Cortex-wide response mode of VIP-expressing inhibitory neurons by reward and punishment
Curation statements for this article:-
Curated by eLife
eLife assessment
This paper is of potential interest to neuroscientists expert in cortical circuitry and behavioral role of neuron types. The imaging technique used permitted to detect a specific group of cortical neurons known as vasoactive intestinal peptide (VIP)-expressing interneurons from several cortical regions with high temporal resolution. The main message conveyed by this manuscript is that many VIP-expressing interneurons respond to reward and punishment but also show regional differences. The conclusions drawn are generally supported by the data, but some claims and interpretations require further attention and clarification.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
Neocortex is classically divided into distinct areas, each specializing in different function, but all could benefit from reinforcement feedback to inform and update local processing. Yet it remains elusive how global signals like reward and punishment are represented in local cortical computations. Previously, we identified a cortical neuron type, vasoactive intestinal polypeptide (VIP)-expressing interneurons, in auditory cortex that is recruited by behavioral reinforcers and mediates disinhibitory control by inhibiting other inhibitory neurons. As the same disinhibitory cortical circuit is present virtually throughout cortex, we wondered whether VIP neurons are likewise recruited by reinforcers throughout cortex. We monitored VIP neural activity in dozens of cortical regions using three-dimensional random access two-photon microscopy and fiber photometry while mice learned an auditory discrimination task. We found that reward and punishment during initial learning produce rapid, cortex-wide activation of most VIP interneurons. This global recruitment mode showed variations in temporal dynamics in individual neurons and across areas. Neither the weak sensory tuning of VIP interneurons in visual cortex nor their arousal state modulation was fully predictive of reinforcer responses. We suggest that the global response mode of cortical VIP interneurons supports a cell-type-specific circuit mechanism by which organism-level information about reinforcers regulates local circuit processing and plasticity.
Article activity feed
-

-

Author Response
Reviewer #1 (Public Review):
Building upon the previous evidence of activation of auditory cortex VIP interneurons in response to non-classical stimuli like reward and punishment, Szadai et al., extended the investigation to multiple cortical regions. Use of three-dimensional acousto-optical two-photon microscopy along with the 3D chessboard scanning method allowed high-speed signal acquisition from numerous VIP interneurons in a large brain volume. Additionally, activity of VIP interneurons in deep cortical regions was obtained using fiber photometry. With the help of these two imaging methods authors were able to extract and analyze the VIP cell signal from different cortical regions. Study of VIP interneuron activity during an auditory go-no-go task revealed that more than half of recorded cortical VIP interneurons …
Author Response
Reviewer #1 (Public Review):
Building upon the previous evidence of activation of auditory cortex VIP interneurons in response to non-classical stimuli like reward and punishment, Szadai et al., extended the investigation to multiple cortical regions. Use of three-dimensional acousto-optical two-photon microscopy along with the 3D chessboard scanning method allowed high-speed signal acquisition from numerous VIP interneurons in a large brain volume. Additionally, activity of VIP interneurons in deep cortical regions was obtained using fiber photometry. With the help of these two imaging methods authors were able to extract and analyze the VIP cell signal from different cortical regions. Study of VIP interneuron activity during an auditory go-no-go task revealed that more than half of recorded cortical VIP interneurons were responding to both reward and punishment with high reliability. Fiber photometry data revealed similar observations; however, the temporal dynamics of reinforcement stimuli-related response in mPFC was slower than in the auditory cortex. The authors performed detailed analysis of individual cell activity dynamics, which revealed five categories of VIP cells based on their temporal profiles. Further, animals with higher performance on the discrimination task showed stronger VIP responses to 'go trials' possibly suggesting the role of VIP interneurons in discrimination learning. Authors found that reinforcement related response of VIP interneurons in visual cortex was not correlated with their sensory tuning, unveiling an interesting idea that VIP interneurons take part in both local as well as global processing. These observations bring attention to the possible involvement of VIP interneurons in reinforcement stimuli-associated global signaling that would regulate local connectivity and information processing leading to learning.
The state-of-the-art imaging technique allowed authors to succeed in imaging VIP interneurons from several cortical regions. Advanced analyses revealed the nuances, similarities and differences in the VIP activity trend in various regions. The conclusions about reinforcement stimuli related activity of VIP interneurons made by the authors are well supported by the results obtained, however some claims and interpretations require more attention and clarification.
We thank Reviewer #1 for the positive general comments.
Reviewer #2 (Public Review):
In recent years the activity of cortical VIP+ interneurons in relation to learning and sensory processing has raised great interest and has been intensely investigated. The ability of VIP+ interneurons in the auditory cortex to respond to both reward and punishment was already reported a few years ago by some of the authors (Pi et al., 2013, Nature). However, this work importantly adds to their previous study demonstrating a largely similar and synchronous response of a large fraction of these interneurons across the neocortex to salient stimuli of different valence during the performance of an auditory discrimination task.
An additional strength of this study is the analysis and identification of the general pattern of VIP+ interneuron responses associated to specific behaviors in the different layers of the neocortex depth.
Interestingly, the authors also identified using cluster analysis 5 different classes of VIP+ interneurons, based on the dynamic of their responses, that were unequally distributed in distinct cortical areas.
This is a well performed study that took advantage of a cutting-edge imaging approach with high recording speed and good signal-to-noise ratio. Experiments are well performed and the data are properly analyzed and nicely illustrated. However, one shortcoming of this paper, in my opinion, is the "case report" structure of the data. Essentially for each neocortical area the activity of VIP+ interneurons was analyzed only in one animal. This limits the assessment of the stability of the response/recruitment of these interneurons. I appreciate the high number of recorded VIP+ interneurons per area/animal and I do understand that it would be excessively laborious to perform 3D random-access two-photon microscopy in several mice for each cortical area. On the other hand, it would be important to have some knowledge of the general variability of the responses of these neurons among animals.
In conclusion, despite the findings described in this manuscript being generally sound, additional experiments are recommended to further substantiate the conclusions.
Thank you for pointing out this potential misunderstanding. Although we mentioned the number of animals the recordings were obtained from (n=22 total), we repeated this multiple times to alleviate the potential confusion. The data recorded with the 2-photon microscope are from 16 animals, and fiber photometry was performed on a separate 6 animals. Each animal was recorded in one (14 mice) or two areas (8 mice, 2 AOD, 6 photometry). We aimed to acquire data from at least 3 recordings per area (4 in the primary somatosensory cortex, 6 in the primary and secondary motor cortices, 4 in the lateral and medial parietal cortices, 3 in the primary visual cortices, 6 in the auditory and medial prefrontal cortices). In the revised manuscript this information can be found at the beginning of the results section and in the figure legends:
“To probe the behavioral function of VIP interneurons, we trained head-fixed mice (n=22 in total, n=16 for 2-photon microscopy and n=6 for fiber photometry) on a simple auditory discrimination task (Figure 1A).”
“Among the 811 neurons imaged in 18 imaging sessions from 16 mice,”
“Ca2+ responses of individual VIP interneurons recorded separately from 18 different cortical regions from 16 mice using fast 3D AO imaging were averaged for Hit (thick green), FA (thick red), Miss (dark blue), and CR (light blue). Fiber photometry data were recorded simultaneously from mPFC and ACx regions and are shown in gray boxes. Functional map (Kirkcaldie, 2012) used with the permission of the author. Speaker symbols represent the average time of tone onset, and gray triangles mark the reinforcement onset for Hit and FA. Averages of Miss and CR trials were aligned according to the expected reinforcement delivery calculated on the basis of the average reaction time. mPFC: medial prefrontal cortex (n=6 mice), ACx: auditory cortex (n=6), S1Hl/S1Tr/S1Bf/S1Sh: primary somatosensory cortex, hindlimb/trunk/barrel field/shoulder region (n=4), M1/M2: primary/secondary motor cortex (n=6), Mpta/Lpta: medial/lateral parietal cortex (n=4), V1: primary visual cortex (n=3).”
“This approach allowed us to simultaneously measure bulk calcium-dependent signals from VIP interneurons located in the right medial prefrontal (mPFC) and left auditory cortices (ACx) by implanting two 400 µm optical fibers at these locations (n=6 sessions from n=6 mice, Figure 1–figure supplement 1C).”
“Raster plot of the trial-to-trial activation of the responsive VIP neurons in Hit and FA trials during the two-photon imaging sessions (n=18 sessions, n=16 mice, n=746 cells).”
Subregional labels, for example on Figure 2, should be considered as additional information to orient the readers, even if they were very precisely defined on the basis of the coordinates. All analyses considering regional differences were conducted on the level of the main functional areas of the dorsal cortex (motor, somatosensory, parietal, and visual). Despite some location-dependent heterogeneity in the late response phase (Figures 2G and H), even these main dorsal cortical regions were all similar from the perspective of responsiveness to reinforcers and auditory cues.
Reviewer #3 (Public Review):
In this study Szadai et al. show reliable, relatively synchronous activation of VIP neurons across different areas of dorsal cortex in response to reward and punishment of mice performing an auditory discrimination task. The authors use both a relatively fast 2 photon imaging, as well as fiber photometry for some deeper areas. They cluster neurons according to their temporal response profiles and show that these profiles differ across areas and cortical depths. Task performance, running behavior and arousal are all related to VIP response magnitude, as has been previously shown.
Methodologically, this paper is strong: the described imaging technique allows for fairly fast sampling rates, they sample VIP cells from many different areas and the analyses are sophisticated and touch on the most relevant points. The figures are of high quality.
However, as the manuscript is now, the presentation could be clearer, the methods more complete and it is not clear whether their conclusions are entirely supported by the data.
The main issue is that reinforcement and arousal are hard to distinguish in this study. It is well known that VIP activity is correlated with arousal. And it is fairly clear that the reinforcement they use in this study - air puffs to the eye, as well as water rewards - cause arousal. It is possible that the reinforcer responses they observe in VIP neurons throughout all areas merely reflect the increases in arousal caused by these behaviorally salient events. They do discuss this caveat (albeit not fully convincingly) and in their abstract even state that the arousal state was not predictive of reinforcer responses. However their data clearly shows the tight relationship of the VIP reinforcer responses to both arousal (as measured by pupil diameter), as well as running speed of the animal. Both of these variables are well known to be tightly coupled to VIP activity.
Although barely mentioned, the authors do appear to sometimes present uncued reward (Figure S2F). If responses were noticeably different from the same events in the task context (as actual reinforcers) this could at least hint towards the reinforcement signal being distinct from mere arousal. However, this data is only mentioned in one supplementary figure in a different context (comparison with PV cells) and neither directly compared to cued reward, nor is this discussed at all. Were uncued air puffs also presented? How do the responses compare to cued air puffs/punishment?
Our original approach to distinguish between reinforcement- and arousal-related responses aimed:
to show that VIP cells with both low and high correlation coefficients with arousal produce large signals upon reinforcement presentation (Figure 3B),
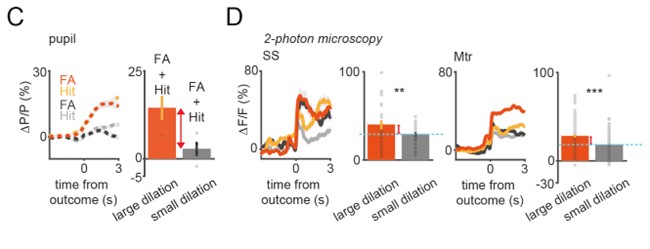
the high differences of low and high arousal changes were reflected in a limited way in the VIP activity (Figures 3C and D): as highlighted in Figure R1, where we also added bars to show ∆P/P in high and low pupil change conditions, the difference in ∆P/P is ~5-fold, while it is only ~1.5-fold for ∆F/F. This disproportionality suggests that a large part of the signal below the dashed blue line is independent of arousal. We have added these modifications to the new version of Figure 3 for clarity.
Figure R1 = Figure 3C-D with modification. Comparison of pupil changes and corresponding calcium averages.
We collected further evidence to support our claims. In Figure 3–figure supplement 2 we depicted Hit and FA trials in which the reinforcement didn’t elevate the arousal level any further. Many of these trials were associated with locomotion prior to the reinforcement, but it was also common that the animals remained still during the whole trial. Trials with increased locomotion upon reinforcement presentation were excluded. Reinforcement-related calcium signals were still present under these conditions, indicating that these signals are not simple reflections of arousal. Moreover, we estimate the distinct contributions of arousal, locomotion, and reinforcers in Figure 3–figure supplement 2D in a systematic way with a generalized linear model. This model also confirmed our view about the reinforcement-related coding.
We now say in the results:
“Finally, to assess the motor- and reinforcement-related contributions to VIP interneuronal activity, we built a generalized linear model using the behavior and imaging data of the SS and Mtr recordings (Figure 3–figure supplement 2D, n=3 mice). This model was able to explain 18.8 ± 11.1% of the variance of the VIP population calcium signal, and highlighted that arousal was the best predictor, followed by reward, punishment, locomotion velocity, and auditory cue (weights = 0.055, 0.031, 0.028, 0.020, 0.018 respectively; all predictors, except the auditory cue in the case of one animal, contributed significantly, p<0.001). These observations indicate that running and arousal changes alone cannot fully explain the recruitment of VIP interneurons by reinforcers.”
We apologize for not describing the rational and the result from the uncued reward experiments. Briefly, while recording reinforcement related signals in auditory cortex in our task, we realized that the cue delivery, and the resulting purely sensory response could alter the measurement of the reward-related responses. Hence, in order to disentangle the reward and sensory-related responses, we presented the animals with simple, uncued reward and observed a similar and robust recruitment of VIP interneurons. Based on the same rational, we made similar measurement for PV neurons.
We now say in the results:
“We did not further analyze the FA responses in auditory cortex as those responses also had a sensory component linked to the white noise-like sound created by the air puff delivery. Because the cue delivery could prove as a confound to measure reward-mediated responses from VIP interneurons in auditory cortex (see also methods), we delivered random reward in separate sessions. Water droplets delivery recruited VIP interneurons in both auditory and medial prefrontal cortex in a similar fashion as water delivery during the discrimination task (Figure 2–figure supplement 1G). Like our single cell results, PV-expressing neuronal population in ACx did not show any significant change in activity upon similar random reward delivery (Figure 2–figure supplement 1G).”
Regarding the difference between cued and uncued responses, we definitely agree with the reviewer that it is an important point. The goal of this manuscript is however to study how reward and punishment are being represented by VIP interneurons in cortex.
The imaging method appears well suited for their task, however the improvements listed in table S1 make the method appear far superior to existing methods in many aspects. Published or preprinted papers with 2 photon imaging of VIP populations (eg. from Scanziani lab (Keller et al.), Carandini lab (Dipoppa et al.), deVries lab (Millman et al.), Adesnik lab (Mossing et al.), which use the much more common resonant scanning, seem to be able to image 4-7 layers at 4-8Hz with a good enough SNR and potentially bigger neuronal yield of approximately 100-200 VIP cells, depending on the field of view. While not every single cell in a volume would be captured by these studies, the only main advantage of the here-used technique appears to be the superior temporal resolution.
We thank the reviewer for the positive comment and we agree that interpretation must be improved. We agree that the imaging methods in the papers listed above have good SNR and were proper to address the scientific questions that had arisen. As the reviewer points out, 3D-AOD imaging allows fast 3D measurement that cannot be achieved otherwise. We used these advantages to address the critical question of layer specificity in the response of VIP interneurons to reinforcer presentation (Figure 2–figure supplement 1F, but see also the new Figure 1–figure supplement 1B). Regarding the comparison and quantification of the factual advantages of AOD microscopy over other imaging methods, the reviewer and readers can refer to the methods section (3D AO microscopy), Table S1 and Szalay et al., 2016. We agree with the reviewer that one of the main advantages is the superior temporal resolution. The second main advantage is the improved SNR. This originates from the fact that the entire measurement time is spent on regions of interest; measurement of unnecessary background areas is not required. More specifically, SNR is improved even in the case of 2D imaging by the factor of:
((area of the entire frame )/(area of the recorded VIP cells))^0.5
which is about (100)0.5=10 as VIP interneurons represent about 1% of the brain. We used this second advantage of AO scanning when we determined the activation ratio (e.g., see Figure 2D).
As the resolution of single or a few action potentials is challenging in behaving mice labelled with the GCaMP6 sensor, any improvement in SNR will improve the detection threshold. The higher SNR achieved here improved the detection threshold, which also explains the relatively high activation ratio in our work.
In the case of asynchronous activity patterns, there is negligible contribution of individual small neuropil structures to somatic activities because of the relatively high volume-ratio of a soma and a given small neuropil structure: this minimizes the error during ∆F/F calculation of somatic responses. However, reinforcement, arousal, and running can generate highly synchronous neuronal activities which can synchronize neuropil activity around a given soma and, therefore, effectively and systematically modulating the somatic ∆F/F responses. To avoid this error, we used a high NA objective with proper neuropil resolution and combined it with motion correction. The use of the high NA also decreased the total scanning volume to about 689 µm × 639 µm × 580 µm and, therefore, it limited the maximum number of VIP cells which could be recorded. It is also possible to use a low-NA objective with a much higher FOV and scanning volume and record over 1000 VIP cells, but the extension of the PSF along the z dimension is inversely and quadratically proportional to the NA of the objective, therefore neuropil resolution will be at least partially lost. In summary, using the high-NA Olympus objective we maximized the 2P resolution which, in combination with off-line motion artifact elimination, allowed precise recording of somatic signals without any neuropil contamination: this provided correct activation ratio values.
Even though this is not mentioned at all, it certainly appears possible, that the accousto-optical scanning emits audible noise. In this case it would be good to know the frequency range and level of this background noise, whether there are auditory responses to the scanning itself and if it interferes with the performance of the animals in the auditory task in any way. If this is not the case, this should probably simply be mentioned for non-experts.
While the name of the acousto-optical deflectors seems to refer to “acoustic noise”, these devices are driven in the range of 55-120 MHz, which is 3 orders of magnitude higher frequency than the hearing threshold of animals: mice don’t hear them. Moreover, we developed water-cooled AODs ten years ago which means that ventilators are also not required, therefore AOD-based scanning can be used with zero noise emission. In contrast, galvo, resonant, and piezo scanning work in the kHz frequency range, which is in the middle of the hearing range of mice. Moreover, these technologies can’t be used in a vacuum and the scanner is just a few tens of centimeters away from the mice, which means that acoustic noise can’t be canceled but can only be partially suppressed with white noise. We thank the reviewer for the helpful comment and have added one sentence about the absence of acoustic noise during acousto-optical scanning:
“The deflectors are driven in the 55-120 MHz frequency range, therefore the noise emitted does not interfere with the auditory cues, as mice can’t hear it. This, in combination with the water cooling of the deflectors, makes the AOD-based scanning the quietest technology for in-vivo imaging.”
The authors show a strong correlation between task performance (hit rate) and the response to the auditory cue on hit trials. Was there any other significant correlations of VIP cells' responses to other trial types? Was reinforcer response correlated to behavioral variables at all?
We have not found any remarkable correlations between VIP cell activity and behavioral variables except the one mentioned above.
For example, we tested discrimination rate (hit rate/FA rate) correlation with ∆F/Ftone in Hit trials, but this was not significant (R2=0.03, F=0.49, p=0.69), just like Hit rate vs. ∆F/Ftone in FA trials (R2=0.19, F=3.8, p=0.07), and discrimination rate vs. ∆F/Ftone in FA trials (R2=0.07, F=1.1, p=0.31).
-

eLife assessment
This paper is of potential interest to neuroscientists expert in cortical circuitry and behavioral role of neuron types. The imaging technique used permitted to detect a specific group of cortical neurons known as vasoactive intestinal peptide (VIP)-expressing interneurons from several cortical regions with high temporal resolution. The main message conveyed by this manuscript is that many VIP-expressing interneurons respond to reward and punishment but also show regional differences. The conclusions drawn are generally supported by the data, but some claims and interpretations require further attention and clarification.
-

Reviewer #1 (Public Review):
Building upon the previous evidence of activation of auditory cortex VIP interneurons in response to non-classical stimuli like reward and punishment, Szadai et al., extended the investigation to multiple cortical regions. Use of three-dimensional acousto-optical two-photon microscopy along with the 3D chessboard scanning method allowed high-speed signal acquisition from numerous VIP interneurons in a large brain volume. Additionally, activity of VIP interneurons in deep cortical regions was obtained using fiber photometry. With the help of these two imaging methods authors were able to extract and analyze the VIP cell signal from different cortical regions. Study of VIP interneuron activity during an auditory go-no-go task revealed that more than half of recorded cortical VIP interneurons were responding to …
Reviewer #1 (Public Review):
Building upon the previous evidence of activation of auditory cortex VIP interneurons in response to non-classical stimuli like reward and punishment, Szadai et al., extended the investigation to multiple cortical regions. Use of three-dimensional acousto-optical two-photon microscopy along with the 3D chessboard scanning method allowed high-speed signal acquisition from numerous VIP interneurons in a large brain volume. Additionally, activity of VIP interneurons in deep cortical regions was obtained using fiber photometry. With the help of these two imaging methods authors were able to extract and analyze the VIP cell signal from different cortical regions. Study of VIP interneuron activity during an auditory go-no-go task revealed that more than half of recorded cortical VIP interneurons were responding to both reward and punishment with high reliability. Fiber photometry data revealed similar observations; however, the temporal dynamics of reinforcement stimuli-related response in mPFC was slower than in the auditory cortex. The authors performed detailed analysis of individual cell activity dynamics, which revealed five categories of VIP cells based on their temporal profiles. Further, animals with higher performance on the discrimination task showed stronger VIP responses to 'go trials' possibly suggesting the role of VIP interneurons in discrimination learning. Authors found that reinforcement related response of VIP interneurons in visual cortex was not correlated with their sensory tuning, unveiling an interesting idea that VIP interneurons take part in both local as well as global processing. These observations bring attention to the possible involvement of VIP interneurons in reinforcement stimuli-associated global signaling that would regulate local connectivity and information processing leading to learning.
The state-of-the-art imaging technique allowed authors to succeed in imaging VIP interneurons from several cortical regions. Advanced analyses revealed the nuances, similarities and differences in the VIP activity trend in various regions. The conclusions about reinforcement stimuli related activity of VIP interneurons made by the authors are well supported by the results obtained, however some claims and interpretations require more attention and clarification.
-

Reviewer #2 (Public Review):
In recent years the activity of cortical VIP+ interneurons in relation to learning and sensory processing has raised great interest and has been intensely investigated. The ability of VIP+ interneurons in the auditory cortex to respond to both reward and punishment was already reported a few years ago by some of the authors (Pi et al., 2013, Nature). However, this work importantly adds to their previous study demonstrating a largely similar and synchronous response of a large fraction of these interneurons across the neocortex to salient stimuli of different valence during the performance of an auditory discrimination task.
An additional strength of this study is the analysis and identification of the general pattern of VIP+ interneuron responses associated to specific behaviors in the different layers of …
Reviewer #2 (Public Review):
In recent years the activity of cortical VIP+ interneurons in relation to learning and sensory processing has raised great interest and has been intensely investigated. The ability of VIP+ interneurons in the auditory cortex to respond to both reward and punishment was already reported a few years ago by some of the authors (Pi et al., 2013, Nature). However, this work importantly adds to their previous study demonstrating a largely similar and synchronous response of a large fraction of these interneurons across the neocortex to salient stimuli of different valence during the performance of an auditory discrimination task.
An additional strength of this study is the analysis and identification of the general pattern of VIP+ interneuron responses associated to specific behaviors in the different layers of the neocortex depth.
Interestingly, the authors also identified using cluster analysis 5 different classes of VIP+ interneurons, based on the dynamic of their responses, that were unequally distributed in distinct cortical areas.
This is a well performed study that took advantage of a cutting-edge imaging approach with high recording speed and good signal-to-noise ratio. Experiments are well performed and the data are properly analyzed and nicely illustrated. However, one shortcoming of this paper, in my opinion, is the "case report" structure of the data. Essentially for each neocortical area the activity of VIP+ interneurons was analyzed only in one animal. This limits the assessment of the stability of the response/recruitment of these interneurons. I appreciate the high number of recorded VIP+ interneurons per area/animal and I do understand that it would be excessively laborious to perform 3D random-access two-photon microscopy in several mice for each cortical area. On the other hand, it would be important to have some knowledge of the general variability of the responses of these neurons among animals.
In conclusion, despite the findings described in this manuscript being generally sound, additional experiments are recommended to further substantiate the conclusions.
-

Reviewer #3 (Public Review):
In this study Szadai et al. show reliable, relatively synchronous activation of VIP neurons across different areas of dorsal cortex in response to reward and punishment of mice performing an auditory discrimination task. The authors use both a relatively fast 2 photon imaging, as well as fiber photometry for some deeper areas. They cluster neurons according to their temporal response profiles and show that these profiles differ across areas and cortical depths. Task performance, running behavior and arousal are all related to VIP response magnitude, as has been previously shown.
Methodologically, this paper is strong: the described imaging technique allows for fairly fast sampling rates, they sample VIP cells from many different areas and the analyses are sophisticated and touch on the most relevant points. …
Reviewer #3 (Public Review):
In this study Szadai et al. show reliable, relatively synchronous activation of VIP neurons across different areas of dorsal cortex in response to reward and punishment of mice performing an auditory discrimination task. The authors use both a relatively fast 2 photon imaging, as well as fiber photometry for some deeper areas. They cluster neurons according to their temporal response profiles and show that these profiles differ across areas and cortical depths. Task performance, running behavior and arousal are all related to VIP response magnitude, as has been previously shown.
Methodologically, this paper is strong: the described imaging technique allows for fairly fast sampling rates, they sample VIP cells from many different areas and the analyses are sophisticated and touch on the most relevant points. The figures are of high quality.
However, as the manuscript is now, the presentation could be clearer, the methods more complete and it is not clear whether their conclusions are entirely supported by the data.
The main issue is that reinforcement and arousal are hard to distinguish in this study. It is well known that VIP activity is correlated with arousal. And it is fairly clear that the reinforcement they use in this study - air puffs to the eye, as well as water rewards - cause arousal. It is possible that the reinforcer responses they observe in VIP neurons throughout all areas merely reflect the increases in arousal caused by these behaviorally salient events. They do discuss this caveat (albeit not fully convincingly) and in their abstract even state that the arousal state was not predictive of reinforcer responses. However their data clearly shows the tight relationship of the VIP reinforcer responses to both arousal (as measured by pupil diameter), as well as running speed of the animal. Both of these variables are well known to be tightly coupled to VIP activity.
Although barely mentioned, the authors do appear to sometimes present uncued reward (Figure S2F). If responses were noticeably different from the same events in the task context (as actual reinforcers) this could at least hint towards the reinforcement signal being distinct from mere arousal. However, this data is only mentioned in one supplementary figure in a different context (comparison with PV cells) and neither directly compared to cued reward, nor is this discussed at all. Were uncued air puffs also presented? How do the responses compare to cued air puffs/punishment?
The imaging method appears well suited for their task, however the improvements listed in table S1 make the method appear far superior to existing methods in many aspects. Published or preprinted papers with 2 photon imaging of VIP populations (eg. from Scanziani lab (Keller et al.), Carandini lab (Dipoppa et al.), deVries lab (Millman et al.), Adesnik lab (Mossing et al.), which use the much more common resonant scanning, seem to be able to image 4-7 layers at 4-8Hz with a good enough SNR and potentially bigger neuronal yield of approximately 100-200 VIP cells, depending on the field of view. While not every single cell in a volume would be captured by these studies, the only main advantage of the here-used technique appears to be the superior temporal resolution.
Even though this is not mentioned at all, it certainly appears possible, that the accousto-optical scanning emits audible noise. In this case it would be good to know the frequency range and level of this background noise, whether there are auditory responses to the scanning itself and if it interferes with the performance of the animals in the auditory task in any way. If this is not the case, this should probably simply be mentioned for non-experts.
The authors show a strong correlation between task performance (hit rate) and the response to the auditory cue on hit trials. Was there any other significant correlations of VIP cells' responses to other trial types? Was reinforcer response correlated to behavioral variables at all?
-