotspots were discovered with potential for future rational design …
Reviewer #3 (Public review):
The authors explore the role of Rec domains in a thermophilic Cas9 enzyme. They report on the crystal structure of part of the recognition lobe, its dynamics from NMR spin relaxation and relaxation-dispersion data, its interaction mode with guide RNA, and the effect of two single-point mutations hypothesised to enhance specificity. They find that mutations have small effects on Rec domain structure and stability but lead to significant rearrangement of micro- to milli-second dynamics which does not translate into major changes in guide RNA affinity or DNA cleavage specificity, illustrating the inherent tolerance of GeoCas9. The work can be considered as a first step towards understanding motions in GeoCas9 recognition lobe, although no clear hotspots were discovered with potential for future rational design of enhanced Cas9 variants.
Strengths:
- Detailed biophysical and structural investigation, despite a few technical limitations inherent with working with complex targets, provides converging evidence that molecular dynamics embedded in the recognition lobes allow GeoCas9 to operate on a broad range of substrates.
- Since the authors and others have shown that substrate specificity is dictated by equivalent hotspot mutations in other Cas9 variants, we are one step closer to understanding this phenomenon.Weaknesses:
- Since the mutations investigated here do not significantly affect substrate binding or enzymatic activity, it is difficult to rationalize anything for enzyme engineering at this point.
- Further investigation of the determinants of the observed dynamic modes, and follow-up with rationally designed mutations would hopefully allow to create a real model of the mechanism, but I do understand that this goes beyond the scope of this study. -

Author response:
The following is the authors’ response to the original reviews
Public Reviews:
Reviewer #1 (Public Review):
Summary:
In this study from Belato, Knight, and co-workers, the authors investigated the Rec domain of a thermophilic Cas9 from Geobacillus stearothermophilus (GeoCas9). The authors investigated three constructs, two individual subdomains of Rec (Rec1 and Rec2) and the full Rec domain. This domain is involved in binding to the guide RNA of Cas9, as well as the RNA-DNA duplex that is formed upon target binding. The authors performed RNA binding and relaxation experiments using NMR for the wild-type domain as well as two-point mutants. They observed differences in RNA binding activities as well as the flexibility of the domain. The authors also performed experiments on fulllength GeoCas9 to determine whether these …
Author response:
The following is the authors’ response to the original reviews
Public Reviews:
Reviewer #1 (Public Review):
Summary:
In this study from Belato, Knight, and co-workers, the authors investigated the Rec domain of a thermophilic Cas9 from Geobacillus stearothermophilus (GeoCas9). The authors investigated three constructs, two individual subdomains of Rec (Rec1 and Rec2) and the full Rec domain. This domain is involved in binding to the guide RNA of Cas9, as well as the RNA-DNA duplex that is formed upon target binding. The authors performed RNA binding and relaxation experiments using NMR for the wild-type domain as well as two-point mutants. They observed differences in RNA binding activities as well as the flexibility of the domain. The authors also performed experiments on fulllength GeoCas9 to determine whether these biophysical differences affect the RNA binding or cleavage activity. Although the authors observed some changes in the thermal stability of the mutant GeoCas9-gRNA complex, they did not observe substantial differences in the cleavage activities of the mutant GeoCas9 variants.
Overall, this manuscript provides a detailed biophysical analysis of the GeoCas9 Rec domain. The NMR assignments for this construct should prove very useful, and the results may provide the grounds for future engineering of higher fidelity variants of GeoCas9. While the NMR results are generally well presented, it is unclear how the results on the isolated Rec domain related to the overall function of full-length GeoCas9. In addition, some conclusions are overstated and not fully supported by the evidence provided. The following major points should be addressed by the authors.
(1) Many of the results rely on the backbone resonance assignments of the three constructs that were used, and the authors have done an excellent job of assigning the Rec1 and Rec2 constructs. However, it is unclear from the descriptions in the text how the full-length Rec construct was assigned. Were these assignments made based on assignments for the individual domains? The authors state that the spectra of individual domains and RecFL overlay very well, but there appear to be many resonances that have chemical shift differences or are only present in one construct. As it stands, it is unclear how the resonances were assigned for residues whose chemical shifts were perturbed, making it difficult to interpret many of the results.
The Reviewer raises an important oversight. In Lines 491-493, we clarify that we were able to transfer the assignments using spectral overlays of the individual domains with GeoRec (i.e. careful analysis of the data in Figure S3). We also cite two new references where a similar approach was applied to Cas9.
(2) The minimal gRNA that was used for the Rec-gRNA binding experiments is unlikely to be a good mimic for the full-length gRNA, as it lacks any of the secondary structure that is most specifically recognized by the REC lobe and the rest of the Cas9 protein. The majority of this RNA is a "spacer" sequence, but spacers are variable, so this sequence is arbitrary. Thus, the interactions that the authors are observing most likely represent non-specific interactions between the Rec domains and RNA. The authors also map chemical shift perturbations and line broadening on structural models with an RNA-DNA duplex bound, but this is not an accurate model for how the Rec domain binds to a single-stranded RNA (for which there is no structural model). Thus, many of the conclusions regarding the RNA binding interface are overstated.
The Reviewer again raises an important point. We have added a section of text explaining the rationale for truncating the gRNA for binding experiments with NMR (Lines 223-235). We chose the 5’end of the gRNA containing the spacer sequence based on crystal structures of NmeCas9 and SpCas9 that show the Rec lobe interacting with this section of nucleic acid. The newly published GeoCas9 cryo-EM structure bound to gRNA, which overlaid well with the NmeCas9 structure, also suggested that this portion of the gRNA could interact with Rec.
Figures S11 and S12 show our gradual truncation of the gRNA and Rec construct to achieve useful atomic detail. Ultimately, a 39nt gRNA containing a 23 base pair spacer sequence was chosen for this study to retain the NMR signal of the complex and because several structures suggested this 39nt sequence would be long enough to interact with the entire Rec lobe.
To investigate the effect of the spacer sequence, we have now measured binding affinities via MST between GeoRec and a 39nt Tnnt2 gRNA and a 39nt gRNA from PDB: 8UZA, containing a different spacer sequence used in the very recent GeoCas9 cryo-EM structure. The observed trends for each gRNA are consistent across the samples. We also measured WT, K267E, and R332A GeoCas9 affinity for the full-length Tnnt2 and PDB:8UZA gRNAs.
Lastly, we used a new cryo-EM structure of GeoCas9 bound to gRNA (PDB: 8JTR) to better define the interface for NMR CSPs and line broadening and have adjusted the language in this section.
(3) The authors include microscale thermophoresis (MST) data for the Rec constructs binding to the minimal gRNA. These data suggest that all three Rec variants have very similar Kd's for the RNA. Given these similarities, it is unclear why the RNA titration experiments by NMR yielded such different results. Moreover, in the Discussion, the authors state that the NMR titration data are consistent with the MST-derived Kd values. This conclusion appears to be overstated given the very small differences in affinities measured by MST.
MST and NMR experiments describing the weakened binding affinity of GeoRec and GeoRec2 for the Tnnt2 gRNA agree with each other (Figure 5). However, additional MST experiments with a different gRNA sequence (from PDB: 8UZA) and with fulllength GeoCas9 (new Figure 7) have provided new insight for us to soften and reframe the Discussion to avoid overstatement. See Lines 263-282 and 375-385.
(4) While the authors have performed some experiments to help place their findings on the isolated Rec domain in the context of the full-length protein, these experiments do not fully support the conclusions that the authors draw about the meaning of their NMR results. The two Cas9 variants that were explored via NMR have no effect on Cas9 cleavage activity, and it is unclear from the data provided whether they have any effect on GeoCas9 binding to the full sgRNA. This makes it difficult to determine whether the observed differences in RNA binding and dynamics of the isolated Rec domain have any consequence in the context of the full protein.
We have since measured the binding affinities of full-length GeoCas9 to full-length gRNA. (new Figure 7) We have also added a comment in the Discussion section describing how both GeoRec and GeoRec2 domain variants bind the truncated RNA with weaker affinity than the WT, but this biophysical effect does not translate to GeoCas9 with its full-length gRNA. We describe this finding as an explanation for why the single-point mutants have minimal effect of GeoCas9 cleavage activity. See Lines 375-385.
(5) The authors state in multiple places that the K267E/R332A mutant enhanced GeoCas9 specificity. Improved specificity refers to a situation in which the efficiency of cleavage of a perfectly matched target improves in comparison to a mismatched target. This is not what the authors observed for the double mutant. Instead, the cleavage of the perfect target was drastically reduced, in some cases to a larger degree than for the mismatched target. The double mutant does not appear to have improved specificity, it has simply decreased cleavage efficiency of the enzyme.
The conclusion has been reframed to suggest that the K267E/R332A double mutant has decreased cleavage efficiency of the enzyme but does not enhance GeoCas9 specificity. We discuss an interesting contrast, namely that mutations in the SpCas9 Rec lobe alter its specificity, which is at times accompanied by a loss of overall activity. We also speculate on why this may not be the case in GeoCas9, considering some very recent (unpublished at the time of initial submission) structural and biochemical data. See Lines 414-418.
Reviewer #2 (Public Review):
Summary:
The manuscript from Belato et al. used advanced NMR approaches and a mutagenesis campaign to probe the conformational dynamics of the recognition lobe (Rec) of the CRISPR Cas9 enzyme from G. stearothermophilus (GeoCas9). Using truncated and full-length constructs they assess the impacts of two different point mutations have on the redistribution and timescale of these motions and assess gRNA recognition and specificity. Single point mutations in the Rec domain in a Cas9 from a related species had profound impacts on- and off-target DNA editing, therefore the authors reasoned analogous mutations in GeoCas9 would have similar effects. However, despite a redistribution of local motions and changes in global stability, their chosen mutations had little impact on DNA editing in the context of the full-length enzyme. Their studies highlight the species-specific complexity of interdomain communication and allosteric mechanisms used by these multi-domain endonucleases. Despite these negative results, their study is highly rigorous, and their approach will broadly support understanding how the activity and specificity of these enzymes can be engineered to tune activity and limit off-target cleavage by these enzymes.
Strengths:
(1) Atomistic investigation of the conformational dynamics of the GeoCas9 gRNA recognition lobe (GeoRec), probing dynamics on a broad range of timescales from ps to ms using advanced NMR approaches will be broadly interesting to both the structural biology and CRISPR engineering communities.
(2) Highly rigorous biophysical studies that push the boundaries of current techniques, provide insight into local dynamics of the GeoRec domain that serve to propagate allosteric information and potentially regulate enzymatic activity.
(3) The study highlights the complexities of understanding interdomain communication in Cas9 enzymes since analogous mutations in different species have different effects on target recognition and cleavage.
(4) The type of structural and dynamic insights derived from this study design could serve as foundational information to guide a rational design strategy aimed at improving the selectivity and reducing the off-target effects of Cas9 enzymes.
Weaknesses:
(1) Despite the rigor of the experiments, the mutations chosen by the authors do not have a profound effect on the overall substrate affinity or activity of GeoCas9 rendering little mechanistic insight into allosteric communication in this particular Cas9. However, the double mutant K267E/R332A has a more pronounced effect on the cleavage of WT and mismatched (at nucleotides 19 and 20) DNA substrates while minimally affecting the cleavage of mismatched (at nucleotides 5 and 6), suggesting more could be learned about the allosteric mechanism from the detailed characterization of this mutant.
We thank the Reviewer for this comment. While we have included new binding experiments with full-length GeoCas9 and gRNAs (new Figure 7), the addition of new MD simulations (new Figure 6) better address this point. MD examined our single and double mutants, as well as the recently published high-specificity iGeoCas9, and reported the degree of conformational sampling and nucleic acid contacts and binding energies.
The simulations show that our mutations induce some, but not the full extent of the effect of iGeoCas9 (with one mutation in GeoRec and many others in the adjacent WED domain), implying that further engineering of GeoRec to mimic iGeoCas9’s properties can have profound functional outcomes. Future efforts to mutate GeoRec will be leverage this strategy. See Lines 309-342.
(2) Follow-up experiments with other residues that were identified as being highly dynamic might affect substrate recognition and cleavage activity in different ways providing additional insight.
The Reviewer is correct. While beyond this initial scope, new MD simulations (see the response directly above) and NMR resonances distally affect by gRNA (via CSP or relaxation dispersion) will be used identify the primary targets for this analysis.
(3) Details regarding the authors' experimental approach are incomplete such as a description of the model used to fit the CD data, a detailed explanation of the global fitting of the relaxation dispersion data describing how the best-fit model was selected, and the description of the ModelFree fitting of fast timescale dynamics is incomplete.
We thank the Reviewer for pointing out these oversights. We have now included the fitting equation in the CD Methods section.
We included new Figures S8-S10 with the individual relaxation dispersion curves and note in the Methods that global fits were deemed superior based on the Akaike Information Criterion. For WT, the AIC showed the global fit to be ~10-fold better. For K267E, the global model was 4-fold better, and for R332A, the global model was 6-fold better.
We have included a more detailed description of CPMG and Model-free fitting. See Lines 520-526.
Reviewer #3 (Public Review):
The authors explore the role of Rec domains in a thermophilic Cas9 enzyme. They report on the crystal structure of part of the recognition lobe, its dynamics from NMR spin relaxation and relaxation-dispersion data, its interaction mode with guide RNA, and the effect of two single-point mutations hypothesised to enhance specificity. They find that mutations have small effects on Rec domain structure and stability but lead to significant rearrangement of micro- to milli-second dynamics which does not translate into major changes in guide RNA affinity or DNA cleavage specificity, illustrating the inherent tolerance of GeoCas9. The work can be considered as a first step towards understanding motions in GeoCas9 recognition lobe, although no clear hotspots were discovered with potential for future rational design of enhanced Cas9 variants.
Recommendations for the authors:
Reviewer #1 (Recommendations For The Authors):
Suggestions for improved or additional experiments, data, or analyses
(1) Please update the sentences on lines 100-105 and the Methods to clarify how the RecFL assignments were obtained. If RecFL was assigned based on the assignments for Rec1 and Rec2, please describe in the Methods how the shifted resonances were handled. Please also provide chemical shift perturbation profiles for the truncated constructs versus the full-length Rec construct.
We have now added text (Lines 491-493) and two new references explaining the GeoRec (full-length) assignment.
We appreciate this point. We have now provided a new Figure S9 with analysis of CSPs and line broadening in truncated constructs (GeoRec2 only). See also Lines 263-282. We also show a similar structural response to mutation in full-length GeoRec and GeoRec2 NMR CSPs (Figure 2 and Figure S5).
We have provided the CSPs for each construct, relative to the full-length GeoRec domain, Author response image 1. In most cases, the largest CSPs occur at resonances on the periphery of the spectra, retaining the ability to unambiguously assign it.
Author response image 1.
(2) It is unclear whether the differences in Kd's for the Rec-gRNA interactions are statistically significant, given the errors associated with the values. Can the authors further analyze these data to determine statistical significance? If they are not found to be significantly different, the authors should soften all conclusions related to the observed differences.
Statistical significance was calculated for all MST data and Figures 5 and 7 have been updated to reflect this
(3) As mentioned above, it seems likely that the Rec-RNA binding that is observed is non-specific. Have the authors tried MST with another 39 nt RNA? Are there differences in affinities for the Rec constructs?
We have done MST with another 39nt RNA. The affinity for each gRNA (Tnnt2 vs 8UZA) is similar for WT and K267E, and a factor of ~4 weaker for R332A with 8UZA gRNA. The trend is the same, that WT Rec has a (statistically significant) stronger affinity for the gRNA compared to the mutants.
(4) Have the authors tried MST with full-length GeoCas9 and the sgRNA? The current data on the thermal stability of the RNP's is interesting, but a more direct measurement of the affinity of the Cas9-sgRNA complexes would provide stronger evidence of the effects of the mutations.
The Reviewer makes an excellent suggestion. We have now generated Cy5-labeled full-length gRNAs and conducted MST with full-length GeoCas9 (new Figure 7). The binding affinities to multiple guides do not vary significantly. We have discussed this, and its implications, in Lines 376-385.
(5) One potential issue with not observing differences between the three Cas9 variants' cleavage activity is that the activity of these purified proteins appears to be very low in comparison to previous studies of GeoCas9. There are significant differences in the expression protocol used by the authors of the current study and previous studies. Have the authors attempted to replicate the expression and purification protocol of previous reports? This may improve the enzymatic activity and allow for a more detailed investigation of cleavage between the three variants (e.g. by performing time-course cleavage assays).
The expression protocol of GeoCas9 is identical to those of previous studies. This was a written mistake on our part, which has now been corrected in the methods section. We apologize for this oversight.
Recommendations for improving the writing and presentation
The introduction of the manuscript is reasonable for specialists who are very familiar with Cas9 function, but it does not contain important details that may be unknown to most readers. The authors do not introduce the domains of Cas9 in the Introduction section. A brief description of the domains that are important to this work should be provided. For example, what is the role of the Rec lobe? This is not introduced until lines 110-111, after some discussion of the authors' initial work on these domains. For a broad audience, it would also be helpful to define the two catalytic domains of the protein. A paragraph describing the general architecture of Cas9 and the overall mechanism of Cas9, including allostery and domain movement, would be very helpful to a general audience. There are elements of this throughout the manuscript, but it would be better to have everything described in a single location at the beginning of the Introduction.
The Reviewer makes an excellent point. We have added significant clarifying text to the Introduction (Lines 42-47, 52-58, and 61-66). We have also amended Figure 1 to highlight the domain arrangement of GeoCas9 and construct domain boundaries.
Minor corrections to the text
(1) Lines 37-38: The statement about GeoCas9 activity should reference citation.
We have added two references here.
(2) Line 39-40: "The widely-studied SpCas9, as well as GeoCas9, are Type-II CRISPR systems". Cas9 is only a single component of a larger system that contains other proteins and DNA elements, so it would be more appropriate to say "are effectors of type II CRISPR systems" or "are signature proteins of type II CRISPR systems". Also, please define the organism from which SpCas9 is derived. It may be more appropriate to use the three-letter abbreviation "SpyCas9" to be consistent with the abbreviation used for GeoCas9.
We have revised the initial suggestion and specified the organisms. We have, however, chosen to keep “SpCas9” for consistency with our prior work and the work of many several others, including Doudna et al and Zhang et al.
(3) Lines 39-42: "only the Type II-C class to which GeoCas9 belongs has been rigorously validated for mammalian genome editing". SpCas9 is from a type II-A system and is by far the most commonly used ortholog for genome editing, including in ongoing clinical trials. It is unlikely that any of the type II-C Cas9 orthologs have been more rigorously validated than SpCas9. The reference cited in this sentence also does not support this statement and is a review written in 2017, so would be unlikely to reflect the current state of the art. Please revise this sentence.
We have softened and revised this text (Lines 42-47).
(4) Lines 48-52: It would be helpful to describe the dynamic movement of the HNH domain (and cite appropriate references) prior to describing the authors' previous work. As it stands, it is unclear how this sentence would be understood by a non-specialist.
We have added text in Lines 61-68
(5) Lines 44-45: The wording is a little unclear, as it sounds like the guide RNA, rather than the nuclease domains, is responsible for dsDNA cleavage. The sentence could be adjusted to remove "and cleave". Cleavage by the HNH and RuvC domains could be described in a separate sentence.
We have revised this text. See Lines 49-50.
(6) Lines 46-48: This segment of the sentence suggests that PAM recognition triggers the allosteric events that result in the movement of the nuclease domain (HNH). This is misleading, as HNH movement is triggered by the complete formation of an R-loop, rather than initial PAM recognition. Please revise this sentence.
We have revised the text in Lines 52-58.
(7) Lines 62-65: The first sentence is unclear. The specificity of many protein-nucleic acid complexes is well understood and is also readily quantified by several wellestablished methods. Are the authors specifically referring to the structural basis for Cas9 specificity? Although Cas9 specificity is highly complex, it has been studied structurally in great detail and should not be described as "poorly understood" without some discussion of what is already known. These sentences also elide the fact that Cas9 specificity has been successfully altered via rational design, based on our general framework for understanding protein-nucleic acid interactions. Please clarify these statements.
The Reviewer makes an important point. We have softened this statement (Lines 8081). We have clarified that we intended to refer to structural characterization of large, multidomain proteins and nucleic acid complexes via NMR. We agree that many critical structural studies comment on Cas9 dynamics and specificity in great detail, including at the domain-level.
(8) Lines 62-68: It seems like the citations do not match up with the references in this section. The references for citations 8-10 are not about DNA repair complexes, references 11-14 are not papers about the directed evolution of Cas9 (should these be 16-17?), and the references for the HNH domain movements should be for citations 1821.
We apologize for the confusion, and the references have been updated
(9) Lines 116-119: The description of the RNAs used is unclear, as the segments that are described add up to 141 not 101. Also, what is meant by "110-nt guide sequence intrinsic to GeoCas9"? Is this referring to the tracrRNA segment? It may be helpful if the RNA sequences shown in the accompanying figures were replaced with cartoons of the RNAs that were used, with the different segments labeled.
We now describe the gRNA sequences in detail in new Table S4. We also expanded a bit in the text (Lines 224-235).
(10) Line 121-123: This sentence should contain reference(s).
We have changed the sentence.
(11) Line 156-158: Reference 19 did not report or investigate any higher specificity SpCas9 variants, is this citation correct?
We have removed the reference from this line. Ref. 19 (now Ref 23, Slaymaker et al) should be correct.
(12) Lines 162-166: Please provide a sequence and structural alignment for SpCas9 and GeoCas9 to support the claim that the amino acid substitutions are equivalent between the two orthologs.
We have updated Figure 1 to display the similarity in domain arrangement between SpCas9 and GeoCas9 and have noted similarity in structure and sequence of these proteins in Figure S1.
(13) Lines 234-236: There is insufficient evidence to conclude that the alterations in protein dynamics caused the changes in gRNA interaction. The substitutions are charge swap substitutions, and it is equally (if not more) feasible that these substitutions decrease the potential for favorable electrostatic interactions.
(14) Lines 261-265: While the RNP stability for R332A is clearly decreased in comparison to WT, the authors' conclusions regarding K267E seem overstated. The difference in Tm for the K267E mutant and WT RNPs is not very large and may be within error, especially given that the CD data are noisy. Similarly, on lines 321-322, only one of the mutations really impacted the stability of the full-length RNP.
We have softened this text in Lines 303-305.
(15) Lines 336-338: HiFi-SpCas9 does not contain four mutations, it is a single R691A point mutation, as reported in reference 17. This sentence and subsequent sentences should be updated.
Here, the “final form” of HiFi SpCas9 contains the R691A and three additional mutations. The Reviewer is correct, though, that the R691A mutation alone was enough to enhance the specificity of WT SpCas9. We have clarified this point on Line 156.
Minor corrections to the figures
(16) The cryo-EM structures of GeoCas9 have recently been released on the PDB. The authors may now update figures to include the experimentally determined structure, rather than an AlphaFold model and update the text accordingly.
We have made this change.
(17) For Figure S4, please describe what the red dashed lines are in the top three graphs. Are these the Tm values determined for the two individual Rec domains? How do these compare to the inflection points for the two transitions in the full Rec construct (could be determined by plotting the first derivative data)? Please provide information in the Methods on how the temperature-dependent CD spectral data were fit and Tm's were determined.
We have made these changes in the Figure S4 caption and Methods section.
(18) The blue box denoting the unassigned region is missing from Figure 2C-D, although it is mentioned in the figure legend.
We have added the blue box denoting the unassigned linker.
Reviewer #2 (Recommendations For The Authors):
The manuscript is well-written and generally clear and concise. The following recommendations will help improve the readability and include details important for interpreting the results.
(1) In general, the figures are too small and difficult to interpret, it was hard to discern the differences described in the text (e.g. Figure 1A, E, 4A, etc.), the text labels are illegible in several panels (e.g. Figure 4A, S8B, C, etc.), the chosen colors were difficult to interpret in the structures (Figure 4C, S8G, H, etc.), as well as residues with motion (as balls) were difficult to make out due to size and color usage. Similar story for the dispersion curves (Fig 3A), the plots are chaotically crowded, and it is impossible to interpret (or see) the undelaying data.
We apologize for these difficulties. We have now revised the Figures in several ways. First, we greatly simplified Figure 1, such that it now includes only the domain arrangement, structure, and initial NMR details for GeoRec (essentially A-B of the old Figure 1).
Second, we have reformatted Figure 3 to make the structure maps a bit easier to see.
We certainly appreciate the point made by the Reviewer about the dispersion curves. Our intent here is to illustrate the number of curves that can be fit globally, which substantially increase for K267E and R332A GeoRec3, versus WT. As a compromise, we have included the individual dispersion curves in the SI for each variant. We have also thinned the line weights for each fit, and added NMR order parameters to the main figure to round out the discussion of dynamics.
Third, we have compiled the gRNA titration into Figure 4, removing the CD analysis (to SI), MST data (new Fig 5), and unclear structure maps to focus only on the NMR spectra here.
Fourth, we have created a new Figure 5 focusing on MST studies of two gRNAs with GeoRec, which now include bar charts of affinities with appropriate statistics.
Much of the data trimmed from the prior version of the manuscript figures has been moved to Supporting Information. We have also created two new main text Figures (6 & 7) based on MD simulations and MST studies of full-length GeoCas9 and gRNAs to provide additional context for interpreting the results in prior figures.
(2) Line 39 - this sentence is awkward, could you rephrase it?
We have rephrased this sentence.
(3) There is inconsistent labeling, in Figure S2 the full-length construct is referred to as GeoRecFL while in other places in the text and in Figure 1 it is called GeoRec.
We have changed all references to the intact Rec lobe to “GeoRec.”
(4) It would be helpful to include a cartoon of the domain organization of GeoCas9 and indicate the truncation mutants that were studied in this manuscript.
We included the domain organization in Figure 1A and indicated the amino acid boundaries for each construct on the figure and in the Methods section.
(5) There is significant line broadening that occurs during the titration, not all line broadening is due to changes in rotational correlation time, and differential line broadening may reveal interactions of residues that are in the intermediate regime, certainly, uM affinities measured by the authors, would suggest this, therefore, a plot of I/Io might inform on binding sites, and it might be useful to look at differential broadening as a function of titrant added.
The Reviewer makes a very good point. In addition to the data in Figure 4, which show a clear reduction in gRNA-induced line broadening in larger GeoRec constructs, we included new titration data on smaller GeoRec2 domains (Figure S12). Here, we conducted an I/I0 analysis and added some clarifying language about the possible nature of line broadening in these samples. See new Figure S12 and Lines 268-274.
(6) Line 126 "Importantly, many resonances are also minimally impacted." This statement is unclear since from the plots shown in Figure 1D, it seems that many of the residues are impacted by RNA titration, see the point about differential broadening above, this sort of plot may help pick apart residues that broaden due to RNA contacts (rather than changing rotational correlation).
We have removed this statement, in addition to our revisions above regarding the line broadening.
(7) Line 137 - I am not sure that a max chemical shift of 0.15 ppm constitutes "strong chemical shift perturbations"
The Reviewer makes a good point. We have changed “strong” to “significant” which refers to 1 standard deviation above the 10% trimmed mean of the data. See Line 237.
(8) Line 144 - change to "...experimentally determined structure...".
We have added new lines 135-136 to make this point clear. We reinforced that initial predictions were based on the Alphafold2, since an experimental structure was lacking, but we have now discussed the mutations in context of the new structural data.
(9) The section from lines 150 - 166, comparison of the effect of different mutations in different Cas9 seems more appropriate for the discussion section.
We have added additional text on this point in the Discussion section, within several new paragraphs.
(10) In Figure S6, chemical shifts are observed at the distal site away from the mutations, could the authors discuss?
The Reviewer makes an important observation. Indeed, the CSPs caused by K267E and R332A extend beyond the mutation site. These shifts are mostly close in 3D space to the mutation, and consistent in Figures 2 and S5. New titrations of gRNA into isolated GeoRec2 also activate some distal sites, and new MD simulations suggests the mutations disrupt RNA and DNA contacts, where these distal effects may play a role with full-length gRNAs.
We agree it would be worth mutating distal sites undergoing CSPs to examine their impact on function, but two complicating factors are 1) the lack of substantial gRNA affinity differences in experiments with full-length GeoCas9 and 2) the lack of functional changes in the mutants. In this initial study, it appears difficult to assign an effect to these distal sites in GeoCas9 (beyond speculation). We do have a brief discussion of the distal sites (Lines 293-298) and will follow up this work with more comprehensive mutagenesis studies of these sites.
(11) It appears that the authors fitted the Tm data to some model although this is not mentioned in the text, figure captions, or methods. In the caption for Figure 4D the authors refer to "Fitted thermal denaturation profiles...".
We have added the relevant Equation in the Methods and referenced it in Figure S6 and S14 captions.
(12) Details of the ModelFree fitting are needed, how many residues fit with the minimal models, and how many invoked Rex and other terms? How does the statement in line 191 about the elevated S2 values arising from global tumbling compare with an experimental estimation of rotational correlation eg. from R2/R1 ratios?
We have included an expanded description of the Model-free protocol (Lines 521-527). The best diffusion tensor was an ellipsoid model. The number of residues utilizing Rex was 81, though Rex contribution was very small. The mean and errors for the fast motion (S2f), slow motion (S2z) and generalized order parameter were 0.97 ± 0.15, 0.84 ± 0.14, and 0.91 ± 0.20, respectively.
R2/R1 ratios for each of the samples (relaxation conducted on GeoRec2 in isolation) corresponded to an estimated tc of 16.3 ns for all data sets. This value is a bit larger than would be expected for a compact globular protein of 25 kDa, though our X-ray structure of GeoRec2 shows a somewhat elongated domain.
(13) Line 221 - referring to two different figures at the end of the sentence is confusing, maybe place the figure references immediately after the referral in the sentence.
We have resolved due to reshuffling of the Figures.
(14) Line 234 - Fig 4E is mentioned before fig 4D, in fact Fig 4D is not mentioned in the text.
We have reordered and edited many of the Figures, this is now resolved.
(15) Line 243 - what is the saturating concentration to which the authors are referring?
We have amended the Results section to more clearly discuss the effect of gRNA on the GeoRec and (now) GeoRec2 domains. We meant 3-fold excess gRNA-to-protein by “saturating” in the prior version. At that point, CSPs held stable and the degree of line broadening at certain sites had completely obscured the resonance from view.
(16) Fig 4E caption - mentions error of 1.34 while the figure is labeled 1.1 for the R332A GeoRec mutant.
This has been resolved due to additional MST trails as well as the editing and reordering of many Figures.
(17) Line 253 - the authors are discussing regions of allosteric hotspots, how do the motions of these predicted hotspots compare with the relaxation dispersion data? There seems to be some overlap.
The Reviewer makes a keen observation. Yes, there is overlap in these data. For example, hotspot residue R269 is bracketed by L268 and L270 with relaxation dispersion. Also, hotspot L279 surrounded by C275, A276, R277, and D281 with dispersion in both variants. Further, D403 and E408 reside in a stretch of ms timescale flexibility comprised of N404, L406, N412, and L413. We have yet to fully understand the functional significance of this overlap, but have added a note in Line 298 to draw the reader’s attention to it.
Reviewer #3 (Recommendations For The Authors):
Although the scope of the manuscript is rather limited due to the minor effects observed for the selected mutations, it is clear that a lot of work was done in spearheading the investigation of dynamic modes in GeoCas9 Rec2. In my view, the data will still be of relevance and interest to the general structural and chemical biology communities.
However, there are a few technical shortcomings that need to be addressed and some statements that are poorly supported by data, necessitating either more experimental proofs or rephrasing of the conclusions.
Major points:
X-ray structure - No PDB ID, structural statistics, or validation report is given for the structure, so it is impossible to judge of the quality. Please provide these. Furthermore, it would be commendable to determine the structure of the point mutant Rec2 domains, this would greatly strengthen the claim that mutations affect only dynamics and do not change structure.
We apologize for this oversight. We absolutely had these data at the time of submission but must have forgotten to upload them. The validation report is now attached.
Regarding the mutant structures, the Reviewer’s point is well taken. In the absence of these structures, we have adjusted the language to include the possibility of structural change. We have also included new MD simulations (new Figure 6 and associated text) that provide comment on possible structural and dynamic changes due to mutation. We note that NMR spectral changes are quite modest, beyond the site of mutation. Further, the new binding data with full-length GeoCas9 (new Figure 7) shows very little change in gRNA affinity with mutations, implying that a profound structural rearrangement does not take place.
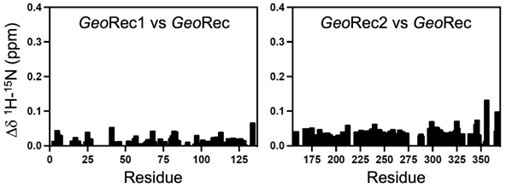
Translating isolated Rec2 findings to FL GeoCas9 - This is an important point and I do appreciate that the authors discuss this. I agree that working on FL samples for NMR would not be feasible, but I am not convinced by the statement that "GeoRec2 in isolation represents the structure of the subdomain within full-length GeoCas9 very well". The chemical shift perturbations observed between isolated Rec2 and FL Cas9 are relatively sizable. This should be discussed in further detail. Figure 1B should showcase peaks having the highest perturbations. Are they located at termini or interaction interfaces?
We have provided the combined 1H-15N combined CSPs for each construct, relative to the full-length GeoRec domain, Author response image 1. In most cases, the largest CSPs occur at resonances on the periphery of the spectra, retaining the ability to unambiguously assign it. The largest CSPs do appear to exist at the termini.
The Rec1 and Rec2 subdomains are connected by a short, but flexible unstructured linker in full-length GeoRec. Thus, the two subdomains do not form a particularly tight non-covalent interface and behave somewhat independently (see Figure S4, for example).
Regarding the statement of “GeoRec2 in isolation...,” we apologize for this confusion.
We were referring to our solved crystal structure in relation to the AlphaFold model. With the new cryo-EM structure of GeoCas9 having been recently published, our X-ray structure of GeoRec2 is still in excellent agreement, but we have clarified our intent on Line 111.
Dynamics and effect of mutations - K267E is more destabilizing and leads to more spread chemical shift perturbations throughout Rec2 and to faster-correlated dynamics but not in significantly lower affinity or cleavage. How do the authors explain this?
The Reviewer raises an interesting question. Regarding the impact of the K267E mutation, new MD simulations also suggest K267E to be quite disruptive of the GeoCas9 structure and dynamics, modulating contacts with the nucleic acids. However, further MD analysis of the recently published (bona fide high specificity) iGeoCas9 variant shows that K267E only imparts a portion of the effect of iGeoCas9, suggesting that even further modulation of GeoRec would be require for substantial functional impact. In addition, new MST binding studies with full-length variants and gRNAs show K267E does not dramatically alter gRNA binding, suggesting that the lack of functional impact, despite biophysical change, is suppressed by the surrounding GeoCas9 domains. We comment on this in the Discussion.
Moreover, the time regime for the fit of the CPMG curves is surprisingly slow given the profiles, how were the minor state populations? Were the dynamics really correlated? Please provide numbers (also see minor points below). In that regime CEST experiments should work, was that done?
The minor state populations were very low in the analysis, <1%.
To examine the correlated dynamics, we compared the global fits to those of the individual fits for each residue and found them to be better for the global fit, based on the Akaike Information Criterion. For WT, the AIC showed the global fit to be ~10-fold better. For K267E, the global model was 4-fold better, and for R332A, the global model was 6-fold better. We have added language clarifying the use of AIC to the Methods section.
We have done CEST experiments on _Geo_HNH (we did not see overly clear evidence for a minor state), but we did not perform these experiments on GeoRec. However, we strongly agree that a detailed follow-up study focusing on CEST and new GeoRec variants should investigate this further.
Since the binding effects with gRNAs differ in the isolated domain and the full-length protein, we have tried not to over-analyze the impact of the relaxation data in this specific context. These data still provide useful information regarding the impact of point mutants on GeoCas9 domain biophysics, and MD simulations support the enhanced dynamics seen in CPMG and other relaxation data. However, the functional implication is clearly more complicated and requires further study.
Mutations affect gRNA affinity - I am not convinced that affinity itself is significantly affected based on the MST data. This data could be reproduced as technical replicates to reduce the error bars, or another technique with less intrinsic noise (ITC, SPR) could be used to better support this claim. However, a 3-fold difference seen from NMR titrations could indicate a change in binding mode, for instance in koff. It would be interesting to obtain SPR or BLI data quantifying the kinetics of the interactions. Anyhow, this point should be more carefully discussed.
We agree with the Reviewer on this point. We conducted additional replicates of MST trials, as well as new MST with a different gRNA sequence. Our updated analysis, including statistics, provides a better measure for “significance” in these data, which is now reported. We have also added some text discussing a possible change in binding mode, see Lines 256-259.
We also carried out MST on full-length GeoCas9 with full-length gRNAs (the same two RNAs used as truncated constructs). We report these data in new Figure 7 and note there is essentially no difference between the gRNAs or the GeoCas9 variants under these conditions.
Further, MD simulations suggest a change in binding energy associated with the gRNA interaction in the context of full-length GeoCas9. Since experimental studies are not able to parse these differences, collectively, we describe a scenario where the highly stable structure of GeoCas9 resists substantial mutation-induced change seen for analogous perturbations in SpCas9. See Lines 309-342, 414-418, and 448-461.
Minor points:
• Please detail how the error on R1 and R2 rates was calculated.
We have included new text in Lines 514-518.
• Please detail how hetNOE values were calculated (simply Isat/Iref?) and what values were used for Model Free.
Yes, the Reviewer is correct. We have added specifically that we used Isat/Iref on Line 518.
• Please elaborate on the Model Free analysis. What tensor was used for tumbling? What was the correlation time? This is needed to judge the trustworthiness of S2 parameters.
We have included new text on Lines 520-526. The diffusion tensor used was an ellipsoid and the correlation time was 15.4 ns. The correlation time estimated from R2/R1 ratios was 16.3 ns.
• Figure 1: Please indicate where Rec1 and Rec2 are located on panel A and indicate the residue assignments for each peak showcased in panel B.
We have indicated the boundary of Rec1 and Rec2 in the new cartoon of Figure 1A. We have also noted the exact amino acids used for each construct in the Methods. We also added resonance labels to the spectral overlays in Figure 1B. We have done the same
• Line 187: I believe this should refer to Figure S8C rather than Figure 3A.
We have made this change.
• Some fits of the CPMG curves look strange, e.g. R343 in Fig. 3B WT definitely does not contain significant us-ms dynamics and should be excluded from the analysis. Please double-check each profile. Were other models besides CR72 not providing better fits?
The Reviewer has made a very careful observation. Our intent was to highlight these sites on purpose to show differences in CPMG relaxation dispersion between WT and variant samples. This was provided as some evidence for the redistribution of dynamics between samples, as many different sites found to be “rigid” on the ms timescale in WT GeoRec2 were flexible in GeoRec2 variants. We agree, however, that this Figure panel was confusing and have therefore removed it in favor of simple discussion in the text.
• To what degree are the CPMG dynamics correlated, can you provide statistical measures for the global fits?
We compared the global fits to those of the individual fits for each residue and found them to be better for the global fit, based on the Akaike Information Criterion. For WT, the AIC showed the global fit to be ~10-fold better. For K267E, the global model was 4fold better, and for R332A, the global model was 6-fold better.
We have added language clarifying the use of AIC to the Methods section.
• Error measured from replicates and p-values should be reported for DNA cleavage assays.
We thank the Reviewer for pointing out this omission. We have included error bars on these plots.
-

eLife assessment
This study provides useful insights into the conformational dynamics of the nucleic acid recognition lobe of GeoCas9, a thermophilic Cas9 from Geobacillus stearothermophilus. The influence of local dynamics and allosteric regulation on guide RNA binding affinity and DNA cleavage specificity is investigated via cutting-edge NMR approaches and mutagenesis. While backed by rigorous biophysical analyses, evidence supporting the proposed mechanistic model is found to be incomplete due to the limited impact of the studied mutations on GeoCas9 cleavage activity. This work will be of interest to biochemists and biophysicists interested in interdomain communication and allosteric mechanisms in Cas9 enzymes.
-

Reviewer #1 (Public Review):
Summary:
In this study from Belato, Knight, and co-workers, the authors investigated the Rec domain of a thermophilic Cas9 from Geobacillus stearothermophilus (GeoCas9). The authors investigated three constructs, two individual subdomains of Rec (Rec1 and Rec2) and the full Rec domain. This domain is involved in binding to the guide RNA of Cas9, as well as the RNA-DNA duplex that is formed upon target binding. The authors performed RNA binding and relaxation experiments using NMR for the wild-type domain as well as two-point mutants. They observed differences in RNA binding activities as well as the flexibility of the domain. The authors also performed experiments on full-length GeoCas9 to determine whether these biophysical differences affect the RNA binding or cleavage activity. Although the authors …
Reviewer #1 (Public Review):
Summary:
In this study from Belato, Knight, and co-workers, the authors investigated the Rec domain of a thermophilic Cas9 from Geobacillus stearothermophilus (GeoCas9). The authors investigated three constructs, two individual subdomains of Rec (Rec1 and Rec2) and the full Rec domain. This domain is involved in binding to the guide RNA of Cas9, as well as the RNA-DNA duplex that is formed upon target binding. The authors performed RNA binding and relaxation experiments using NMR for the wild-type domain as well as two-point mutants. They observed differences in RNA binding activities as well as the flexibility of the domain. The authors also performed experiments on full-length GeoCas9 to determine whether these biophysical differences affect the RNA binding or cleavage activity. Although the authors observed some changes in the thermal stability of the mutant GeoCas9-gRNA complex, they did not observe substantial differences in the cleavage activities of the mutant GeoCas9 variants.
Overall, this manuscript provides a detailed biophysical analysis of the GeoCas9 Rec domain. The NMR assignments for this construct should prove very useful, and the results may provide the grounds for future engineering of higher fidelity variants of GeoCas9. While the NMR results are generally well presented, it is unclear how the results on the isolated Rec domain related to the overall function of full-length GeoCas9. In addition, some conclusions are overstated and not fully supported by the evidence provided. The following major points should be addressed by the authors.
(1) Many of the results rely on the backbone resonance assignments of the three constructs that were used, and the authors have done an excellent job of assigning the Rec1 and Rec2 constructs. However, it is unclear from the descriptions in the text how the full-length Rec construct was assigned. Were these assignments made based on assignments for the individual domains? The authors state that the spectra of individual domains and RecFL overlay very well, but there appear to be many resonances that have chemical shift differences or are only present in one construct. As it stands, it is unclear how the resonances were assigned for residues whose chemical shifts were perturbed, making it difficult to interpret many of the results.
(2) The minimal gRNA that was used for the Rec-gRNA binding experiments is unlikely to be a good mimic for the full-length gRNA, as it lacks any of the secondary structure that is most specifically recognized by the REC lobe and the rest of the Cas9 protein. The majority of this RNA is a "spacer" sequence, but spacers are variable, so this sequence is arbitrary. Thus, the interactions that the authors are observing most likely represent non-specific interactions between the Rec domains and RNA. The authors also map chemical shift perturbations and line broadening on structural models with an RNA-DNA duplex bound, but this is not an accurate model for how the Rec domain binds to a single-stranded RNA (for which there is no structural model). Thus, many of the conclusions regarding the RNA binding interface are overstated.
(3) The authors include microscale thermophoresis (MST) data for the Rec constructs binding to the minimal gRNA. These data suggest that all three Rec variants have very similar Kd's for the RNA. Given these similarities, it is unclear why the RNA titration experiments by NMR yielded such different results. Moreover, in the Discussion, the authors state that the NMR titration data are consistent with the MST-derived Kd values. This conclusion appears to be overstated given the very small differences in affinities measured by MST.
(4) While the authors have performed some experiments to help place their findings on the isolated Rec domain in the context of the full-length protein, these experiments do not fully support the conclusions that the authors draw about the meaning of their NMR results. The two Cas9 variants that were explored via NMR have no effect on Cas9 cleavage activity, and it is unclear from the data provided whether they have any effect on GeoCas9 binding to the full sgRNA. This makes it difficult to determine whether the observed differences in RNA binding and dynamics of the isolated Rec domain have any consequence in the context of the full protein.
(5) The authors state in multiple places that the K267E/R332A mutant enhanced GeoCas9 specificity. Improved specificity refers to a situation in which the efficiency of cleavage of a perfectly matched target improves in comparison to a mismatched target. This is not what the authors observed for the double mutant. Instead, the cleavage of the perfect target was drastically reduced, in some cases to a larger degree than for the mismatched target. The double mutant does not appear to have improved specificity, it has simply decreased cleavage efficiency of the enzyme.
-

Reviewer #2 (Public Review):
Summary:
The manuscript from Belato et al. used advanced NMR approaches and a mutagenesis campaign to probe the conformational dynamics of the recognition lobe (Rec) of the CRISPR Cas9 enzyme from G. stearothermophilus (GeoCas9). Using truncated and full-length constructs they assess the impacts of two different point mutations have on the redistribution and timescale of these motions and assess gRNA recognition and specificity. Single point mutations in the Rec domain in a Cas9 from a related species had profound impacts on- and off-target DNA editing, therefore the authors reasoned analogous mutations in GeoCas9 would have similar effects. However, despite a redistribution of local motions and changes in global stability, their chosen mutations had little impact on DNA editing in the context of the …
Reviewer #2 (Public Review):
Summary:
The manuscript from Belato et al. used advanced NMR approaches and a mutagenesis campaign to probe the conformational dynamics of the recognition lobe (Rec) of the CRISPR Cas9 enzyme from G. stearothermophilus (GeoCas9). Using truncated and full-length constructs they assess the impacts of two different point mutations have on the redistribution and timescale of these motions and assess gRNA recognition and specificity. Single point mutations in the Rec domain in a Cas9 from a related species had profound impacts on- and off-target DNA editing, therefore the authors reasoned analogous mutations in GeoCas9 would have similar effects. However, despite a redistribution of local motions and changes in global stability, their chosen mutations had little impact on DNA editing in the context of the full-length enzyme. Their studies highlight the species-specific complexity of interdomain communication and allosteric mechanisms used by these multi-domain endonucleases. Despite these negative results, their study is highly rigorous, and their approach will broadly support understanding how the activity and specificity of these enzymes can be engineered to tune activity and limit off-target cleavage by these enzymes.
Strengths:
(1) Atomistic investigation of the conformational dynamics of the GeoCas9 gRNA recognition lobe (GeoRec), probing dynamics on a broad range of timescales from ps to ms using advanced NMR approaches will be broadly interesting to both the structural biology and CRISPR engineering communities.
(2) Highly rigorous biophysical studies that push the boundaries of current techniques, provide insight into local dynamics of the GeoRec domain that serve to propagate allosteric information and potentially regulate enzymatic activity.
(3) The study highlights the complexities of understanding interdomain communication in Cas9 enzymes since analogous mutations in different species have different effects on target recognition and cleavage.
(4) The type of structural and dynamic insights derived from this study design could serve as foundational information to guide a rational design strategy aimed at improving the selectivity and reducing the off-target effects of Cas9 enzymes.
Weaknesses:
(1) Despite the rigor of the experiments, the mutations chosen by the authors do not have a profound effect on the overall substrate affinity or activity of GeoCas9 rendering little mechanistic insight into allosteric communication in this particular Cas9. However, the double mutant K267E/R332A has a more pronounced effect on the cleavage of WT and mismatched (at nucleotides 19 and 20) DNA substrates while minimally affecting the cleavage of mismatched (at nucleotides 5 and 6), suggesting more could be learned about the allosteric mechanism from the detailed characterization of this mutant.
(2) Follow-up experiments with other residues that were identified as being highly dynamic might affect substrate recognition and cleavage activity in different ways providing additional insight.
(3) Details regarding the authors' experimental approach are incomplete such as a description of the model used to fit the CD data, a detailed explanation of the global fitting of the relaxation dispersion data describing how the best-fit model was selected, and the description of the ModelFree fitting of fast timescale dynamics is incomplete.
-

Reviewer #3 (Public Review):
The authors explore the role of Rec domains in a thermophilic Cas9 enzyme. They report on the crystal structure of part of the recognition lobe, its dynamics from NMR spin relaxation and relaxation-dispersion data, its interaction mode with guide RNA, and the effect of two single-point mutations hypothesised to enhance specificity. They find that mutations have small effects on Rec domain structure and stability but lead to significant rearrangement of micro- to milli-second dynamics which does not translate into major changes in guide RNA affinity or DNA cleavage specificity, illustrating the inherent tolerance of GeoCas9. The work can be considered as a first step towards understanding motions in GeoCas9 recognition lobe, although no clear hotspots were discovered with potential for future rational design …
Reviewer #3 (Public Review):
The authors explore the role of Rec domains in a thermophilic Cas9 enzyme. They report on the crystal structure of part of the recognition lobe, its dynamics from NMR spin relaxation and relaxation-dispersion data, its interaction mode with guide RNA, and the effect of two single-point mutations hypothesised to enhance specificity. They find that mutations have small effects on Rec domain structure and stability but lead to significant rearrangement of micro- to milli-second dynamics which does not translate into major changes in guide RNA affinity or DNA cleavage specificity, illustrating the inherent tolerance of GeoCas9. The work can be considered as a first step towards understanding motions in GeoCas9 recognition lobe, although no clear hotspots were discovered with potential for future rational design of enhanced Cas9 variants.
-

-