Beyond the Focus of Expansion: Retinal curl as a functional signal for heading estimation
Curation statements for this article:-
Curated by eLife
eLife assessment
This study provides an important and biologically plausible account of how human perceptual judgments of heading direction are influenced by a specific pattern of motion in optic flow fields known as retinal curl. By combining psychophysical experiments and neural modeling, the authors demonstrate that what was previously considered an incidental "nuisance" signal actually serves as a functional control signal for estimating heading and steering toward a fixated target. While the evidence for the role of curl signals is convincing and advances our understanding of vision-based navigation, the work's impact would be strengthened by situating these findings among other cues that contribute to heading estimation, and by clarifying both the time course of these computations and their generalizability across different navigational contexts.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
Prevailing models aiming at explaining heading assume that humans need to recover the Focus of Expansion (FOE) while filtering out rotational flow (curl) caused by eye movements. We propose an alternative: the visual system utilizes retinal curl directly to estimate heading, rendering the explicit recovery of the FOE unnecessary. Stationary participants viewed simulated walking paths on a large screen while fixating on ground targets at varying eccentricities—a natural behavior inducing sustained retinal curl. Participants continuously reported perceived heading. To isolate the role of rotational flow, we employed a real-time manipulation that kept translational flow constant while the foveal curl component was either intact, cancelled, or overcancelled. Under natural conditions, participants exhibited systematic heading biases opposite the direction of gaze. Crucially, these biases vanished in the ‘cancelled curl’ condition, identifying retinal curl as the specific driver of perceptual bias. We modeled these results using a simple feedback controller and a ring-attractor neural network featuring gaze-contingent inhibition and a ‘straight-ahead’ prior. These findings suggest the brain exploits the geometry of gaze stabilization to simplify navigation, treating retinal curl as a functional signal rather than noise to be filtered.
Article activity feed
-

-

-

eLife assessment
This study provides an important and biologically plausible account of how human perceptual judgments of heading direction are influenced by a specific pattern of motion in optic flow fields known as retinal curl. By combining psychophysical experiments and neural modeling, the authors demonstrate that what was previously considered an incidental "nuisance" signal actually serves as a functional control signal for estimating heading and steering toward a fixated target. While the evidence for the role of curl signals is convincing and advances our understanding of vision-based navigation, the work's impact would be strengthened by situating these findings among other cues that contribute to heading estimation, and by clarifying both the time course of these computations and their generalizability across different …
eLife assessment
This study provides an important and biologically plausible account of how human perceptual judgments of heading direction are influenced by a specific pattern of motion in optic flow fields known as retinal curl. By combining psychophysical experiments and neural modeling, the authors demonstrate that what was previously considered an incidental "nuisance" signal actually serves as a functional control signal for estimating heading and steering toward a fixated target. While the evidence for the role of curl signals is convincing and advances our understanding of vision-based navigation, the work's impact would be strengthened by situating these findings among other cues that contribute to heading estimation, and by clarifying both the time course of these computations and their generalizability across different navigational contexts.
-

Reviewer #1 (Public review):
Summary:
This carefully executed study uncovers the functional relevance of curl signals that impinge on the retina every time an observer's gaze direction and movement direction are not aligned.
Strengths:
This finding is important, highlighting the functional role of an abundant incidental signal (curl in retinal motion) that has thus far believed to be a nuisance that needs to be filtered out of the retinal motion stream.
The study's evidence is compelling: a combination of psychophysical experiments and critical manipulations, control theory and neural modeling, which together make an internally consistent and biologically plausible case for the role of curl signals in estimating heading direction.
This study uncovers the functional relevance of curl signals that occur on the retina when an observer is …
Reviewer #1 (Public review):
Summary:
This carefully executed study uncovers the functional relevance of curl signals that impinge on the retina every time an observer's gaze direction and movement direction are not aligned.
Strengths:
This finding is important, highlighting the functional role of an abundant incidental signal (curl in retinal motion) that has thus far believed to be a nuisance that needs to be filtered out of the retinal motion stream.
The study's evidence is compelling: a combination of psychophysical experiments and critical manipulations, control theory and neural modeling, which together make an internally consistent and biologically plausible case for the role of curl signals in estimating heading direction.
This study uncovers the functional relevance of curl signals that occur on the retina when an observer is moving, and gaze is not straight ahead. The experimental and modeling results clearly go beyond previous studies and significantly advance our understanding of vision-based navigation.
Another clear strength is that the study uses tightly controlled experimental manipulation to provide strong test cases for the hypothesis that curl is used for visual navigation. These conditions are important to constrain the proposed model (and future models) of heading control.
The modeling is very clearly described, and the modeling and analysis code is published and freely available. The authors go beyond a back-of-the-envelope control model and show how it might be implemented at the neural-circuit level. The model is biologically plausible.
Weaknesses:
The discussion would benefit from an extension of the implications of the study and predictions of their model.
-

Reviewer #2 (Public review):
This study examines how curl in the retinal flow field can be used as a control variable for estimating and controlling the heading of a moving observer. The basic idea (which is not entirely new, see Matthis et al. 2022) is that translation along a path with eccentric gaze (meaning that the subject is not heading toward the point they are looking at) produces a pattern of optic flow on the retina with a rotational component around the point of fixation (which can be captured by the mathematical "curl" operator). The sign and magnitude of retinal curl vary with heading relative to the point of fixation, such that curl can be used as a control variable to steer rightward or leftward to move toward the fixated target. The authors perform behavioral experiments and show that there are biases in perceived …
Reviewer #2 (Public review):
This study examines how curl in the retinal flow field can be used as a control variable for estimating and controlling the heading of a moving observer. The basic idea (which is not entirely new, see Matthis et al. 2022) is that translation along a path with eccentric gaze (meaning that the subject is not heading toward the point they are looking at) produces a pattern of optic flow on the retina with a rotational component around the point of fixation (which can be captured by the mathematical "curl" operator). The sign and magnitude of retinal curl vary with heading relative to the point of fixation, such that curl can be used as a control variable to steer rightward or leftward to move toward the fixated target. The authors perform behavioral experiments and show that there are biases in perceived heading that seem to be largely governed by retinal curl. They also show that a simple controller model can use curl to steer toward a target, and they provide a neural network model that provides a biologically plausible implementation of the controller (although there are some questions about that).
There is a core of interesting work here that I think can be important to the field. However, there is a lack of clarity on several important fronts, including design of the behavioral experiments, presentation of the behavioral data, conceptual framing of what curl can and cannot do, etc. Equally importantly, the manuscript is not written in a manner that will make it accessible to most vision scientists. I consider myself to be pretty knowledgeable about optic flow, and I had to read most of the manuscript 3 or 4 times to be able to understand the bulk of it. And my experience is that most vision scientists do not understand optic flow well, so I fear that most of the readers that the authors should want to reach would struggle to understand the work. As written, this is mainly going to make an impact on a handful of optic flow gurus. Thus, I consider that this manuscript will need a major overhaul to clarify important issues and make it more accessible.
Major issues:
(1) The manuscript contains inconsistent, if not misleading, messaging about what information retinal curl does, and does not, provide regarding heading estimation. In the Abstract, the authors state: "We propose an alternative: the visual system utilizes retinal curl directly to estimate heading, rendering the explicit recovery of the FOE unnecessary." Based on my understanding of the rest of the manuscript, I find this statement to be a misrepresentation for two main reasons:
a) To "directly estimate heading" relative to what? When not qualified, most people interpret "heading" to mean an observer's heading relative to the world (or some allocentric reference frame). But retinal curl only gives information about an observer's heading relative to the point on which their eyes are fixated. Moreover, that point of fixation will change every few hundred milliseconds in natural viewing, so the retinal curl will change with each new fixation even as heading relative to the world remains unchanged. So I think most readers would grossly misinterpret the claim that retinal curl can be used "directly to estimate heading". Indeed, in the authors' controller model, the initial heading needs to be given, and then the controller can work. But from where does the visual system get the initial heading, since it does not come from curl? These issues are left hanging. Thus, while curl can provide a very useful input for steering toward a fixated target, other signals are needed to estimate heading relative to the world. This has to be made much clearer early on, and a conceptual schematic diagram might help. Also, the authors generally do not specify the reference frame of the variables they are talking about, leaving lots of room for misinterpretations. It should be clear each time they are talking about a variable, such as heading, whether it is relative to the fixation target, body, world, etc.
b) It seems to me that retinal curl will depend on other variables, in addition to heading relative to the fixation target. For example, it seems to me that the magnitude of retinal curl will depend on self-motion speed, the depth structure of the scene, the angle of elevation of the fixated target, and perhaps others. This is not discussed at all, and many readers would get the misguided impression that there is a 1:1 mapping from curl to heading (relative to fixation). If I am right that this is not correct, it means that retinal curl can tell the observer whether to steer right or left to move toward the fixated target, but it cannot tell them how much to steer. Indeed, in the authors' controller model, there is a free parameter that calibrates curl to angle. It makes sense that this works to fit trajectory data that are given from a fixed environment, but it is unclear how the brain would use retinal curl to control steering when these other variables are uncertain or changing unpredictably. Moreover, how does the system change the mapping from curl to steering command as the location of fixation changes relative to the current heading? These are issues that need to be brought up in framing the problem and discussed at some length. If the authors can show mathematically that retinal curl is only dependent on heading (relative to fixation) and not any of these other variables, it would be very valuable to show the equations for this relationship.
(2) The description of the behavioral experiment and presentation of behavioral data leaves a lot to be desired.
a) First, it is stated (line 158) that "Participants continuously reported their perceived direction of self-motion while maintaining fixation on the yellow dot." Again, the reference frame is completely unspecified. Participants were reporting their perceived heading relative to what? The fixation target? The world? What exactly were the instructions given to the subjects to perform the task? Based on the description of how perceived paths are computed (line 166-), it seems to be presumed that subjects are reporting their heading relative to the world because those angles are then converted into x and z coordinates in what I presume is a world-centered reference frame. But how do we know that subjects are accurately reporting their heading relative to the world? What if they are biased in their reports by the location of the fixation target relative to the scene, or by some other reference signal? Is it possible for the authors to rule out the possibility that perceptual biases seen in the unaltered curl condition result from observers not fully adopting the assumed reference frame of the task? If this cannot be firmly excluded, it seems to create problems for the rest of the study.
b) I also feel that there is a mismatch between what the behavioral task requires and what the controller model does. Subjects are apparently asked to report their heading relative to the world, but the controller model only controls their heading relative to the point that they are fixating. I understand how this is resolved in the model, but I think this type of distinction is buried and will not be apparent to most readers. Again, the reference frames of what is being measured and controlled need to be specified explicitly in all parts of the paper, and the authors need to explain how the system would combine curl-based control with some other measures of (at least initial) heading for world-centered heading to be computed. All of the assumptions need to be clearly specified.
c) In addition, I found it frustrating that the authors never present raw perceptual data from the observers. Rather, in Figure 2, we see reconstructed trajectories that are perfectly smooth with no indications of noise whatsoever. Since these paths are computed from the perceptual reports, there must be some noise inherent in them. The figures should represent this uncertainty somehow, and it should be explained how these perfectly smooth trajectories are obtained.
(3) "...the magnitude of retinal curl in the fovea can specify the body trajectory relative to gaze (Matthis et al., 2022)." The main idea put forward by the authors here seems to overlap heavily with this statement that they attribute to Matthis et al. 2022. While I think this paper still adds importantly to the topic, the authors do not discuss how their findings are different from those of Matthis et al. 2022, why they are an important extension, etc. Readers should not have to go read this other paper to have any idea how the present findings are placed in importance relative to the literature.
(4) The analysis and treatment of eye movements is extremely weak. The authors discarded trials for which gaze deviated from the fixation point by more than 3 degrees (which is a LOT given that the eye speeds are generally in the neighborhood of 0.5 deg/sec), and they provide basic stats on the distribution of positions. But this largely misses the point: it is not small position errors that are likely to matter, but rather velocity errors. Even a small amount of retinal slip of the target while it is being pursued will cause image motion that is going to alter the optic flow field around the fixation target. So, for example, the retinal curl field may no longer be centered on the fixation target. How do we know that some of the perceptual biases are not influenced by image motion resulting from imperfect tracking of the fixation target? This needs to be analyzed and discussed.
(5) I found the sections of text comparing the separate and joined fits (starting line 287) to be a bit too rosy. The authors show the separate fits in the main text, and it is not very surprising that these fits are good, given that the model has 30 parameters, and these data are pretty low-dimensional. The authors only show the joined fits in the supplement, and they say that they are almost as good as the separate fits (indeed, they are better in a model comparison sense, but this is 30 parameters vs. 2 parameters). However, when I look at the fits of the joined model in the supplement, I don't find them to be very impressive. In particular, the model grossly misses the data for the straight paths for several subjects (e.g., id5, id6, id8, id10). And fitting the straight paths would presumably be easiest. This implies that the joined model is really missing something and that fitting the curved paths interacts strongly with fitting the data for different fixation target locations on the straight path. I think that the authors should discuss the results a bit more soberly and tone down their conclusions here.
(6) The section of the paper on neural simulations (starting line 387) has a few weaknesses. First, why are only straight paths simulated here? This does not seem to provide a very rigorous test of the model. Second, it is awkward that the simulation results are presented in units of pixels, rather than degrees. Third, the authors seem to downplay the fact that the neural estimates of heading seem to oscillate rather wildly (over a range of hundreds of pixels, whatever that means, see especially Figure S16). It was far from clear to me how an estimate of heading with these large oscillations is useful. It would seem to require that heading estimates are integrated over substantial lengths of time to be reliable. It was therefore unclear how the model produces such smooth paths from these oscillating estimates.
-

Reviewer #3 (Public review):
Summary:
This manuscript uses a novel paradigm to demonstrate that rotational motion patterns in the retinal image, called curl, directly influence perception of heading direction. This means that it is not necessary to recover the focus of expansion, defined by the point of zero motion when moving along a straight trajectory toward a target, as is commonly thought.
Strengths:
It has long been accepted that the focus of expansion of the optic flow field generated by self-motion is used to guide heading direction. While there have been many challenges to the need to recover the focus of expansion when gaze is not in the direction of travel, it is still not well understood how retinal motion patterns contribute to heading perception. Recent work has demonstrated the complexity of the retinal motion patterns …
Reviewer #3 (Public review):
Summary:
This manuscript uses a novel paradigm to demonstrate that rotational motion patterns in the retinal image, called curl, directly influence perception of heading direction. This means that it is not necessary to recover the focus of expansion, defined by the point of zero motion when moving along a straight trajectory toward a target, as is commonly thought.
Strengths:
It has long been accepted that the focus of expansion of the optic flow field generated by self-motion is used to guide heading direction. While there have been many challenges to the need to recover the focus of expansion when gaze is not in the direction of travel, it is still not well understood how retinal motion patterns contribute to heading perception. Recent work has demonstrated the complexity of the retinal motion patterns during natural walking, where body motion adds a rotational component. A rotational component also results from curved paths as well as gaze off the direction of travel. This rotational component is called curl. The primary contribution of this manuscript is to demonstrate convincingly that curl influences perception of heading, and that it is not necessary to recover the focus of expansion.
A strength of the manuscript is that realistic retinal motion patterns are generated by recording the image sequences generated by a walker in a virtual environment, and then using those patterns as stimuli in the experiment. This allows the creation of the more complex flow patterns that are a consequence of the bob and sway of natural walking, which are often considered a minor factor. The elegant experimental design allows direct manipulation of the curl signal, and this in turn directly influences measured heading perception. Another strength is that the authors ground their findings in control theory and neural computations, using a model that produces human-like path trajectories.
The study is timely, given the long history of this question, together with the growing understanding of the complexity of naturally generated retinal motion and the absence of direct evidence for the way that these motion patterns are used in heading perception. It adds an important piece of evidence for how retina-centered optic flow may be used by the visual system, which is critical for our understanding of motion processing in the brain.
Weaknesses:
The primary limitation of the paper is that it avoids discussion of some of the inevitable complexities of heading perception. The main issue is what exactly is meant by heading. Different behaviors evolve over different timescales. The geometry of retinal motion defines instantaneous heading, which varies widely through the gait cycle. Time-varying information like this is known to be important in the momentary control of balance. Heading can also be thought of as steering the body toward a distant goal, which evolves over longer timescales. The current manuscript appears to be concerned with heading information integrated over a few seconds and seems to provide evidence that heading is indeed integrated over the gait cycle. The issue of the time scale of the computation is touched on, but it is not related to how it might be used in normal walking or what situations it might apply to. Steering toward a distant goal during walking is not a very difficult problem and may not require evaluation of retinal motion, but control of balance is more challenging and may depend critically on curl. Consequently, the timescale of the computation needs to be considered in order to understand what is meant by heading.
-

Author Response:
Public Reviews:
Reviewer #1 (Public review):
We appreciate Reviewer #1’s very positive feedback. Incorporating the perspective of ‘incidental’ sensory signals is a valuable suggestion that aligns perfectly with our findings. We agree that this perspective significantly strengthens the impact of our paper.
In the revised version, we will update the manuscript to bridge these perspectives (the functional role of incidental” sensory signals and the role of retinal flow in navigation). In addition we will elaborate on the potential predictions of the model and possible manipulations that might affect the integration between sensory evidence (curl signal) and straight-ahead prior.
Reviewer #2 (Public review):
We appreciate the reviewer’s feedback regarding the formalization of our reference frames. We agree that certain …
Author Response:
Public Reviews:
Reviewer #1 (Public review):
We appreciate Reviewer #1’s very positive feedback. Incorporating the perspective of ‘incidental’ sensory signals is a valuable suggestion that aligns perfectly with our findings. We agree that this perspective significantly strengthens the impact of our paper.
In the revised version, we will update the manuscript to bridge these perspectives (the functional role of incidental” sensory signals and the role of retinal flow in navigation). In addition we will elaborate on the potential predictions of the model and possible manipulations that might affect the integration between sensory evidence (curl signal) and straight-ahead prior.
Reviewer #2 (Public review):
We appreciate the reviewer’s feedback regarding the formalization of our reference frames. We agree that certain definitions were implicitly assumed rather than explicitly stated. We will revise the manuscript to provide all necessary self-contained information, ensuring that the geometry of the task response and the definition of heading are unambiguous. Also, we will address the gap between the task response (in world coordinates) and the functional role of the controller, as well as the other points raised by the reviewer.
Major issues:
(1a), (2a) Clarification of Reference Frames
The reviewer asks: “To ‘directly estimate heading’ relative to what?”
In our study, participants were instructed to report their “perceived direction of self-motion” by aligning a rotational encoder (steering wheel) with the direction they felt they were moving within the 3D simulated scene. Consequently, participants reported their instantaneous heading in a world-centered reference frame, from which the 3D trajectories were reconstructed. Since the reviewer had to infer this information, it should be clarified to ensure it is immediately evident.
Participants were informed that the initial heading (i.e. θ0 in our controller nomenclature) was oriented “straight ahead” relative to their body which was aligned longitudinally with the experimental room. We will modify Figure 1B and revise the Methods section to explicitly clarify this initial alignment and the instructions provided to participants.
In the revised manuscript, we will clarify that while the participant’s report is world-centered, the retinal curl provides a gaze-relative heading signal. Although this was already mentioned, we will emphasize this point. In natural navigation toward a fixated target, a world-centered vector is often unnecessary; an error signal indicating heading relative to fixation is sufficient (as the reviewer also notes). However, the initial alignment of the heading within the 3D scene allows the brain to “calibrate” this internal controller, mapping the retinal curl signal onto the 3D world coordinates required for the task.
The reviewer also asks how we can be certain that participants were reporting in world coordinates rather than an alternative frame, such as “heading relative to the fixation target.” We believe our “Cancelled Curl” (and over-cancelled) conditions provide the most compelling evidence to rule out this alternative. In these conditions, the physical position of the fixation target in the scene remained identical to the unaltered flow condition. If participants were simply reporting heading relative to the fixation target’s spatial location, the observed biases should have persisted regardless of the flow manipulation. Instead, the bias vanished when the curl was removed. This causal evidence proves that the bias is driven by the retinal motion signal (curl) rather than the spatial orientation of the eyes or the target’s position in the scene. Furthermore, the temporal evolution of the response supports a world-centered integration. For simulated straight paths, the perceived heading remains straight for the first few seconds (consistent with the initial world-centered alignment), with biases only emerging after approximately 3 seconds of integration (a point we elaborate on in our response to Reviewer #3). Had participants been responding based on a simple gaze-relative reference frame from the onset, these biases would have manifested significantly earlier. We will incorporate these points into the revised Discussion to better frame our findings alongside other cues, such as the Focus of Expansion (FOE), that contribute to heading estimation.
(1b) The reviewer notes that we must be clear about the relationship between curl and heading (relative to fixation) and the variables that affect curl.
Beyond the discrepancy between heading (θ) and gaze (ψ), curl is geometrically determined by translational self-motion speed (υ), eye height (h), and pitch (α). More specifically curl = (υ sin_ψcos α)/h). The derivation will be included in the Supplementary Information. Since h = d_sinα, where d is the 3D distance to the fixation point, we could express cos α as a function of distance. Certainly, there is not a 1:1 map from curl signal to heading relative to gaze (e.g. θ – ψ). Participant would need to know υ and eye height plus extra-retinal information. Frenz et al (2003, Vis Res.) showed that people can estimate self-motion directly from optic flow, across different simulated eye height and gaze angle; extra-retinal information can, in addition, provide knowledge to (ψ) and (α). It is then plausible that the visual system can use and transform the curl signal from a qualitative directional cue (i.e. steering left or right of fixation) into a quantitative steering command. By combining curl with knowledge of gaze orientation and eye height, the visual system can resolve ambiguities in the flow field and utilize curl as a more precise error signal for locomotor control. These aspects will be included in the new version.
(2b) Mismatch between task and controller
We thank the reviewer for this point. We have addressed the alignment of the reference frames in our response to Issues 1a and 2a. Once the initial orientation () is established in the world frame, the controller model generates steering adjustments that directly translate into heading predictions within that same world reference frame. By treating the perceptual report as an output of the locomotor controller, we resolve the discrepancy between the steering task and the reported heading.
(2c) No raw data provided
We respectfully disagree with the reviewer’s interpretation regarding data smoothing. The thin lines in Figure 2 represent the mean 3D paths derived directly from the response variable (θ0) across trials of identical conditions for each participant (as detailed in the ‘Computation of Perceived Path’ section). No smoothing or filtering has been applied to these plotted trajectories other than computing the mean across trials. We also wish to remind the reviewer that the raw data and analysis code remain publicly accessible for further inspection. Regarding the visual representation: in earlier versions of the manuscript, we included shaded 95% Confidence Intervals (CIs) in Figure 2. However, this addition rendered the plot overly cluttered and obscured the individual trajectories. We therefore elected to present individual participant means (thin lines) alongside group averages (thick lines) to emphasize inter-subject variability. For clarity, the 95% CIs are explicitly displayed in Figure 3, where the data density is more conducive to shaded areas.
(3) Difference with Matthis et al (2022)
While Matthis et al. (2022) described the existence of retinal curl during walking and which information can provide relative to gaze, Our paper provides the causal link, since we manipulate in real-time (the ‘cancelled & overcancelled curl’ condition) providing the critical evidence that perceived heading is affected by this signal.
(4) Eye movements analysis
We thank the reviewer for noting that retinal slip (velocity error) is a more critical metric than positional gaze error. We agree that tracking inaccuracies can introduce translational noise into the flow field. The 3° threshold was established based on the eye tracker’s specifications and the naturalistic setup (1-meter viewing distance without head stabilization). Across all participants, the mean positional error ranged from 1.016° to 1.5° (1 deg is 2.08 cm in our setup). We also calculated retinal slip values, which ranged from 0.12 to 0.27 deg/s (X dimension) and 0.12 to 0.23 deg/s (Y dimension). These values are comparable to natural oculomotor drift (Kowler et al., 1979) and are understandably small given the low velocity of the fixation target. Consequently, it is highly unlikely that retinal slip influenced the results. Furthermore, assuming that tracking error remained consistent across fixation conditions, any present retinal slip cannot explain why the bias followed the retinal curl manipulation as predicted by the controller. We therefore consider retinal slip to be an unlikely confounding factor.
(5) the separate and joined fits
We thank the reviewer for the opportunity to clarify the logic behind our modeling choices. We acknowledge that the “separate fits” are inherently less informative due to the high number of free parameters relative to the data. Our primary scientific goal was not to achieve perfect descriptive accuracy via 30 parameters, but to test a specific functional hypothesis through the “joint fit.”
The Logic of the Joint Fit:
We agree with the reviewer that the joint fit misses some paths in some conditions. Of course, the joint fit reflects a significant compromise. The “Gain” (the weighting of the curl signal) is likely not a static constant but is dynamically tuned based on task demands, confidence in the visual signal, simulated speed, and so on. By using a single Gain parameter, we intentionally ignore this contextual variability to see how much of the behavior can be explained by a “minimalist” controller. In this sense, the 2-parameter joint model is a deliberate attempt to test this limit. By forcing a single Gain parameter to account for all conditions across both straight and curved paths within one flow manipulation (e.g. unaltered flow) we are asking if a single, fixed linear relationship between retinal curl and steering effort/gain can explain the results. We view the joint fit not as a “perfect” model, but as a stronger test of the curl-based control theory. The fact that a 2-parameter model can capture the direction and scale of biases across such a diverse set of conditions (straight/curved paths, five fixation eccentricities) suggests that retinal curl is a robust signal. Upon closer analysis, these discrepancies between the joint model and the data are most pronounced in the over-cancelled condition which is the one when sensory evidence becomes more ecologically inconsistent with the extra-retinal information (gaze direction). While the joint fit successfully demonstrates that a single parameter can capture the general functional role of curl, it fails to account for the complex sensory re-weighting that occurs in ecologically inconsistent conditions (like ‘over-cancelled’ flow). We will update the manuscript to discuss these limitations, framing the model as a parsimonious first-order approximation rather than a complete description of human heading perception based on a minimal set of parameters.
(6) On the neural simulations
We acknowledge that the presentation of the neural model requires more clarity regarding its objectives and its relationship to the behavioral data.
We first wish to clarify the intended scope of the neural ring-attractor model. Our primary goal was not to provide a comprehensive account of behavioral performance across all conditions (which is the role of the controller model), but rather to demonstrate a biologically plausible mechanism that explains the emergence of the “Opposite-to-Gaze” bias. While the controller demonstrates that the bias follows a specific control law, the neural model shows how such a law can emerge from known primate neurophysiology, specifically, spiral-tuned MSTd neurons, gaze-contingent inhibition, and an egocentric “straight-ahead” prior.
Why Straight Paths are Sufficient for this Objective. The reviewer asks why only straight paths were simulated. In our study, the straight-path condition with eccentric gaze is the purest test of the bias mechanism. Simulating the straight paths allowed us to isolate the interaction between foveal inhibition and the straight-ahead prior without the confounding variable of path-curvature flow. Given the complexity of the neural network’s parameter space, we focused on these conditions to provide a clear neuro-plausible explanation.
Units: Pixels vs. Degrees. We acknowledge that the use of “pixels” in the plots of internal neural dynamics may appear awkward. The neural network operates on input stimuli that are defined by the pixel resolution of the videos used in the simulations, we used pixels as the native coordinate system to describe the movement of activity peaks within the network’s internal “map.”
Behavioral Output (Meters): Importantly, the final heading estimates produced by the network are not left in pixels. We use a pinhole camera model to reconstruct the 3D trajectories from the neural activity. These results are expressed in meters, allowing for a direct comparison with the human behavioral data.
Addressing Wild Oscillations and Smooth Paths. The oscillations observed in the instantaneous heading estimates reflect the stochastic nature of the population peak when tracking high-frequency sensory inputs. In our model, the synaptic time constant (τ) was kept relatively small to ensure a fast, low-latency response to changes in self-motion. While increasing τ would have produced smoother internal dynamics, it would also have introduced delays into the control loop. Instead, we chose to maintain this high sensory responsiveness and applied a temporal moving average later to the network’s decoding to reconstruct the 3D trajectories.
In addition, the neural activity over time is shown in two ways: the heatmap shows the neuron with preferred heading (one can see more oscillations, specially when the fixation point is closer to the centre (eccentricities -2 and 2), due to larger competition between the sensory evidence and the straight-ahead prior. The other way is the decoded heading. In the ring-attractor model, the decoded heading
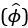 is not determined by a single neuron but is calculated using a population vector average (equation 19). By summing across the entire population, the decoder effectively integrates sensory evidence from many neurons simultaneously. One can appreciate (see e.g. Fig. 5B) that averaged decoding, leads to a smoother resulting estimate (the white dashed line, whose visibility will be improved in the revised version). Behavioral work by Burr and Santoro (2001) suggests that global motion signals (divergence and rotation in optic flow) are integrated over much longer timescales—roughly 1000ms to 3000ms—compared to local motion units (~200ms).
is not determined by a single neuron but is calculated using a population vector average (equation 19). By summing across the entire population, the decoder effectively integrates sensory evidence from many neurons simultaneously. One can appreciate (see e.g. Fig. 5B) that averaged decoding, leads to a smoother resulting estimate (the white dashed line, whose visibility will be improved in the revised version). Behavioral work by Burr and Santoro (2001) suggests that global motion signals (divergence and rotation in optic flow) are integrated over much longer timescales—roughly 1000ms to 3000ms—compared to local motion units (~200ms).See also our comment on temporal integration in the responses to reviewer #3.
Reviewer #3 (Public review):
We thank Reviewer #3 the comments regarding the definition of heading at different time scales, the role of the gait cycle, and the temporal integration of the curl signal. They will help us refine the manuscript’s core arguments.
We agree that “heading” must be precisely defined within the context of the differing temporal demands of balance and steering. While instantaneous retinal motion provides the high-frequency feedback necessary for momentary postural adjustments and balance, our study is concerned with heading as a gaze-relative signal used for the continuous control of a locomotor trajectory. As such, we will revise the manuscript to specify that the perceived heading measured in our task reflects a signal integrated over the gait cycle to filter out the oscillatory noise induced by head bob and sway.
The reviewer correctly notes that gait-induced head bob and sway produce high-frequency oscillations in the curl signal, yet our behavioral results show smooth, slowly evolving biases. The visual system does not react to “instantaneous” curl, which would lead to jittery, unstable heading estimates. Instead, it integrates flow over a timescale roughly commensurate with a full gait cycle (~500–1000ms). This implies a significant temporal integration process. This temporal integration is consistent with evidence (Burr and Santoro,2001, Vis Res) indicating that optic flow signals (radial and rotational components) are integrated over windows of approximately up to 3 seconds to ensure perceptual stability. Neurally, this likely involves the projection from area MSTd to the Ventral Intraparietal area (VIP), a pathway where fast, eye-centered sensory inputs are transformed into stable, body-centered representations suitable for guiding long-term steering behavior (Chen et al. 2011, JNeurosci.). By grounding our definition of heading in these specific temporal and neural constraints, we aim to clarify how the visual system exploits retinal curl for goal-directed action in natural, dynamic environments and relate our findings to recent studies addressing the role of retinal motion on balance (Powell et al. 2026 Bioarx).
In our implementation, we explicitly address the high-frequency noise introduced by gait dynamics by smoothing the retinal curl signals computed from the stimulus videos before they are fed into the controller. This temporal filtering allows the fit of the controller’s prediction to the response data while remaining robust to the rapid fluctuations of head bob and sway. In contrast, the neural ring-attractor model would not require an external smoothing step; instead, the integration is an emergent property of the system’s architecture that can be controlled with different parameters. The dynamics of the synaptic weights and the characteristic “leak” in the population activity naturally implement a leaky integration of sensory evidence, ensuring that the decoded heading reflects a sustained estimate rather than an instantaneous response to visual noise.
-
