Circadian photoreceptor CRYPTOCHROME promotes wakefulness under short winter-like days via a GABAergic circuitry
Curation statements for this article:-
Curated by eLife
eLife Assessment
Winter months with short days are commonly associated with seasonal depression and hypersomnolence; the mechanisms behind this hypersomnolence however, remain unclear. Chen and colleagues identify a genetic basis for this phenomenon in the fly Drosophila - mutations in the circadian photoreceptor cryptochrome resulted in increased sleep under short photoperiods. These findings are valuable insights into the genetic mechanisms regulating sleep under short days. The data supporting the precise neurobiological basis of these effects however, remains incomplete.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
A cardinal symptom of seasonal affective disorder (SAD, also known as winter depression) is hypersomnolence, while the cause of this “winter sleepiness” is not known. Here we found that lack of the circadian photoreceptor cryptochrome (cry) leads to increased sleep under short winter-like days in fruit flies, reminiscent of the hypersomnolence in SAD. CRY functions in neurons that synthesize the major inhibitory neurotransmitter GABA, including the small ventral lateral neurons which are known to be circadian pacemakers, and down-regulates the GABAergic tone. This in turn leads to increased neural activity of the wake-promoting large ventral lateral neurons, a subset of circadian neurons that are inhibited by GABA-A receptor. CRY protein is known to be degraded by light, thus rendering CRY to be functional within this GABAergic circuitry to enhance wakefulness only under short day length. Taken together, we demonstrate a mechanism that specifically regulates wakefulness under short winter-like days, which may provide insights regarding the winter sleepiness in SAD.
Article activity feed
-

eLife Assessment
Winter months with short days are commonly associated with seasonal depression and hypersomnolence; the mechanisms behind this hypersomnolence however, remain unclear. Chen and colleagues identify a genetic basis for this phenomenon in the fly Drosophila - mutations in the circadian photoreceptor cryptochrome resulted in increased sleep under short photoperiods. These findings are valuable insights into the genetic mechanisms regulating sleep under short days. The data supporting the precise neurobiological basis of these effects however, remains incomplete.
-

Reviewer #1 (Public review):
Summary:
In this paper, Chen et al. identified a role for the circadian photoreceptor CRYPTOCHROME (CRY) in promoting wakefulness under short photoperiods. This research is potentially important as hypersomnolence is often seen in patients suffering from SAD during winter times. The mechanisms underlying these sleep effects are poorly known.
Strengths:
The authors clearly demonstrated that mutations in cry lead to elevated sleep under 4:20 Light-Dark (LD) cycles. Furthermore, using RNAi, they identified GABAergic neurons as a primary site of CRY action to promote wakefulness under short photoperiods. They then provide genetic and pharmacological evidence demonstrating that CRY acts on GABAergic transmission to modulate sleep under such conditions.
Weaknesses:
The authors then went on to identify the neuronal …
Reviewer #1 (Public review):
Summary:
In this paper, Chen et al. identified a role for the circadian photoreceptor CRYPTOCHROME (CRY) in promoting wakefulness under short photoperiods. This research is potentially important as hypersomnolence is often seen in patients suffering from SAD during winter times. The mechanisms underlying these sleep effects are poorly known.
Strengths:
The authors clearly demonstrated that mutations in cry lead to elevated sleep under 4:20 Light-Dark (LD) cycles. Furthermore, using RNAi, they identified GABAergic neurons as a primary site of CRY action to promote wakefulness under short photoperiods. They then provide genetic and pharmacological evidence demonstrating that CRY acts on GABAergic transmission to modulate sleep under such conditions.
Weaknesses:
The authors then went on to identify the neuronal location of this CRY action on sleep. This is where this reviewer is much more circumspect about the data provided. The authors hypothesize that the l-LNvs which are known to be arousal promoting may be involved in the phenotypes they are observing. To investigate this, they undertook several imaging and genetic experiments.
While the authors have made improvements in this resubmitted manuscript, there are still multiple concerns about the paper. I think the authors provide enough evidence suggesting that CRY plays a role in sleep under short photoperiod. The data also supports that CRY acts in GABAergic neurons. However, there are still major issues with the quality of the confocal images presented throughout the paper. In many cases it appears that the images are oversaturated with poor resolution, making it hard to understand what is going on. In addition, none of the drivers used in this study are specific to the neurons the authors aim to manipulate. Therefore, the identity of the GABAergic neurons involved in this CRY dependent sleep mechanism remains unclear. Similarly, whether l-LNvs are the target of this GABA mediated sleep regulation under short photoperiod is not fully demonstrated. The data presented suggests that but does not prove it.
Major concerns:
(1) While the authors provided sleep parameters like consolidation or waking activity for some experiments. These measurements are still not shown for several experiments (for example Figures 2E, 3, 4, 5, and 6). These data are essential, these metrics must be reported for all sleep experiments.
(2) Line 144 "We fed flies with agonists of GABA-A (THIP) and GABA-B receptor (SKF-97541) (Ki and Lim, 2019; Matsuda et al., 1996; Mezler et al., 2001). Both drugs enhance sleep in WT," The proper citation is needed here, Dissel et al., 2015 PMID:25913403. Both THIP and SKF-97541 were used in that paper.
(3) Figure 2C and 2F: it appears that the control data is the same in both panels. That is not acceptable.
(4) Figure 4A: With the quality of the images, it is impossible to assess whether GABA levels are increased at the l-LNvs soma.
(5) Fig 4 S1A shows colabeling of l-LNvs and Gad1-Gal4 expressing neurons. They are almost 100% overlapping signals. This would indicate that the l-LNvs are GABAergic themselves, or that there is a problem with this experiment.
(6) Fig 4 S1B: Again, I can see colabelling of the GFP and PDF staining, suggesting that Gad1-Gal4 expresses in l-LNvs.
(7) Line 184: "Consistently, knocking down Rdl in the l-LNvs rescues the long sleep phenotype of cry mutants (Figure 4-figure supplement 1D)." This statement is incorrect as the driver used for this experiment, 78G01-GAL4 is not specific to the l-LNvs, so it is possible that the phenotypes observed are not coming from these neurons.
(8) Figure 4G-K: None of these manipulations are specific to the l-LNvs. The authors describe 10H10-GAL4 and 78G01-GAL4 as l-LNvs specific tools, but this is not the case. Why not use the SS00681 Split-GAL4 line described in Liang et al., 2017 PMID: 28552314? It is possible that some of the effects reported in this manuscript are not caused by manipulating the l-LNvs.
(9) Similarly for the manipulation of s-LNvs, the authors cannot rule out effect that are coming from other cells as R6-GAL4 is not specific to s-LNvs.
(10) The staining presented in Fig 5 S1 is not very convincing. Difficult to see whether Gad1-GAL4 only expresses in the s-LNvs.
-

Reviewer #3 (Public review):
Summary:
In humans, short photoperiods are associated with hypersomnolence. The mechanisms underlying these effects is however, unknown. Chen et al. use the fly Drosophila to determine the mechanisms regulating sleep under short photoperiods. They find that mutations in the circadian photoreceptor cryptochrome (cry) increase sleep specifically under short photoperiods (e.g. 4h light : 20 h dark). They go on to show that cry is required in GABAergic neurons and that the effects of the cry mutation on sleep are mediated by alterations in GABA signalling. Further, they suggest that the relevant subset of GABAergic neurons are the well-studied small ventral lateral neurons that they suggest inhibit the arousal promoting large ventral neurons via GABA signalling
Strengths:
Genetic analysis to show that …
Reviewer #3 (Public review):
Summary:
In humans, short photoperiods are associated with hypersomnolence. The mechanisms underlying these effects is however, unknown. Chen et al. use the fly Drosophila to determine the mechanisms regulating sleep under short photoperiods. They find that mutations in the circadian photoreceptor cryptochrome (cry) increase sleep specifically under short photoperiods (e.g. 4h light : 20 h dark). They go on to show that cry is required in GABAergic neurons and that the effects of the cry mutation on sleep are mediated by alterations in GABA signalling. Further, they suggest that the relevant subset of GABAergic neurons are the well-studied small ventral lateral neurons that they suggest inhibit the arousal promoting large ventral neurons via GABA signalling
Strengths:
Genetic analysis to show that cryptochrome (but not other core clock genes) mediates the increase in sleep in short photoperiods, and circuit analysis to localise cry function to GABAergic neurons.
Weaknesses:
The authors' have substantially revised their manuscript, and the manuscript is better for the revisions. However, the conclusion that the sLNvs are GABAergic is unfortunately still not well supported by the data. A key sticking point remains the anti GABA immunostaining, and specific driver lines for sLNvs and lLNvs.
The authors should tone down their conclusions to reflect the fact that their data, as presented, does not support the model that cry acts in sLNvs to modulate GABA signalling onto lLNvs and thus modulate sleep.
-

Reviewer #4 (Public review):
Summary:
Short photoperiod is an important experimental manipulation in neurobiology, endocrinology, and metabolism studies. However, the molecular mechanisms by which short photoperiod gives rise to behavioral phenotypes that are seen in seasonal affective disorders remain unknown. Using the classic circadian model organism Drosophila, this study examines short photoperiod-induced hypersomnolence and identifies the circadian photoreceptor cryptochrome as a regulator of GABAergic tone within the clock neural circuit to promote wakefulness under short photoperiod conditions. The discovery has broad implications for understanding how short photoperiod modulates neural inhibition in circadian circuits in regulating sleep.
Strengths:
The Drosophila model provided a powerful platform to dissect the molecular …
Reviewer #4 (Public review):
Summary:
Short photoperiod is an important experimental manipulation in neurobiology, endocrinology, and metabolism studies. However, the molecular mechanisms by which short photoperiod gives rise to behavioral phenotypes that are seen in seasonal affective disorders remain unknown. Using the classic circadian model organism Drosophila, this study examines short photoperiod-induced hypersomnolence and identifies the circadian photoreceptor cryptochrome as a regulator of GABAergic tone within the clock neural circuit to promote wakefulness under short photoperiod conditions. The discovery has broad implications for understanding how short photoperiod modulates neural inhibition in circadian circuits in regulating sleep.
Strengths:
The Drosophila model provided a powerful platform to dissect the molecular mechanisms underlying short photoperiod-induced hypersomnolence. A battery of behavioral, imaging, circuit-manipulation approaches was employed to test the novel hypothesis that the circadian photoreceptor cryptochrome modulates GABAergic tone within the clock neural circuit to promote wakefulness under short photoperiod conditions.
Weaknesses:
The current model proposed by the authors suggests that the small ventral lateral neurons of the Drosophila clock circuit are GABAergic; however, this remains unclear. At present, the field lacks sufficient data and validated reagents to definitively establish the GABAergic identity of these neuropeptidergic neurons.
-

Author response:
The following is the authors’ response to the original reviews
Public Reviews:
Reviewer #1 (Public Review):
Summary:
In this paper, Chen et al. identified a role for the circadian photoreceptor CRYPTOCHROME (cry) in promoting wakefulness under short photoperiods. This research is potentially important as hypersomnolence is often seen in patients suffering from SAD during winter times. The mechanisms underlying these sleep effects are poorly known.
Strengths:
The authors clearly demonstrated that mutations in cry lead to elevated sleep under 4:20 Light-Dark (LD) cycles. Furthermore, using RNAi, they identified GABAergic neurons as a primary site of cry action to promote wakefulness under short photoperiods. They then provide genetic and pharmacological evidence demonstrating that cry acts on GABAergic transmission to …
Author response:
The following is the authors’ response to the original reviews
Public Reviews:
Reviewer #1 (Public Review):
Summary:
In this paper, Chen et al. identified a role for the circadian photoreceptor CRYPTOCHROME (cry) in promoting wakefulness under short photoperiods. This research is potentially important as hypersomnolence is often seen in patients suffering from SAD during winter times. The mechanisms underlying these sleep effects are poorly known.
Strengths:
The authors clearly demonstrated that mutations in cry lead to elevated sleep under 4:20 Light-Dark (LD) cycles. Furthermore, using RNAi, they identified GABAergic neurons as a primary site of cry action to promote wakefulness under short photoperiods. They then provide genetic and pharmacological evidence demonstrating that cry acts on GABAergic transmission to modulate sleep under such conditions.
Weaknesses:
The authors then went on to identify the neuronal location of this cry action on sleep. This is where this reviewer is much more circumspect about the data provided. The authors hypothesize that the l-LNvs which are known to be arousal-promoting may be involved in the phenotypes they are observing. To investigate this, they undertook several imaging and genetic experiments.
Major concerns:
(1) Figure 2 A-B: The authors show that knocking down cry expression in GABAergic neurons mimics the sleep increase seen in cryb mutants under short photoperiod. However, they do not provide any other sleep parameters such as sleep bout numbers, sleep bout duration, and more importantly waking activity measurements. This is an essential parameter that is needed to rule out paralysis and/or motor defects as the cause of increased "sleep". Any experiments looking at sleep need to include these parameters.
Thank you for bringing up these points. We have now included these sleep parameters in Figure 2—figure supplement 3.
(2) For all Figures displaying immunostaining and imaging data the resolution of the images is quite poor. This makes it difficult to assess whether the authors' conclusions are supported by the data or not.
We apologize for the poor resolution. This is probably due to the compression of the figures in the merged PDF file. We are now uploading the figures individually and hopefully this can resolve the resolution issue.
(3) In Figure 4-S1A it appears that the syt-GFP signal driven by Gad1-GAL4 is colabeling the l-LNvs. This would imply that the l-LNvs are GABAergic. The authors suggest that this experiment suggests that l-LNvs receive input from GABAergic neurons. I am not sure the data presented support this.
We agree that this piece of data alone is not sufficient to demonstrate that the l-LNvs receive GABAergic inputs rather than the l-LNvs are GABAergic. However, when nlsGFP signal is driven by two independent Gad1-GAL4 lines (one generated by P element insertion while the other generated by GAL4 inserted into the Gad1 locus), we do not observe any prominent signal in the l-LNvs (Figure 5A and B; Figure 5-figure supplement 1A). We have also co-labeled using Gad1GAL4 and PdfLexA (Figure 5-figure supplement 1B). As can be seen, Gad1GAL4-driven GFP signal is present only in the s-LNvs but not the l-LNvs. This further supports the idea that the l-LNvs are not GABAergic, and that the syt-GFP signal likely arises from GABAergic neurons projecting to the l-LNvs.
(4) In Figure 4-S1B. The GRASP experiment is not very convincing. The resolution of the image is quite poor. In addition, the authors used Pdf-LexA to express the post t-GRASP construct in l-LNvs, but Pdf-LexA also labels the s-LNvs, so it is possible that the GRASP signal the authors observe is coming from the s-LNvs and not the l-LNvs. The authors could use a l-LNvs specific tool to do this experiment and remove any doubts. Altogether this reviewer is not convinced that the data presented supports the conclusion "All in all, these results demonstrate that GABAergic neurons project to the l-LNvs and form synaptic connections." (Line 176). In addition, the authors could have downregulated the expression of Rdl specifically in l-LNvs to support their conclusions. The data they are providing supports a role for RDL but does not prove that RDL is involved in l-LNvs.
Thank you for these wonderful suggestions. Again we apologize for the poor resolution and hopefully by uploading the images separately we can resolve this issue. We agree that the GRASP signal could be coming from the s-LNvs and not the l-LNvs but unfortunately we are not able to find a LexA that is specifically expressed in the l-LNvs. We believe the trans-Tango data further support the idea that GABAergic neurons project to and form synaptic connections with the l-LNvs. Nonetheless, we have changed our conclusion to “All in all, these results strongly suggest that GABAergic neurons project to the l-LNvs and form synaptic connections” to be more rigorous. In addition, we have obtained R78G01GAL4 which is specifically expressed in the l-LNvs, and using this GAL4 to knock down Rdl rescues the long-sleep phenotype of cry mutants (Figure 4—figure supplement 1D).
(5) In Figures 4 A and C: it appears that GABA is expressed in the l-LNvs. Is this correct? Can the authors clarify this? Maybe the authors could do an experiment where they co-label using Gad1-GAL4 and Pdf-LexA to clearly demonstrate that l-LNvs are not GABAergic. Also, the choice of colors could be better. It is very difficult to see what GABA is and what is PDF.
Thank you for this wonderful suggestion. We have now co-labeled using Gad1GAL4 and PdfLexA (Figure 5-figure supplement 1B). As can be seen, Gad1GAL4-driven GFP signal is present only in the s-LNvs but not the l-LNvs. We suspect the GABA signal at the l-LNvs may arise from the GABAergic projections received by these cells. We have now changed the color of the GABA/PDF signals in these images and have reduced the intensity of the PDF signal. Hopefully, it would be easier to visualize in this revised version.
(6) Figure 4G: Pdf-GAL4 expresses in both s-LNvs and l-LNvs. So, in this experiment, the authors are silencing both groups, not only the l-LNvs. Why not use a l-LNvs specific tool?
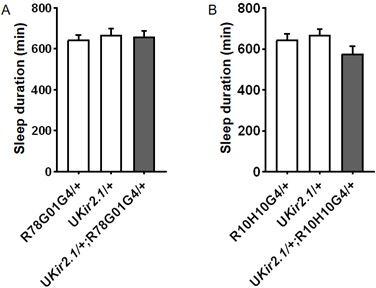
Thank you for bring up this important point. We have previously used c929GAL4 to express Kir2.1 and this led to lethality. We have now used two l-LNv-specific GAL4 drivers (R78G01GAL4 and R10H10GAL4) that we newly obtained to express Kir2.1 but did not observe significant effect on sleep. Please see Author response image 1 for the results.
Author response image 1.
Daily sleep duration of male flies expressing Kir2.1 in l-LNvs using R78G01GAL4 (A)(n = 40, 41, 30 flies) and R10H10GAL4 (B) (n = 40, 41, 32 flies) and controls, monitored under 4L20D. One-way ANOVA with Bonferroni multiple comparison test was used to calculate the difference between experimental group and control group.
(7) Figure 4H-I: The C929-GAL4 driver expresses in many peptidergic neurons. This makes the interpretation of these data difficult. The effects could be due to peptidergic cells being different than the l-LNvs. Why not use a more specific l-LNvs specific tool? I am also confused as to why some experiments used Pdf-GAL4 and some others used C929-GAL4 in a view to specifically manipulate l-LNvs? This is confusing since both drivers are not specific to the l-LNvs.
Thank you for bring up these important points. We have now used the l-LNv-specific R10H10GAL4 and the results are more or less comparable with that of c929GAL4 (Figure 4I and K), i.e. activating the l-LNvs blocks the long-sleep phenotype of cry mutants. The reason PdfGAL4 is used in 4G is because c929GAL4 leads to lethality while the l-LNv-specific GAL4 lines do not alter sleep.
(8) Figure 5-S1B: Why does the pdf-GAL80 construct not block the sleep increase seen when reducing expression of cry in Gad1-GAL4 neurons? This suggests that there are GABAergic neurons that are not PDF expressing involved in the cry-mediated effect on sleep under short photoperiods.
Yes, this is indeed the conclusion we draw from this result, and we commented on this in the Discussion: “Moreover, inhibiting cry RNAi expression in PDF neurons does not eliminate the long-sleep phenotype of Gad1GAL4/UAScryRNAi flies. Therefore, we suspect that cry deficiency in other GABAergic neurons is also required for the long-sleep phenotype. Given that the s-LNvs are known to express CRY and appear to be GABAergic based on our findings here, we believe that CRY acts at least in part in the s-LNvs to promote wakefulness under short photoperiod.”
In conclusion, it is not clear that the authors demonstrated that they are looking at a cry-mediated effect on GABA in s-LNvs resulting in a modulation of the activity of the l-LNvs. Better images and more-suited genetic experiments could be used to address this.
Thank you very much for all the comments. They are indeed quite helpful for improving our manuscript. Hopefully, with images of higher quality and the additional experiments described above, we have now provided more evidence supporting our major conclusion.
Reviewer #2 (Public Review):
Summary:
The sleep patterns of animals are adaptable, with shorter sleep durations in the winter and longer sleep durations in the summer. Chen and colleagues conducted a study using Drosophila (fruit flies) and discovered that a circadian photoreceptor called cryptochrome (cry) plays a role in reducing sleep duration during day/night cycles resembling winter conditions. They also found that cry functions in specific GABAergic circadian pacemaker cells known as s-LNvs inhibit these neurons, thereby promoting wakefulness in the animals in the winter. They also identified l-LNvs, known as arousal-promoting cells, as the downstream neurons.
Strengths:
Detailed mapping of the neural circuits cry acts to mediate the shortened sleep in winter-like day/night cycles.
Weaknesses:
The supporting evidence for s-LNvs being GABAergic neurons is not particularly strong. Additionally, there is a lack of direct evidence regarding changes in neural activity for s-LNvs and l-LNvs under varying day/night cycles, as well as in cry mutant flies.
Thank you very much for all the comments. We have now expressed nlsGFP by two independent Gad1-GAL4 lines (one generated by P element insertion while the other generated by GAL4 inserted into the Gad1 locus), and positive signals in the s-LNvs can be observed (Figure 5A and B; Figure 5-figure supplement 1A). Hopefully, this can provide some further support regarding the s-LNvs being GABAergic neurons.
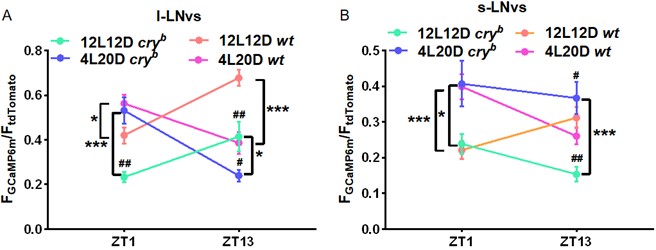
We have now examined GCaMP signals in the l- and s-LNvs of WT and cry mutants under 4L20D/12L12D. Please see Author response image 2 for the results. As can be seen, both WT and cry mutants show photoperiod-dependent changes. Interestingly, cry mutants show more prominent reduction of GCaMP signal in the l-LNvs compared to WT under 12L12D vs. 4L20D, but the sleep duration phenotype is observed only under 4L20D. Moreover, GCaMP signal is elevated in the s-LNvs of cry mutants relative to WT under 4L20D but decreased under 12L12D. These results indicate that there are distinct mechanisms regulating sleep under short vs. normal photoperiod (with CRY being dispensable under 12L12D), and the role of CRY in modulating the activity of these neurons are also photoperiod-dependent. Further in-depth characterizations are need to delineate these complex issues.
Author response image 2.
Quantification of GCaMP6m signal intensity normalized to that of tdTomato under 12L12D and 4L20D (n = 25-45 cells). Student’s t-test: compared to WT, #P < 0.05, ##P < 0.01; 12L12D vs. 4L20D, *P < 0.05, ***P < 0.001.Reviewer #3 (Public Review):
Summary:
In humans, short photoperiods are associated with hypersomnolence. The mechanisms underlying these effects are, however, unknown. Chen et al. use the fly Drosophila to determine the mechanisms regulating sleep under short photoperiods. They find that mutations in the circadian photoreceptor cryptochrome (cry) increase sleep specifically under short photoperiods (e.g. 4h light: 20 h dark). They go on to show that cry is required in GABAergic neurons. Further, they suggest that the relevant subset of GABAergic neurons are the well-studied small ventral lateral neurons that they suggest inhibit the arousal-promoting large ventral neurons via GABA signalling.
Strengths:
Genetic analysis to show that cryptochrome (but not other core clock genes) mediates the increase in sleep in short photoperiods, and circuit analysis to localise cry function to GABAergic neurons.
Weaknesses:
The authors' conclusion that the sLNvs are GABAergic is not well supported by the data. Better immunostaining experiments and perhaps more specific genetic driver lines would help with this point (details below).
(1) The sLNvs are well known as a key component of the circadian network. The finding that they are GABAergic would if true, be of great interest to the community. However, the data presented in support of this conclusion are not convincing. Much of the confocal images are of insufficient resolution to evaluate the paper's claims. The Anti-GABA immunostaining in Fig 4 and 5 seem to have a high background, and the GRASP experiments in Fig 4 supplement 1 low signal.
We apologize for the poor resolution. This is probably due to the compression of the figures in the merged PDF file. We are now uploading the figures individually and hopefully this can resolve the resolution issue. Unfortunately, the GABA immunostaining does not work very well in our hands and thus the background is high. We have now adjusted the images by changing the minimum lookup table (LUT) value in the green channel to 213, which removes all pixels below 213. This can remove background without changing the gray values, so the analysis is not affected. We have modified all images the exact same way and hopefully this can improve the contrast. Furthermore, we have now expressed nlsGFP by two independent Gad1-GAL4 lines (one generated by P element insertion while the other generated by GAL4 inserted into the Gad1 locus), and positive signals in the s-LNvs can be observed (Figure 5A and B; Figure 5-figure supplement 1A). Hopefully, this can provide some further support regarding the s-LNvs being GABAergic neurons.
Transcriptomic datasets are available for the components of the circadian network (e.g. PMID 33438579, and PMID 19966839). It would be of interest to determine if transcripts for GAD or other GABA synthesis/transport components were detected in sLNvs. Further, there are also more specific driver lines for GAD, and the lLNvs, sLNVs that could be used.
Thank you for these wonderful suggestions. Based on PMID 19966839, both the s-LNvs and l-LNvs express Gad1 and VGAT at a relatively low level, although here in our study Gad1GAL4 expression is observed only in the s-LNvs and not l-LNvs. We have commented on this in the 4th paragraph of Discussion: “One study using cell-type specific gene expression profiling demonstrates Gad1 and VGAT expression in both s-LNvs and l-LNvs, although with relatively low signal (Nagoshi et al., 2010). Here we observed that Gad1GAL4 is expressed in the s-LNvs, and their GABA intensity is reduced when we use R6GAL4 to knock down VGAT in these cells.” PMID 33438579 does not report expression of these genes in either s-LNvs or l-LNvs, likely due to insufficient sequencing depth. Furthermore, we have now used two l-LNv-specific GAL4 lines (R78G01GAL4 and R10H10GAL4) to conduct some of the experiments that we previously used c929GAL4 for, and obtained comparable results (Figure 4I and K).
(2) The authors' model posits that in short photoperiods, cry functions to suppress GABA secretion from sLNvs thereby disinhibiting the lNVs. In Fig 4I they find that activating the lLNvs (and other peptidergic cells) by c929>NaChBac in a cryb background reduces sleep compared to activating lLNVs in a wild-type background. It's not clear how this follows from the model. A similar trend is observable in Fig 4H with TRP-mediated activation of lNVs, although it is not clear from the figure if the difference b/w cryb vs wild-type background is significant.
Thank you for bring up this important point. This does appear to be counterintuitive. We suspect that in cry mutants, there is more inhibition occurring at the l-LNvs and thus the system may be particularly sensitive to their activation. Therefore, activating these neurons on the mutant background can result in a more prominent wake-promoting effect compared to that of WT.
Recommendations for the authors:
Our major concern centers around the claim that the sLNvs are GABAergic and secrete GABA onto the lLNVs. As it stands, this is not well supported by the data.
The authors could substantiate these findings by using more specific driver lines for GAD / vGAT (MiMic based lines are available that should better recapitulate endogenous expression). Transcriptomic data for circadian neurons are available, the FlyWire consortium also predicts neurotransmitter identities for specific neural circuits. These datasets could be mined for evidence to support the claim of sLNvs being GABAergic
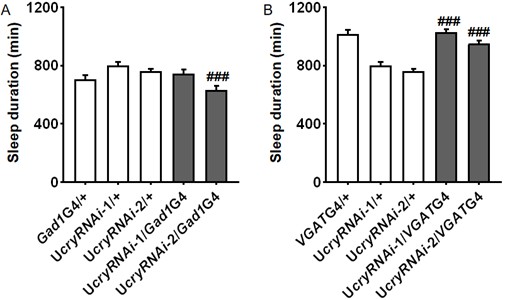
Thank you for these wonderful suggestions. We have now used MiMic-based lines for Gad1 (BS52090, Mi{MIC}Gad1MI09277) and VGAT (BS23022, Mi{ET1}VGATMB01219) to knock down cry but unfortunately were not able to observe changes in sleep. Please see Author response image 3 for the results.
Author response image 3.
Daily sleep duration of male flies with cry knocked down in GABAergic neurons by Gad1GAL4 (A) (n = 30, 38, 50, 18, 31 flies) or VGATGAL4 (B) (n = 28, 38, 50, 18, 30 flies) monitored under 4L20D.One-way ANOVA with Bonferroni multiple comparison test: compared to UAS control, ###P < 0.001.
Furthermore, we have now included another Gad1GAL4 line which is generated by knocking GAL4 transgene into the Gad1 locus. We are also able to observe increased sleep when using this GAL4 to knock down cry, and positive signals in the s-LNvs can be observed when using this GAL4 to drive nlsGFP (Figure 2B; Figure 5-figure supplement 1A).
Based on PMID 19966839, both the s-LNvs and l-LNvs express Gad1 and VGAT at a relatively low level, although here in our study Gad1GAL4 expression is observed only in the s-LNvs and not l-LNvs. We have commented on this in the 4th paragraph of Discussion: “One study using cell-type specific gene expression profiling demonstrates Gad1 and VGAT expression in both s-LNvs and l-LNvs, although with relatively low signal (Nagoshi et al., 2010). Here we observed that Gad1GAL4 is expressed in the s-LNvs, and their GABA intensity is reduced when we use R6GAL4 to knock down VGAT in these cells.” The FlyWire does not have prediction for this particular circuit that we are interested in.
Further, many of the immunostaining images have high background / low signal - so better confocal images would help, as would the use of more specific driver lines for the lNVs as it is sometimes hard to distinguish the lLNvs from sLNvs.
We have now adjusted all images by changing the minimum lookup table (LUT) value in the green channel to 213 and that of the red channel to 279, which removes all pixels below 213 and 279, respectively. This can remove background without changing the gray values, so the analysis is not affected. We have modified all images the exact same way and hopefully this can improve the signal to noise ratio. We were not able to find a LexA line that is specifically expressed in the l-LNvs but we have found two l-LNv-specific GAL4 lines (R78G01GAL4 and R10H10GAL4). We used these lines to conduct some of the experiments that we previously used c929GAL4 for, and obtained comparable results (Figure 4I and 4K).
Additional specific comments are in the reviews above.
Minor points:
(1) Line 55: CRYPTOCHROME is misspelled.
This has been fixed.
(2) Line 140: The authors need to provide the appropriate references for the use of THIP and SKF-97541.
This has been added.
(3) Line 149: there are multiple GABA-A receptors in flies, the authors should acknowledge that. What about LccH3 or Grd?
Thank you for bring up this important point. Here we focused only on Rdl because it is the only GABA-A receptor known to be involved in sleep regulation. We have modified our description regarding this issue: “We tested for genetic interaction between cry and Resistant to dieldrin (Rdl), a gene that encodes GABA-A receptor in flies and has previously been shown to be involved in sleep regulation.”
-

-

-

-

eLife assessment
Winter months with short days are commonly associated with seasonal depression and hypersomnolence; the mechanisms behind this hypersomnolence however remain unclear. Chen and colleagues identify a genetic basis for this phenomenon in the fly Drosophila - mutations in the circadian photoreceptor cryptochrome resulted in increased sleep under short photoperiods. These findings are potentially valuable insights into the genetic mechanisms regulating sleep under short days. The data supporting the neurobiological basis of these effects is however incomplete.
-

Reviewer #1 (Public Review):
Summary:
In this paper, Chen et al. identified a role for the circadian photoreceptor CRYPTOCHROME (cry) in promoting wakefulness under short photoperiods. This research is potentially important as hypersomnolence is often seen in patients suffering from SAD during winter times. The mechanisms underlying these sleep effects are poorly known.Strengths:
The authors clearly demonstrated that mutations in cry lead to elevated sleep under 4:20 Light-Dark (LD) cycles. Furthermore, using RNAi, they identified GABAergic neurons as a primary site of cry action to promote wakefulness under short photoperiods. They then provide genetic and pharmacological evidence demonstrating that cry acts on GABAergic transmission to modulate sleep under such conditions.Weaknesses:
The authors then went on to identify the neuronal …Reviewer #1 (Public Review):
Summary:
In this paper, Chen et al. identified a role for the circadian photoreceptor CRYPTOCHROME (cry) in promoting wakefulness under short photoperiods. This research is potentially important as hypersomnolence is often seen in patients suffering from SAD during winter times. The mechanisms underlying these sleep effects are poorly known.Strengths:
The authors clearly demonstrated that mutations in cry lead to elevated sleep under 4:20 Light-Dark (LD) cycles. Furthermore, using RNAi, they identified GABAergic neurons as a primary site of cry action to promote wakefulness under short photoperiods. They then provide genetic and pharmacological evidence demonstrating that cry acts on GABAergic transmission to modulate sleep under such conditions.Weaknesses:
The authors then went on to identify the neuronal location of this cry action on sleep. This is where this reviewer is much more circumspect about the data provided. The authors hypothesize that the l-LNvs which are known to be arousal-promoting may be involved in the phenotypes they are observing. To investigate this, they undertook several imaging and genetic experiments.Major concerns:
1. Figure 2 A-B: The authors show that knocking down cry expression in GABAergic neurons mimics the sleep increase seen in cryb mutants under short photoperiod. However, they do not provide any other sleep parameters such as sleep bout numbers, sleep bout duration, and more importantly waking activity measurements. This is an essential parameter that is needed to rule out paralysis and/or motor defects as the cause of increased "sleep". Any experiments looking at sleep need to include these parameters.2. For all Figures displaying immunostaining and imaging data the resolution of the images is quite poor. This makes it difficult to assess whether the authors' conclusions are supported by the data or not.
3. In Figure 4-S1A it appears that the syt-GFP signal driven by Gad1-GAL4 is colabeling the l-LNvs. This would imply that the l-LNvs are GABAergic. The authors suggest that this experiment suggests that l-LNvs receive input from GABAergic neurons. I am not sure the data presented support this.
4. In Figure 4-S1B. The GRASP experiment is not very convincing. The resolution of the image is quite poor. In addition, the authors used Pdf-LexA to express the post t-GRASP construct in l-LNvs, but Pdf-LexA also labels the s-LNvs, so it is possible that the GRASP signal the authors observe is coming from the s-LNvs and not the l-LNvs. The authors could use a l-LNvs specific tool to do this experiment and remove any doubts. Altogether this reviewer is not convinced that the data presented supports the conclusion "All in all, these results demonstrate that GABAergic neurons project to the l-LNvs and form synaptic connections." (Line 176). In addition, the authors could have downregulated the expression of Rdl specifically in l-LNvs to support their conclusions. The data they are providing supports a role for RDL but does not prove that RDL is involved in l-LNvs.
5. In Figures 4 A and C: it appears that GABA is expressed in the l-LNvs. Is this correct? Can the authors clarify this? Maybe the authors could do an experiment where they co-label using Gad1-GAL4 and Pdf-LexA to clearly demonstrate that l-LNvs are not GABAergic. Also, the choice of colors could be better. It is very difficult to see what GABA is and what is PDF.
6. Figure 4G: Pdf-GAL4 expresses in both s-LNvs and l-LNvs. So, in this experiment, the authors are silencing both groups, not only the l-LNvs. Why not use a l-LNvs specific tool?
7. Figure 4H-I: The C929-GAL4 driver expresses in many peptidergic neurons. This makes the interpretation of these data difficult. The effects could be due to peptidergic cells being different than the l-LNvs. Why not use a more specific l-LNvs specific tool? I am also confused as to why some experiments used Pdf-GAL4 and some others used C929-GAL4 in a view to specifically manipulate l-LNvs? This is confusing since both drivers are not specific to the l-LNvs.
8. Figure 5-S1B: Why does the pdf-GAL80 construct not block the sleep increase seen when reducing expression of cry in Gad1-GAL4 neurons? This suggests that there are GABAergic neurons that are not PDF expressing involved in the cry-mediated effect on sleep under short photoperiods.
In conclusion, it is not clear that the authors demonstrated that they are looking at a cry-mediated effect on GABA in s-LNvs resulting in a modulation of the activity of the l-LNvs. Better images and more-suited genetic experiments could be used to address this.
-

Reviewer #2 (Public Review):
Summary:
The sleep patterns of animals are adaptable, with shorter sleep durations in the winter and longer sleep durations in the summer. Chen and colleagues conducted a study using Drosophila (fruit flies) and discovered that a circadian photoreceptor called cryptochrome (cry) plays a role in reducing sleep duration during day/night cycles resembling winter conditions. They also found that cry functions in specific GABAergic circadian pacemaker cells known as s-LNvs inhibit these neurons, thereby promoting wakefulness in the animals in the winter. They also identified l-LNvs, known as arousal-promoting cells, as the downstream neurons.Strengths:
Detailed mapping of the neural circuits cry acts to mediate the shortened sleep in winter-like day/night cycles.Weaknesses:
The supporting evidence for s-LNvs …Reviewer #2 (Public Review):
Summary:
The sleep patterns of animals are adaptable, with shorter sleep durations in the winter and longer sleep durations in the summer. Chen and colleagues conducted a study using Drosophila (fruit flies) and discovered that a circadian photoreceptor called cryptochrome (cry) plays a role in reducing sleep duration during day/night cycles resembling winter conditions. They also found that cry functions in specific GABAergic circadian pacemaker cells known as s-LNvs inhibit these neurons, thereby promoting wakefulness in the animals in the winter. They also identified l-LNvs, known as arousal-promoting cells, as the downstream neurons.Strengths:
Detailed mapping of the neural circuits cry acts to mediate the shortened sleep in winter-like day/night cycles.Weaknesses:
The supporting evidence for s-LNvs being GABAergic neurons is not particularly strong. Additionally, there is a lack of direct evidence regarding changes in neural activity for s-LNvs and l-LNvs under varying day/night cycles, as well as in cry mutant flies. -

Reviewer #3 (Public Review):
Summary:
In humans, short photoperiods are associated with hypersomnolence. The mechanisms underlying these effects are, however, unknown. Chen et al. use the fly Drosophila to determine the mechanisms regulating sleep under short photoperiods. They find that mutations in the circadian photoreceptor cryptochrome (cry) increase sleep specifically under short photoperiods (e.g. 4h light : 20 h dark). They go on to show that cry is required in GABAergic neurons. Further, they suggest that the relevant subset of GABAergic neurons are the well-studied small ventral lateral neurons that they suggest inhibit the arousal-promoting large ventral neurons via GABA signalling.Strengths:
Genetic analysis to show that cryptochrome (but not other core clock genes) mediates the increase in sleep in short photoperiods, and …Reviewer #3 (Public Review):
Summary:
In humans, short photoperiods are associated with hypersomnolence. The mechanisms underlying these effects are, however, unknown. Chen et al. use the fly Drosophila to determine the mechanisms regulating sleep under short photoperiods. They find that mutations in the circadian photoreceptor cryptochrome (cry) increase sleep specifically under short photoperiods (e.g. 4h light : 20 h dark). They go on to show that cry is required in GABAergic neurons. Further, they suggest that the relevant subset of GABAergic neurons are the well-studied small ventral lateral neurons that they suggest inhibit the arousal-promoting large ventral neurons via GABA signalling.Strengths:
Genetic analysis to show that cryptochrome (but not other core clock genes) mediates the increase in sleep in short photoperiods, and circuit analysis to localise cry function to GABAergic neurons.Weaknesses:
The authors' conclusion that the sLNvs are GABAergic is not well supported by the data. Better immunostaining experiments and perhaps more specific genetic driver lines would help with this point (details below).1. The sLNvs are well known as a key component of the circadian network. The finding that they are GABAergic would if true, be of great interest to the community. However, the data presented in support of this conclusion are not convincing. Much of the confocal images are of insufficient resolution to evaluate the paper's claims. The Anti-GABA immunostaining in Fig 4 and 5 seem to have a high background, and the GRASP experiments in Fig 4 supplement 1 low signal.
Transcriptomic datasets are available for the components of the circadian network (e.g. PMID 33438579, and PMID 19966839). It would be of interest to determine if transcripts for GAD or other GABA synthesis/transport components were detected in sLNvs. Further, there are also more specific driver lines for GAD, and the lLNvs, sLNVs that could be used.
2. The authors' model posits that in short photoperiods, cry functions to suppress GABA secretion from sLNvs thereby disinhibiting the lNVs. In Fig 4I they find that activating the lLNvs (and other peptidergic cells) by c929>NaChBac in a cryb background reduces sleep compared to activating lLNVs in a wild-type background. It's not clear how this follows from the model. A similar trend is observable in Fig 4H with TRP-mediated activation of lNVs, although it is not clear from the figure if the difference b/w cryb vs wild-type background is significant.
-