Involuntary feedback responses reflect a representation of partner actions
Curation statements for this article:-
Curated by eLife
eLife Assessment
This important study concerns the propagation of waves in bacterial biofilms, bridging active matter physics and bacterial biophysics. The experimental observations are solid, and the theoretical interpretation and model validation have been refined with revisions. This work will be of interest to microbiologists, biophysicists, and researchers studying collective behavior in biological systems.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
We have a remarkable ability to seamlessly and rapidly coordinate actions with others, from double dutch to dancing. Humans use high-level partner representations to jointly control voluntary actions, while other work shows lower-level involuntary feedback responses to sudden visual perturbations. Yet, it is unknown if a high-level partner representation can be rapidly expressed through lower-level involuntary sensorimotor circuitry. Here we test the idea that a partner representation influences involuntary visuomotor feedback responses during a cooperative sensorimotor task. Using two experiments and dynamic game theory predictions, we show that involuntary visuomotor feedback responses reflect a partner representation and consideration of a partner’s movement cost (i.e., accuracy and energy). Collectively, our results suggest there is top-down modulation from high-level partner representations to lower-level sensorimotor circuits, enabling fast and flexible feedback responses during jointly coordinated actions.
Article activity feed
-

-

-

eLife Assessment
This important study concerns the propagation of waves in bacterial biofilms, bridging active matter physics and bacterial biophysics. The experimental observations are solid, and the theoretical interpretation and model validation have been refined with revisions. This work will be of interest to microbiologists, biophysicists, and researchers studying collective behavior in biological systems.
-

Reviewer #1 (Public review):
Summary:
Overall, this is an interesting paper. The authors identify several experimental knobs that can perturb mechanical wave behavior driven by pili feedback. They frame these effects in terms of nonreciprocal interactions. While nonreciprocity could indeed play a role, it raises the question of whether mechanical feedback might also contribute. Phenomenological models can be useful, but the model currently lack direct mechanistic insight. It would be more compelling to formulate the model around potential mechanochemical feedback, which could help clarify the underlying microscopic mechanisms.
Strengths:
Report of mechanical waves in bacterial collectives, mechanism has potential application in multicellular context such as morphogenesis.
Weaknesses:
A minor concern about the language of 'left-right …
Reviewer #1 (Public review):
Summary:
Overall, this is an interesting paper. The authors identify several experimental knobs that can perturb mechanical wave behavior driven by pili feedback. They frame these effects in terms of nonreciprocal interactions. While nonreciprocity could indeed play a role, it raises the question of whether mechanical feedback might also contribute. Phenomenological models can be useful, but the model currently lack direct mechanistic insight. It would be more compelling to formulate the model around potential mechanochemical feedback, which could help clarify the underlying microscopic mechanisms.
Strengths:
Report of mechanical waves in bacterial collectives, mechanism has potential application in multicellular context such as morphogenesis.
Weaknesses:
A minor concern about the language of 'left-right asymmetry.' I believe the correct term is simply 'radial asymmetry' which is a distinct concept. Left-right is not well defined in the current context.
-

Reviewer #3 (Public review):
Summary:
The revised manuscript presents a compelling study of radially propagating metachronal waves on the surface of Pseudomonas nitroreducens biofilms, combining experiments with two theoretical descriptions (a local phase-oscillator model and an active solid/active gel model). The central experimental findings-spiral/target/planar wave patterns, their controllability via water/PEG/temperature perturbations, and the correlation between frequency gradients and propagation direction-remain highly interesting and relevant to both bacterial biophysics and active-matter physics. The revised manuscript also adds substantial new material, including additional analyses of defect dynamics and clearer discussion of the relationship between the two models. The study continues to have a strong interdisciplinary …
Reviewer #3 (Public review):
Summary:
The revised manuscript presents a compelling study of radially propagating metachronal waves on the surface of Pseudomonas nitroreducens biofilms, combining experiments with two theoretical descriptions (a local phase-oscillator model and an active solid/active gel model). The central experimental findings-spiral/target/planar wave patterns, their controllability via water/PEG/temperature perturbations, and the correlation between frequency gradients and propagation direction-remain highly interesting and relevant to both bacterial biophysics and active-matter physics. The revised manuscript also adds substantial new material, including additional analyses of defect dynamics and clearer discussion of the relationship between the two models. The study continues to have a strong interdisciplinary appeal and the potential to stimulate further work on collective oscillations in biological active media.
Strengths:
The authors have substantially addressed the major conceptual issue raised in the previous round by clearly distinguishing between nonreciprocity and frequency gradients / global asymmetry. This clarification significantly improves the theoretical interpretation and resolves an important source of confusion in the original version.
The revised manuscript also improves the connection between the phase-oscillator and active-solid descriptions. In particular, the authors now explain more explicitly how the phase variable is defined in the reduced oscillatory dynamics of confined biofilm motion, and they state that they added a schematic illustration and simulation details (including parameter values and the elastic-force definition) to improve reproducibility. This directly addresses one of my previous major concerns.
A notable improvement is the newly added defect-based analysis of waveform transitions (spiral -> target -> planar). The revised text argues that defect motility is a key control parameter, linked experimentally to moisture-dependent elasticity and theoretically to nonreciprocity / defect-pair stability. This provides a more concrete mechanistic bridge between experimental perturbations and the modeling framework than in the previous version.
The manuscript now gives a clearer experimental-theoretical narrative for how environmental manipulations (drying, water addition, PEG, heating) affect wave patterns through changes in effective elasticity and activity, including a useful distinction between short-timescale and long-timescale temperature effects. This added discussion strengthens the biological interpretation and makes the modeling assumptions easier to follow.
Weaknesses:
The main remaining limitation is the level of quantitative correspondence between theory and experiment. The revised manuscript now provides a stronger qualitative/mechanistic link, but the mapping between model parameters (e.g., effective coupling terms / elasto-active parameters) and directly measurable biofilm properties is still limited. The authors acknowledge this point, and I agree that it is technically challenging in the present system. However, this means the theoretical framework is currently most convincing as an effective mechanistic model rather than a quantitatively predictive one.
Relatedly, some conclusions about parameter-level control (especially in connecting moisture/temperature manipulations to specific model parameters) remain qualitative. I do not view this as fatal, but I recommend that the manuscript clearly state this scope and avoid overstating the quantitative predictive power of the theory.
Although the terminology has improved compared with the original version, the revised manuscript still uses "left-right asymmetry" in places where the underlying geometry and symmetry are more general (e.g., radial inward propagation in circular colonies). Since this wording was one of the original points of confusion, I suggest one final pass to ensure the symmetry language is consistently precise throughout the main text and figure captions.
-

Author response:
The following is the authors’ response to the original reviews.
eLife Assessment
This important study concerns the propagation of waves in bacterial biofilms, bridging active matter physics and bacterial biophysics. While the experimental observations are solid, the theoretical interpretation and model validation are currently incomplete and require further refinement. This work will be of interest to microbiologists, biophysicists, and researchers studying collective behavior in biological systems.
In the revised manuscript, we have added new experimental results that strengthen the connection between our observations and the modeling framework used to interpret the collective oscillations. We have not introduced a new theoretical model; rather, we employed established active matter models and sought to link the …
Author response:
The following is the authors’ response to the original reviews.
eLife Assessment
This important study concerns the propagation of waves in bacterial biofilms, bridging active matter physics and bacterial biophysics. While the experimental observations are solid, the theoretical interpretation and model validation are currently incomplete and require further refinement. This work will be of interest to microbiologists, biophysicists, and researchers studying collective behavior in biological systems.
In the revised manuscript, we have added new experimental results that strengthen the connection between our observations and the modeling framework used to interpret the collective oscillations. We have not introduced a new theoretical model; rather, we employed established active matter models and sought to link the observed phenomena to these frameworks. In particular, our new data demonstrate that the transition between the motile and biofilm-forming states specifically modulates the elasticity and elasto active coupling of the bacterial structure. This behavior is in excellent agreement with the predictions of the active solid model. All the experimental details are given below. We believe that the revised version of the manuscript now establishes this connection more clearly and convincingly.
Public Reviews:
Reviewer #1 (Public review):
Summary:
Overall, this is an interesting paper. The authors have found multiple experimental knobs to perturb a mechanical wave behavior driven by pilli feedback. The authors framed this as nonreciprocal interactions - while I can see how nonreciprocity could play a role - what about mechanical feedback? Phenomenological models are fine, but a lack of mechanistic understanding is a weakness. I think it will be more interesting to frame the model based on potential mechanochemical feedback to understand microscopic mechanisms. Regardless, more can be done to better constrain the model through finding knobs to explain experimental observations (in Figures 3, 4, 5, and 7).
We thank the reviewer for the positive assessment and for highlighting this important point. The reviewer is correct that the phenomenological Kuramoto-based model does not explicitly show the detailed cell–cell interactions. However, the active solid model is formulated on detailed elastic couplings and active forces, which inherently represent mechanical feedback within the biofilm structure. In this framework, nonreciprocity emerges naturally from the tensorial nature of active forces between bacteria—a concept already well established in the active matter literature. Importantly, this mechanism is purely mechanical and closely parallels nonreciprocal hydrodynamic interactions among active particles, which also arise from tensorial couplings.
In our system, elastic interactions within the biofilm matrix, combined with pilus-generated active forces, provide a natural origin for nonreciprocal interactions. To further validate this, we improved our imaging to record single-cell dynamics both at the colony edge and on the biofilm surface. (new supplementary Video). These experiments show that motile bacteria at the leading edge of the biofilm structure do not generate waves, whereas stationary bacteria within the biofilm display local oscillations within the elastic network. This observation supports the view that collective oscillations are a property of the elastic biofilm state rather than of freely motile cells.
Moreover, the main control parameter for these oscillations is the ratio between elastic strength and the active force generated by pili. In the active solid model, this ratio is captured by the parameter π and alpha terms. Experimentally, we can tune this ratio simply by adding or removing water from the biofilm, thereby modulating its elasto active coupling. We further motivated the controllability of this feature experimentally. We let the plate dry nonuniformly and observed that the transition between spiral target and plane waves could emerge spontaneously across the plate (see Figure 3a). This observation also states the importance of moisture in the biofilm. Starting from this point we established the connection between experimental observation and modelling. In our new simulations we also noticed that the transition from spiral to target wave is particularly driven by merging processes of different topological charges +/- 1 spiral pairs. This critical point was also confirmed by modelling which links the process to elasto active coupling. Further we supported our claim by imagining the edge and the biofilm structure. These new results clarify that elastic structure of the biofilm is critically important (Supplementary Figure 3). We have clarified this mechanistic link in the revised manuscript and rewritten the relevant sections to make this connection explicit.
Modification in the manuscript:
“To gain deeper insight into the mechanisms underlying wave formation, we imaged the dynamics of individual bacteria from the fingering regions toward the center of the biofilm. This distinction is critical because, unlike the biofilm center, the edges do not generate waves. We observed that bacteria near the fingering regions remain motile and exhibit collective flow. In contrast, bacteria at the biofilm center are surface-attached and undergo periodic lifting motions. This behavior strongly resembles Mexican-wave dynamics.”
“We further found that the central region of the biofilm is mechanically more elastic, whereas the edge regions—where wave formation is absent—are motile. These observations suggest that gradual biofilm maturation is a key factor that transforms motile bacteria into a periodically moving but spatially constrained state. Consistent with this picture, the PAO1 strain, which has a strong biofilm-forming capability, completely suppresses surface oscillations. In contrast, the PA14 strain exhibits intermediate behavior, sustaining a partial transition between motile and locally constrained dynamics. Remarkably, signatures of this transition and wave generation are already detectable at the earliest stages of finger formation.”
Strengths:
The report of mechanical waves in bacterial collectives. The mechanism has potential application in a multicellular context, such as morphogenesis.
We thank the reviewer for the positive assessment and for highlighting this potential broad impact of our findings.
Weaknesses:
My most serious concern is about left-right symmetry breaking. I fail to see how the data in Figure 6 shows LR symmetry breaking. All they show is in-out directionality, which is a boundary condition. LR SM means breaking of mirror symmetry - the pattern cannot be superimposed on its mirror image using only rigid body transformations (translation and rotation) - as far as I am aware, this condition is not satisfied in this pattern-forming system.
We thank the reviewer for pointing out this critical issue. We acknowledge that we overlooked the distinction between biological and physical definitions of left–right symmetry in our initial submission, and we agree that our terminology was confusing.
In developmental biology, the term “left–right symmetry breaking” is often used to describe asymmetric flows generated by nodal cilia, which subsequently establish developmental asymmetry. This usage differs fundamentally from the physical definition of mirror symmetry breaking, which refers to chirality switching upon mirror reflection. As the reviewer correctly noted, our system does not exhibit mirror symmetry breaking in this strict physical sense.
To avoid confusion, we have revised the manuscript and replaced the term left–right symmetry breaking with left–right asymmetry between the edge and the center of the biofilm. This asymmetry arises from frequency gradients across the biofilm and is not a trivial boundary effect. For circular colonies, this phenomenon is more accurately described as radial asymmetry. We have rewritten the relevant sections of the manuscript to clarify this distinction and prevent misinterpretation.
Reviewer #2 (Public review):
Summary:
This manuscript by Altin et al. examines the dynamics of bacterial assemblies, building on previously published work documenting mechanical spiral waves. The authors show that the emergent dynamics can be influenced by various factors, including the strain of bacteria and water content in the sample. While the topic of this paper would be of broad interest, and the preliminary results are certainly interesting, various aspects of this paper are underdeveloped and require further exploration.
Strengths:
One of the nice features of this system is the ability to transition between the different states based on the addition or withdrawal of water. The authors use a similar experimental model system and mathematical model to previously published work (Reference 49), but extend by showing that the behaviour can be modified through simple interventions. Specifically, the authors show that adding water droplets or drying the sample through heating can result in changes in the observed wave structure. This represents a possible way of controlling active matter.
The mathematical model proposed in this paper involves a phase-oscillator model of Kuramotostyle coupling (similar to previously reported models). A non-reciprocal phase lag is introduced in order to facilitate the patterns seen in experiments. The qualitative agreement in the behaviour is quite striking, showing both spiral waves and travelling waves.
We thank the reviewer for the positive assessment and for pointing out areas that required further development. The reviewer is correct that our work builds on previously reported bacterial spiral wave systems; however, there are several significant differences that we now emphasize more clearly in the revised manuscript.
First, our study involves a different bacterial species and reveals a distinct dynamical process: the waves we report are strictly localized on the surface of the biofilm, in contrast to the bulk oscillations detected through density fluctuations in the earlier work (Ref. 49). The surface waves in our system resemble “Mexican wave”-like motions, in which surface bacteria periodically lift upward. To highlight this key distinction, we performed new imaging experiments that directly visualize this process. (New Video 5 and 6, Author response image 1).
Second, we systematically compared different bacterial strains, including pathogenic species such as P. aeruginosa PA14 and PAO1, alongside our BSL-1 strain. This comparative approach demonstrates that the observed phenomenon spans strains with different pathogenicity levels, and genetic variations while also showing that our strain provides a safer and more broadly usable model system for laboratory investigations.
Third, the modeling frameworks differ. Whereas the referred study relied primarily on phase models similar to those used in cilia systems, we combine a delayed Kuramoto-style oscillator model with an active solid model. This combination provides both a phenomenological description and a physical interpretation of the collective dynamics. We acknowledge that, in the original submission, the physical interpretation of the model in relation to our experimental system was underdeveloped. In the revision, we have now established this link explicitly through the elasticity and elasto active coupling of the biofilm. Specifically, we show that the transition from motile to biofilm states is accompanied by changes in elasticity, which directly influence the observed transitions between different types of wave defects. This connection is consistent with prior theoretical works and has even been only studied in robotic active matter systems.
Together, these clarifications and new results reinforce the novelty of our findings and establish a stronger connection between the experiments and the modeling framework.
Author response image 1.
Comparison between the elastic biofilm core and the motile colony edge. Highresolution video recordings revealing individual bacterial motion highlight the key physical differences driving wave-generating. Time-lapse snapshots show that bacteria at the colony edge move freely and form fingering structures, whereas bacteria in the elastic central biofilm periodically lift vertically, producing a Mexican-wave–like collective motion across the surface. See new Video
Weaknesses:
The principal observation of the paper - that spiral waves emerge in these systems and can be controlled in various ways - is not linked to microscale dynamics at the cell level. It is recognised that hydrodynamics can introduce non-reciprocity, an essential ingredient of this model. However, in this work the authors have not identified a physical mechanism for the lag, e.g., either through steric interactions or hydrodynamic disturbances. This is also relevant in the phase oscillator modelling section. In low Reynolds number flows, dynamics are instantaneously determined. In this light, what does the phase lag term represent?
The reviewer is correct that, at low Reynolds numbers, fluid dynamics are instantaneous and do not generate real temporal delays. However, nonreciprocity in hydrodynamic interactions can still emerge from the tensorial structure of the Blake–Oseen Green’s function. In this formalism, the effective asymmetry can be represented mathematically as a phase-lag–like term. This has been theoretically demonstrated in Ref.40. While this is not a literal time delay, it functions analogously by breaking odd symmetry in the coupling.
In our system, strong long-range hydrodynamic interactions are absent, as the bacteria are embedded in an elastic biofilm matrix. Instead, the dominant interactions are active elastic couplings mediated by pili and biofilm structure. The elastic solid model behaves in a way that is conceptually similar to the hydrodynamic case: pili-induced deformations of the elastic medium produce anisotropic stresses that play a role analogous to the tensorial hydrodynamic Green’s function. Thus, the phase-lag term in our Kuramoto-based model can be interpreted as an effective representation of these nonreciprocal elastic interactions.
We have clarified this point in the revised manuscript by explicitly connecting the phenomenological phase-lag term to the underlying elastic coupling in biofilms.
What is the origin of the coupling term, b? Can this be varied systematically or derived from experimental measurements or parameters?
The term b represents the enhanced elasto-active coupling of the pili process. The length of the Pili varies, and the elongated Pili has more potential to modulate the coupling between bacteria which is known to depend on a critical threshold. This process resembles the pinning dynamics and is driven by the activity of molecular motors within the pili machinery. However, the detailed mechanisms that set the effective coupling strength remain highly complex and are not yet fully understood.
At present, we do not have a direct way to systematically manipulate b in experiments. A major technical limitation is the nanoscale nature of type IV pili: these protein assemblies are extremely small and difficult to monitor or manipulate directly. Even basic tools such as GFP-based labeling have proven challenging to implement, which restricts our ability to track the detailed dynamics of these structures in live biofilms.
While we cannot currently derive b directly from experimental parameters, we emphasize in the revised manuscript that b should be understood as an effective parameter capturing the excitability of pili retractions. We also highlight this limitation and note that future advances in molecular imaging and manipulation of pili will be essential for quantitatively linking b to microscopic processes.
Classification of wave properties is an important aspect of this paper, but is not accomplished in a quantitative sense. What is the method for distinguishing between travelling and spiral waves? There is a range of quantitative tools that could be used to investigate these dynamics (and also compare quantitatively with the models). For example, examining the correlation functions and order parameters could assist with the extraction of wave features (see extensive literature on oscillator models).
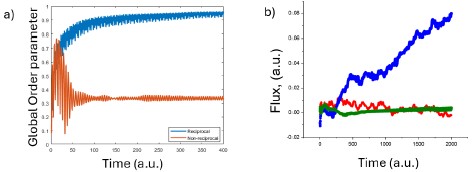
We thank the reviewer for emphasizing this important point. In the revised manuscript, we have incorporated the classic Kuramoto order parameter (S) to characterize the dynamics in our model simulations. However, this metric is not directly applicable to our experimental system, because we cannot resolve the phase of individual bacteria at large scales.
Instead, we have focused on a flux-based parameter, as previously used in Ref. 40, which can be measured experimentally from collective surface dynamics. Interestingly, we find that the directional flux extracted from our experimental movies closely matches the trends predicted by the model order parameter. We suspect that this similarity arises from the combination of our optical illumination method and the characteristic surface modulations of the biofilm. While we currently lack a rigorous theoretical justification for this correspondence, so we want to keep this discussion in the review document.
In summary, we now use the classic Kuramoto order parameter in simulations and rely on the experimentally accessible flux measure for our experimental data. This dual approach allows us to compare model and experiment in a consistent manner.
Author response image 2.
Critical order parameters of the coupled biofilm system. (a) The Kuramoto global order parameter increases continuously as the system becomes globally synchronized. In contrast, in the nonreciprocally coupled system the order parameter saturates at a critical level. (b) In the experimentally observed biofilm, however the flux generated by the coupled oscillations provides a more appropriate measure of synchronization. Blue curves indicate directionally propagating planar waves, red curves correspond to spiral wave formation, and green curves represent the globally synchronized reciprocal system.
Author response image 3.
Comparison of flux profiles of the simulations with experimental measurements. Directional optical illumination enhances the flux term on the surface of the biofilm.
The methodology of changing the dynamics through moisture content appears to be slightly underdeveloped, e.g., adding water involves a droplet, and removing water is accomplished by heating (which presumably could cause other effects). Could the dynamics not be controlled more directly by varying the humidity?
We thank the reviewer for this valuable suggestion. Our results indicate that water content in the biofilm plays a key role in driving the transition to the biofilm state by modulating its elasticity. During the initial submission, we did not know how to systematically vary humidity without simultaneously altering temperature. Standard approaches typically involve water evaporation in controlled chambers, which inherently changes both parameters.
Following the reviewer’s recommendation, we first measured the ambient moisture levels inside closed culture plates. To our surprise, the relative humidity was already ~98%, leaving virtually no room to increase it further. We then attempted to decrease humidity by flowing dry synthetic air, but even under these conditions we could not reduce it below ~85%, and achieving this required unrealistically high flow rates. Moreover, we noticed that in closed-lid NGM plates, evaporation is already substantial, and when the lid is left open the evaporation rate reaches ~1 µm/s. This rapid surface thinning severely limits the quality of long-term time-lapse imaging.
Taken together, these technical constraints explain why we have to reliy on localized perturbations such as water droplets and heating rather than global humidity control. We have clarified this point in the revised manuscript and now explicitly discuss both the challenges and limitations of humidity-based approaches.
At the same time, the authors also mention that temperature itself plays a role in shaping the behaviour. What is the mechanism for this? Is it just through evaporation? Since the frequency increases with temperature, could it just be that activity increases with temperature?
We thank the reviewer for raising this critical point. We believe that temperature has two distinct impacts operating on different timescales.
Short timescale (~minutes): We observed that biofilm oscillations respond to temperature changes very rapidly and in a reversible manner. This timescale is too short to be explained by modulation of water content or bulk elasticity of the biofilm. Instead, we attribute the immediate frequency increase to enhanced biological activity of the bacteria at elevated temperatures.
Long timescale (~tens of minutes to hours): During processes such as the transition from planar to spiral waves, prolonged heating can significantly alter the biofilm structure. These changes are not reversible and likely involve modifications of elasticity and other structural properties.
In the modeling framework, the short-timescale effect is represented as an increase in the active force term, while the long-timescale effect is captured by concurrent changes in both the active force and the elastic properties of the biofilm. We have clarified this mechanism and its representation in the revised manuscript.
Reviewer #3 (Public review):
Summary:
This manuscript presents a novel investigation into unidirectionally propagating waves observed on the surface of Pseudomonas nitroreducens bacterial biofilms. The authors explore how these waves, initially spiral in form, transition into combinations of spiral, target, and planar patterns. The study identifies the periodic extension-retraction cycles of type IV pili as the driving mechanism for wave propagation, which preferentially moves from the colony's edge to its center. Furthermore, the manuscript proposes two theoretical models-a phase-oscillator model and a continuum active solid model-to reproduce these phenomena, and demonstrates how external manipulations (e.g., water droplets, temperature, PEG) can control wave patterns and direction, often correlating with oscillation frequency gradients. The work aims to bridge the fields of activematter physics and bacterial biophysics by providing both experimental observations and theoretical frameworks for understanding these complex biological wave phenomena.
We thank the reviewer for the positive assessment of our work and for highlighting both the novelty and the key contributions of our study.
Strengths:
The experimental discovery of unidirectionally propagating waves on bacterial biofilms is highly intriguing and represents a significant contribution to both microbiology and active-matter physics.
The detailed observations of wave pattern transitions (spiral to target to planar) and their response to various environmental perturbations (water, temperature, PEG) provide valuable empirical data. The identification of type IV pili as the driving force offers a concrete biological mechanism. The observed correlation between frequency gradients and wave direction is a compelling finding with potential for broader implications in understanding biological pattern formation. This work has the potential to stimulate further research in the collective behavior of living systems and the physical principles underlying biological organization.
We thank the reviewer once again for emphasizing the importance of wave directionality. We also believe that this phenomenon may provide insight into early symmetry-breaking processes observed in developmental biology, where oxygen or nutrient gradients in dense environments could play a similar role.
Weaknesses:
The manuscript attempts to link unidirectional wave propagation to non-reciprocal couplings but ultimately shows that the wave direction is determined by the gradient of the oscillation frequency. The couplings in the two theoretical models are both isotropic and thus cannot dictate the wave direction. A clear distinction should be made between non-reciprocity as a source of wave generation and non-uniformity as a controlling factor of wave direction.
We greatly appreciate the reviewer’s careful evaluation, particularly for highlighting this important and often confusing distinction. The relationship between nonreciprocity, spontaneous symmetry breaking, and frequency gradients has also been a challenging concept for us and required significant effort to clarify.
Recent theoretical studies have established that traveling wave formation requires nonreciprocity, which provides a framework for understanding phenomena ranging from spiral to target and planar waves. In our system, nonreciprocity arises between the displacement field (U) and the pili force vector (P): as a result in broken phase U effectively “chases” P, breaking PT symmetry locally and thereby enabling the generation of local directional flux and traveling waves. In this sense, nonreciprocity is essential for travelling wave generation and spontaneous symmetry breaking in either direction.
However, we now agree that global directionality (always from right to left, or edge to center) is set by an independent factor—namely, the oscillation frequency gradient across the biofilm. Thus, while nonreciprocity determines whether waves can travel, frequency gradients determine the large-scale direction in which they propagate. Put differently, PT symmetry is already broken spiral waves due to nonreciprocity, but global asymmetry (frequency gradients) is required to align the overall propagation in one direction.
We have clarified this distinction in the revised manuscript, emphasizing that nonreciprocity is a necessary ingredient for travelling wave generation, whereas global asymmetry controls global wave direction.
Modification in the manuscript:
“We should note that traveling waves indicate broken PT symmetry between these fields triggered by nonreciprocity, with spiral waves serving as a classic signature of this phenomenon. A further transition from spiral to planar waves reflects an overall asymmetry in the frequency profile, which is not directly related to PT-symmetry breaking.”
The relationship between the phase oscillator model and the active solid model is unclear. Given that U and P are both dynamical variables evolving in three-dimensional space, defining the phase Φ precisely in the phase space spanned by U and P could be challenging. A graphical illustration of the definition of Φ would be beneficial. To ensure reproducibility of the numerical results, the parameter values used in the numerical simulations and an explicit definition of the elastic force in the active solid model should be provided.
We agree with the reviewer that the relationship between the phase oscillator model and the active solid model can be confusing, but establishing this link is essential to connect different modeling approaches in the literature. As the reviewer notes, in a fully three-dimensional setting with freely moving bacteria, defining the oscillation phase (Φ) in the phase space spanned by U and P is indeed complicated.
However, our recent imaging results show that bacteria within the biofilm do not undergo large translational motions but instead exhibit periodic “Mexican wave”-like oscillations. These oscillations are confined to a restricted phase space, which allows us to define Φ in a straightforward way. In this context, the phase oscillator model becomes a natural reduction of the dynamics.
Similarly, in the active solid (or active gel) model, we can plot not only the displacement and force vectors but also the local phase, which shows strong agreement with the phenomenological Kuramoto-style model. To make this connection clearer, we have now included a schematic illustration in the revised manuscript that explicitly shows how Φ is defined in the reduced phase space, and we provide the parameter values used in the simulations as well as the explicit definition of the elastic force in the active solid model to ensure reproducibility.
The link between the theoretical models and experimental results is weak. For example, the propagation of the kink from the lower to the higher part of the surface (Figure 1e) could be addressed within the framework of the active solid model. The mechanism of transition from spiral to target waves (Figure 3a), b)) requires clarification, identifying which model parameter is crucial for inducing this transition. The wave propagation toward the lower frequency side is numerically demonstrated using the phase oscillator model, but a physical or intuitive explanation for this phenomenon is missing. Also, the wave transitions induced by the addition of water droplets and temperature rise are not linked to specific parameters in the theoretical models.
We thank the reviewer for highlighting this important weakness, which was also consistently noted by the other reviewers. We fully agree that the link between our theoretical models and experimental results required significant strengthening.
With improved imaging in the revised study, we were able to uncover additional connections that help establish this link more clearly. We acknowledge that our ability to measure detailed biofilm parameters is limited, which restricts us from providing fully quantitative mappings. Nonetheless, based on the reviewers’ suggestions, we carried out additional imaging and simulations to compare bacterial dynamics at the colony edge and within the biofilm surface. These data confirm that cells within the biofilm undergo restricted, “Mexican wave”-like oscillations, emphasizing the critical role of elasticity in governing the collective dynamics.
Experimentally, we found that adding water or PEG, or alternatively inducing drying, strongly modulates the effective elasticity of the biofilm. Within the active solid framework, elasticity and the elasto-active coupling are the key parameters controlling the system. By tuning these parameters in simulations, we could reproduce the qualitative transitions observed experimentally. Specifically, we observed that:
At low elasticity, topological defects are mobile and can move, merge, or annihilate, leading to the emergence of planar waves.
At high elasticity, defects remain pinned, across the biofilm surface, dominating the dynamics.
These observations suggest that the motility of defects is the crucial parameter governing the transition between spiral, target, and planar waves. Although we cannot independently manipulate each parameter in experiments, varying the moisture content provides an effective and experimentally accessible control.
Finally, our simulations and new analyses reveal that spiral defect cores can move and merge to form target waves or annihilate entirely—processes that we also observe experimentally. This rich dynamical behavior underscores the importance of elasticity in shaping pattern transitions, and we believe it warrants further theoretical exploration. We have clarified this connection and its implications in the revised manuscript.
First, we compare defect dynamics in both Kuramoto-based simulations and the active solid model. Both systems exhibit similar defect-survival behavior. As shown in the review , pairs of unlike (+/−) defects can stably persist only at high nonreciprocity. We further quantify this behavior by plotting the separation distances between unlike defect pairs and find that short-range defect separations are possible exclusively in the high-nonreciprocity regime Supplementary Figure 11.
This high-nonreciprocity regime corresponds to the dry biofilm state. Increasing moisture reduces elasticity, leading to the loss of stable defect dynamics and promoting the annihilation of unlike defect pairs, which in turn drives the system toward target-wave formation and ultimately planar waves. Conversely, heating the biofilm removes water, enhances elasticity, and increases the system’s ability to sustain closely separated defect pairs.
Experimentally, we further observe that removing water by heating enhances surface nonuniformities, which readily trigger defect-pair formation. To investigate this mechanism, we performed additional simulations in which local nonuniformities were introduced Supplementary Figure 12. Consistent with experiments, defect-pair generation occurs only at high nonreciprocity, where pairs of unlike defects can be stably maintained. Experimental observation (Author response image 4) also show that surface nonuniformities on the biofilm surface similarly trigger the formation of closely separated defect pairs. We have updated the details of the defect dynamics in the revised manuscript to clarify the transition between these waves.
Author response image 4.
Experimental observation showing that small surface nonuniformities on the biofilm surface trigger the formation of closely separated defect pairs. Arrows indicate the position of the nonuniformities
Modification in the manuscript:
Defect dynamics controlling the transition between spiral to target waves
“To better understand the dynamics of the transition between different form of the waves we focused on numerical simulations. We noticed that the motility of defects is the crucial parameter governing the transition between spiral, target, and planar waves varying the moisture content provides an effective and experimentally accessible control this motility. Our analyses revealed that spiral defect cores can move and merge to form target waves or annihilate entirely—processes that we also observe experimentally. This rich dynamical behavior underscores the importance of elasticity in shaping pattern transitions. First, we compare defect dynamics in both Kuramotobased simulations and the active solid model. Both systems exhibit similar defect-survival behavior. As shown in Supplementary Figure10, pairs of unlike (+/−) defects can stably persist only at high nonreciprocity. We further quantify this behavior by plotting the separation distances between unlike defect pairs and find that short-range defect separations are possible exclusively in the high-nonreciprocity regime (Supplementary Figure11). This high-nonreciprocity regime corresponds to the dry biofilm state. Increasing moisture reduces elasticity, leading to the loss of stable defect dynamics and promoting the annihilation of unlike defect pairs, which in turn drives the system toward target-wave formation and ultimately planar waves. Conversely, heating the biofilm removes water, enhances elasticity, and increases the system’s ability to sustain closely separated defect pairs. Experimentally, we further observe that removing water by heating enhances surface nonuniformities, which readily trigger defect-pair formation (Supplementary Video9). To investigate this mechanism, we performed additional simulations in which local nonuniformities were introduced (Supplementary Video12-13). Consistent with experiments, defect-pair generation occurs only at high nonreciprocity, where pairs of unlike defects can be stably maintained. Experimental observation (Supplementary Video9) also show that surface nonuniformities on the biofilm surface similarly trigger the formation of closely separated defect pairs.”
All the recommended points have been addressed in the revised manuscript.
-

eLife Assessment
This important study combines a two-person joint hand-reaching paradigm with game-theoretical modeling to examine whether, and how, reflexive visuomotor responses are modulated by a partner's control policy and cost structure. The study provides a convincing set of behavioral findings suggesting that involuntary visuomotor feedback is indeed modulated in the context of interpersonal coordination. The work will be of interest to cognitive scientists studying the motor and social aspects of action control.
-

Reviewer #1 (Public review):
Summary:
Sullivan and colleagues examined the modulation of reflexive visuomotor responses during collaboration between pairs of participants performing a joint reaching movement to a target. In their experiments, the players jointly controlled a cursor that they had to move towards narrow or wide targets. In each experimental block, each participant had a different type of target they had to move the joint cursor to. During the experiment, the authors used lateral perturbation of the cursor to test participants' fast feedback responses to the different target types. The authors suggest participants integrate the target type and related cost of their partner into their own movements, which suggests that visuomotor gains are affected by the partner's task.
Strengths:
The topic of the manuscript is very …
Reviewer #1 (Public review):
Summary:
Sullivan and colleagues examined the modulation of reflexive visuomotor responses during collaboration between pairs of participants performing a joint reaching movement to a target. In their experiments, the players jointly controlled a cursor that they had to move towards narrow or wide targets. In each experimental block, each participant had a different type of target they had to move the joint cursor to. During the experiment, the authors used lateral perturbation of the cursor to test participants' fast feedback responses to the different target types. The authors suggest participants integrate the target type and related cost of their partner into their own movements, which suggests that visuomotor gains are affected by the partner's task.
Strengths:
The topic of the manuscript is very interesting, and the authors are using well-established methodology to test their hypothesis. They combine experimental studies with optimal control models to further support their work. Overall, the manuscript is very timely and shows important findings - that the feedback responses reflect both our and our partners tasks.
-

Reviewer #2 (Public review):
Summary:
Sullivan and colleagues studied the fast, involuntary, sensorimotor feedback control in interpersonal coordination. Using a cleverly designed joint-reaching experiment that separately manipulated the accuracy demands for a pair of participants, they demonstrated that the rapid visuomotor feedback response of a human participant to a sudden visual perturbation is modulated by his/her partner's control policy and cost. The behavioral results are well matched with the predictions of the optimal feedback control framework implemented with the dynamic game theory model. Overall, the study provides an important and novel set of results on the fast, involuntary feedback response in human motor control in the context of interpersonal coordination.
Review:
Sullivan and colleagues investigated whether fast, …
Reviewer #2 (Public review):
Summary:
Sullivan and colleagues studied the fast, involuntary, sensorimotor feedback control in interpersonal coordination. Using a cleverly designed joint-reaching experiment that separately manipulated the accuracy demands for a pair of participants, they demonstrated that the rapid visuomotor feedback response of a human participant to a sudden visual perturbation is modulated by his/her partner's control policy and cost. The behavioral results are well matched with the predictions of the optimal feedback control framework implemented with the dynamic game theory model. Overall, the study provides an important and novel set of results on the fast, involuntary feedback response in human motor control in the context of interpersonal coordination.
Review:
Sullivan and colleagues investigated whether fast, involuntary sensorimotor feedback control is modulated by the partner's state (e.g., cost and control policy) during interpersonal coordination. They asked a pair of participants to make a reaching movement to control a cursor and hit a target, where the cursor's position was a combination of each participant's hand position. To examine fast visuomotor feedback response, the authors applied a sudden shift in either the cursor (experiment 1) or the target (experiment 2) position in the middle of movement. To test the involvement of partner's information in the feedback response, they independently manipulated the accuracy demand for each participant by varying the lateral length of the target (i.e., a wider/narrower target has a lower/higher demand for correction when movement is perturbed). Because participants could also see their partner's target, they could theoretically take this information (e.g., whether their partner would correct, whether their correction would help their partner, etc.) into account when responding to the sudden visual shift. Computationally, the task structure can be handled using dynamic game theory, and the partner's feedback control policy and cost function are integrated into the optimal feedback control framework. As predicted by the model, the authors demonstrated that the rapid visuomotor feedback response to a sudden visual perturbation is modulated by the partner's control policy and cost. When their partner's target was narrow, they made rapid feedback corrections even when their own target was wide (no need for correction), suggesting integration of their partner's cost function. Similarly, they made corrections to a lesser degree when both targets were narrower than when the partner's target was wider, suggesting that the feedback correction takes the partner's correction (i.e., feedback control policy) into account.
The strength of the current paper lies in the combination of clever behavioral experiments that independently manipulate each participant's accuracy demand and a sophisticated computational approach that integrates optimal feedback control and dynamic game theory. Both the experimental design and data analysis sound good and the main claim is well supported by the results.
A future direction would be to investigate how this mechanism is implemented in the CNS and to examine whether the same cooperative mechanism also applies to human-AI interactions.
-

Author response:
The following is the authors’ response to the original reviews.
Reviewer #1 (Public review):
Summary
Sullivan and colleagues examined the modulation of reflexive visuomotor responses during collaboration between pairs of participants performing a joint reaching movement to a target. In their experiments, the players jointly controlled a cursor that they had to move towards narrow or wide targets. In each experimental block, each participant had a different type of target they had to move the joint cursor to. During the experiment, the authors used lateral perturbation of the cursor to test participants’ fast feedback responses to the different target types. The authors suggest participants integrate the target type and related cost of their partner into their own movements, which suggests that visuomotor gains are …
Author response:
The following is the authors’ response to the original reviews.
Reviewer #1 (Public review):
Summary
Sullivan and colleagues examined the modulation of reflexive visuomotor responses during collaboration between pairs of participants performing a joint reaching movement to a target. In their experiments, the players jointly controlled a cursor that they had to move towards narrow or wide targets. In each experimental block, each participant had a different type of target they had to move the joint cursor to. During the experiment, the authors used lateral perturbation of the cursor to test participants’ fast feedback responses to the different target types. The authors suggest participants integrate the target type and related cost of their partner into their own movements, which suggests that visuomotor gains are affected by the partner’s task.
Strengths
The topic of the manuscript is very interesting, and the authors are using well established methodology to test their hypothesis. They combine experimental studies with optimal control models to further support their work. Overall, the manuscript is very timely and shows important findings - that the feedback responses reflect both our and our partner’s tasks.
We thank the reviewer for the positive comments regarding our work.
Weaknesses
However, in the current version of the manuscript, I believe the results could also be interpreted differently, which suggest that the authors should provide further support for their hypothesis and conclusions.
Major Comments
(1) Results of the relevant conditions:
In addition to the authors’ explanation regarding the results, it is also possible that the results represent a simple modulation of the reflexive response to a scaled version of cursor movement. That is, when the cursor is partially controlled by a partner, which also contributes to reducing movement error, it can also be interpreted by the sensorimotor system as a scaling of hand-to-cursor movement. In this case, the reflexes are modulated according to a scaling factor (how much do I need to move to bring the cursor to the target). I believe that a single-agent simulation of an OFC model with a scaling factor in the lateral direction can generate the same predictions as those presented by the authors in this study. In other words, maybe the controller has learned about the nature of the perturbation in each specific context, that in some conditions I need to control strongly, whereas in others I do not (without having any model of the partner). I suggest that the authors demonstrate how they can distinguish their interpretation of the results from other explanations.
We thank the reviewer for the thoughtful comment. While it is possible that the change in the visuomotor feedback responses could be just from a scaling factor. This hypothesis could explain the difference between two conditions, but would fail to explain differences between two other conditions. Specifically, this hypothesis could explain a decrease in involuntary visuomotor feedback responses between partner-irrelevant/self-relevant and partner-relevant/self-relevant. Critically, this hypothesis could not explain the difference between partner-irrelevant/self-irrelevant and partner-relevant/self-irrelevant. That is, there is no reason to scale a response to correct for a partner’s relevant target when your own target is irrelevant. However, our finding that there is a greater involuntary visuomotor feedback response in partner-relevant/self-irrelevant compared to partner-irrelevant/self-irrelevant is predicted by the notion that humans form a representation of others and consider their movement costs.
We have added a paragraph in the discussion to justify our hypothesis over the scaling factor hypothesis.
“Our hypothesis that the sensorimotor system uses a representation of a partner and considers the partner’s costs to modify involuntary visuomotor feedback responses can parsimoniously explain all of our experimental findings. There are a few alternative hypotheses that could explain a subset of results. One alternative hypothesis is that participants simply learned the hand to center cursor mapping in each experimental condition. That is, instead of using a model of their partner, participants simply adapted to the dynamics of the center cursor. However, this hypothesis would not predict an increased involuntary visuomotor feedback response in the partner-relevant/self-irrelevant condition compared to the partner-irrelevant/self-irrelevant condition. If participants did not form a model of their partner nor consider their partner’s costs, then they would not display an increased feedback response when they had an irrelevant target and their partner’s target was relevant. An increased feedback response to help a partner achieve their goal is captured by our hypothesis that the sensorimotor system uses a representation of a partner and considers the partner’s costs to modify involuntary visuomotor feedback responses.”
(2) The effect of the partner target:
The authors presented both self and partner targets together. While the effect of each target type, presented separately, is known, it is unclear how presenting both simultaneously affects individual response. That is, does a small target with a background of the wide target affect the reflexive response in the case of a single participant moving? The results of Experiment 2, comparing the case of partner- and self-relevant targets versus partner-irrelevant and self-relevant targets, may suggest that the system acted based on the relevant target, regardless of the presence and instructions regarding the self-target.
We thank the reviewer for bringing up another valid point, which we discussed at length as a group when designing the experiment. The reviewer is correct in pointing out the lack of difference in the involuntary epoch between the partner-relevant/self-relevant and partner-irrelevant/self-relevant could potentially suggest that the sensorimotor system acted based on only relevant targets, irrespective if it was a self or partner relevant target. While the effect of the simultaneous presentation of a narrow and wide target on an individual’s response by themselves is unknown, comparing the differences between our other experimental conditions control for this potential confound. Participants viewed a wide target and a narrow target on the screen, in both the partner-irrelevant/self-relevant condition and the partner-relevant/self-irrelevant condition. Crucially, we found that the visuomotor feedback responses were greater in the partner-irrelevant/self-relevant condition compared to the partner-relevant/self-irrelevant condition in both Experiment 1 and 2. That is, participants were able to distinguish between the self-target and partner target and appropriately modify their feedback responses in both Experiment 1 and 2, despite there being both a wide and narrow target on the screen in both conditions. Given that we found different visuomotor feedback responses between the two conditions that had both a narrow and wide target, this rules out the alternative hypothesis that the sensorimotor system acted based just on a relevant target being present. We have added to our discussion to clarify this point.
“Another alternative hypothesis would be that the sensorimotor system was responding only to the relevant target displayed on the screen. Again, this hypothesis would only explain a subset of our results. In particular, this relevant target hypothesis cannot explain the observed feedback response differences between the partner-relevant/self-irrelevant and partner-irrelevant/self-relevant conditions in both Experiments 1 and 2.”
(3) Experiment instructions:
It is unclear what the general instructions were for the participants and whether the instructions provided set the proposed weighted cost, which could be altered with different instructions.
Our instructions explicitly informed participants that their performance bonus was only based on them stabilizing within their own self-target within the time constraint. We have added the following in the methods to emphasize this instruction.
“In other words, we ensured participants had a clear understanding that their performance in the task was only based on stabilizing the center cursor in their own self-target within the time constraint. Therefore, the instructions and timing constraints did not enforce participants to work together.”
(4) Some work has shown that the gain of visuomotor feedback responses reflects the time to target and that this is updated online after a perturbation (Cesonis & Franklin, 2020, eNeuro; Cesonis and Franklin, 2021, NBDT; also related to Crevecoeur et al., 2013, J Neurophysiol). These models would predict different feedback gains depending on the distance remaining to the target for the participant and the time to correct for the jump, which is directly affected by the small or large targets. Could this time be used to target instead of explaining the results? I don’t believe that this is the case, but the authors should try to rule out other interpretations. This is maybe a minor point, but perhaps more important is the location (&time remaining) for each participant at the time of the jump. It appears from the figures that this might be affected by the condition (given the change in movement lengths - see Figure 3 B & C). If this is the case, then could some of the feedback gain be related to these parameters and not the model of the partner, as suggested? Some evidence to rule this out would be a good addition to the paper - perhaps the distance of each partner at the time of the perturbation, for example. In addition, please analyze the synchrony of the two partners’ movements.
(1) Time to target and forward position
The reviewer raises an interesting point. In our task, the cursor/target jump occurs once the center cursor crosses 6.25 cm from the start. We analyzed the time it took for the center cursor to intercept the targets from perturbation onset (Supplementary D). In Experiment 1, an ANOVA with center cursor time-to-target as the dependent variable showed no main effect of self-target (F[1,47] = 2.45, p = 0.124) or partner target (F[1,47] = 2.50, p=0.120), nor any interaction (F[1,47] = 1.97, p = 0.166). In Experiment 2, an ANOVA with center cursor time-to-target as the dependent variable showed a significant interaction (F[1,47] = 5.87, p = 0.019). Post-hoc mean comparisons showed that only the difference between the partner-irrelevant/self-irrelevant and partner-relevant/self-irrelevant condition was significant (p = 0.006). Given that only one comparison in Experiment 2 showed a difference in time-to-target, we do not believe that time-to-target was a significant driver of the change in involuntary visuomotor feedback responses observed between conditions. While time-to-target is likely a metric the nervous system modifies feedback gains around, our results suggest that the nervous system can also use a partner model to modify feedback gains. We have added a supplemental analysis on time to target
“Previous work by Česonis and Franklin (2020) showed that time to-target is a key variable the sensorimotor system uses to modify feedback responses. In their experiment, they manipulated the time-to-target of the participant’s cursor, while controlling for other movement parameters (e.g., distance from goal) [1]. When compared to classical optimal feedback control models, they showed that a model that modifies feedback responses based on time-to-target best predicted their results. In our task, it’s possible that the time-to-target could have influenced visuomotor feedback responses, since the distance to the center of the target is greater for a narrow target than a wide target on perturbation trials.”
“We calculated the time from perturbation onset to the center cursor reaching the forward position of the targets (Supplementary Fig. S5). In Experiment1, an ANOVA with center cursor time-to-target as the dependent variable showed no main effect of self-target (F[1,47]=2.45,p=0.124) or partner target (F[1,47] = 2.50, p=0.120), nor any interaction (F[1,47] = 1.97, p = 0.166). In Experiment2, an ANOVA with center cursor time-to-target as the dependent variable showed a significant interaction (F [1,47] = 5.87, p = 0.019). Post-hoc mean comparisons showed that only the difference between the partner-irrelevant/self-irrelevant and partner-relevant/self-irrelevant condition was significant (p=0.006). Although time-to-target and hand position are important variables for the control ofmovement,[1,2,3] they are likely not driving factors of the different in voluntary visuomotor feedback responses between our experimental conditions.”
However, it is possible that the participant forward position at perturbation onset could also influence the involuntary feedback response. We show the forward positions at perturbation onset in Supplementary D. Statistical analysis of the forward positions in Experiment 1 showed a main effect of self-target (F[1,47] = 12.72, p < 0.001), main effect of partner target (F[1,47] = 12.82, p < 0.001), and no interaction (F[1,47] = 0.00, P = 0.991). We see the same trend in experiment 2, showing a main effect of self-target (F[1,47] = 12.11, p < 0.001), main effect of partner target (F[1,47] = 12.04, p < 0.001), and no interaction (F[1,47] = 0.00, p = 0.986). The fact that there was no interaction implies that the results could not solely be due to forward position. Nevertheless, given there were main effects, we proceeded to run an ANCOVA on the involuntary visuomotor feedback responses with forward position as a covariate. For experiment 1, we still observed a significant interaction between self and partner target (F[1,47] = 43.14, p < 0.001). Further, we also observed no significant main effect of forward position on the involuntary visuomotor feedback responses. The ANCOVA for Experiment 2 also showed that there was still a significant interaction of self and partner target on the involuntary visuomotor feedback responses (F[1,47] = 9.80, p = 0.002). However, here we did find a significant main effect of the forward position (F[1,47] = 5.06, p = 0.026). Therefore, we ran follow-up mean comparisons with the covariate adjusted means. We found the same statistical trend as reported in the main results. We found significant differences between the partner-irrelevant/self-irrelevant and partner-relevant/self-irrelevant conditions (p = 0.003), partner-relevant/self-irrelevant and partner-irrelevant/self-relevant conditions (p < 0.001), partner-relevant/self-irrelevant and partner-relevant/self-relevant conditions (p < 0.001). We found no significant difference between the partner-irrelevant/self-relevant and partner-relevant/self-relevant conditions (p = 0.381). Given that there was no main effect of forward position in Experiment 1, and that our adjusted mean comparisons in Experiment 2 showed the same trends as the unadjusted mean comparisons in the main manuscript, our results show that the forward position of the participants is not a significant factor in explaining the differences in involuntary visuomotor feedback responses between conditions.
“Supplementary Fig. 6 shows the participant hand forward position at perturbation onset time for Experiment 1 (A) and Experiment 2 (B). It is possible that the participant forward hand position at perturbation onset time could influence their visuomotor feedback responses. Therefore, we ran an ANCOVA with self-target and partner target as factors, and participant forward hand position at perturbation onset time as a covariate. In Experiment 1, we found no main affect of participant forward hand position on involuntary visuomotor feedback responses (F[1,47] = 1.466, p = 0.228). Further, when including the covariate, we still found a significant interaction between self-target and partner target on in voluntary visuomotor feedback responses (F[1,47]=43.2, p<0.001).”
“In Experiment 2, we found a significant main effect of participant forward hand position on involuntary visuomotor feedback responses (F[1,47] = 6.73, p = 0.010). We still found a significant interaction between self-target and partner target (F[1,47] = 9.78, p = 0.002). Since we found a main effect of participant forward hand position, we calculated the adjusted means of the involuntary visuomotor feedback responses. We then performed follow-up mean comparisons on the adjusted means of the involuntary visuomotor feedback responses (using emmeans in R). We found the same significant trends as the unadjusted means in the main manuscript. Specifically we found involuntary visuomotor feedback responses to be: significantly greater in the partner-relevant/self-irrelevant condition compared to the partner-irrelevant/self-irrelevant condition (p =0.003),significantly greater in the partner-relevant/self-irrelevant condition compared to the partner-irrelevant/self-relevant condition (p<0.001), significantly greater in the partner-relevant/self-relevant condition compared to the partner-relevant/self-irrelevant condition (p<0.001),and not different between the partner-irrelevant/self-relevant and partner-relevant/self-relevant conditions (p = 0.824).”
We have also included in the discussion how time-to-target and participant forward hand position are important control variables to consider, and their potential relationship to our findings.
“Finally, we also considered whether time to target [1,2]. (Supplementary D), participant forward hand position (Supplementary E), or learning [4] (Supplementary G-H) influenced feedback responses, but found that none impacted the observed differences between experimental conditions nor changed our interpretation. Our hypothesis that the sensorimotor system uses a representation of a partner and considers the partner’s costs to modify involuntary visuomotor feedback responses parsimoniously accounts for the differences observed between all conditions.”
(2) Synchrony
In our task, participants movements were not self-initiated. We had them begin the movement as soon as they hear an audible tone so that they would begin their movements at as similar a time as possible. We have analyzed the movement onset synchrony between participants within a pair, shown in Supplementary F.
Supplementary: “We calculated movement onset times at the time that the participants left the start target [8]. We then took the absolute value of the difference between the participants within a pair as a measure of movement onset synchrony. For Experiment 1, an ANOVA with movement onset synchrony as the dependent variable showed no main effect of self-target (F[1,47] = 1.38, p = 0.252), no main effect of partner target (F[1,47] = 0.057, p = 0.813), and no interaction (F[1,47] = 0.45, p = 0.508). For Experiment 2, an ANOVA with movement onset synchrony as the dependent variable showed no main effect of self-target (F[1,47] = 0.07, p = 0.788), no main effect of partner target (F[1,47] = 2.75, p = 0.111), and no interaction (F[1,47] = 2.31, p = 0.142).”
Further, we have modified our methods to emphasize that participants within a pair generally began their movement at the same time.
“Instead of self-initiating their movements, we specifically had participants move at the sound of a tone so that the movement onset between participants in a pair was as synchronous as possible (see Supplementary F for movement onset synchrony analysis).”
Reviewer #1 (Recommendations for the authors):
(1) Lines 291-292: One study extensively examined cursor and target jump visuomotor on set times and found no difference (Franklin et al., 2016; J Neuroscience), which strongly argues against this interpretation.
We thank the reviewer for pointing out this work. We have modified the following lines:
“However, other work by Franklin and colleagues (2016) found no difference in visuomotor feedback response latencies between cursor and target jumps [6].”
(2) Line 411: What were the instructions regarding partner performance in terms of the reward? Did you explain that individual performance alone will determine the reward?
As addressed above, we have made the following changes to emphasize the instructions given to participants.
“In other words, we ensured participants had a clear understanding that their performance in the task was only based on stabilizing the center cursor in their own self-target within the time constraint. Therefore, the instructions and timing constraints did not enforce participants to work together.”
(3) Line 506: Ten probe trials in each direction is very low. Can this still be in the transition state of the feedback response, rather than at steady state? There are many studies done looking at the learning of visuomotor responses in which changes are still occurring after several hundred trials (e.g., Franklin et al., 2017 J Neurophysiol; Franklin et al., 2008; J Neuroscience). In this experiment, each block only lasts 151 trials total if my calculations are correct. How certain are you that the results are at a steady state and not continuously changing? Perhaps with further experimental experience, the feedback responses would approach the predictions of a different model.
The reviewer raises an important point. We had run these analyses prior to submitting the manuscript and did not see anything. However, we believe this information is important to include since both we and yourself asked the same question. Specifically, we have analyzed the visuomotor feedback responses over the trials (Supplementary G), which shows little to no learning over time. Additionally, we also found no difference in the visuomotor feedback response trends between the first and second half of trials in each condition (Supplementary H). Therefore, it appears that the sensorimotor system was at steady state behaviour very quickly and we do believe that the feedback responses would approach the predictions of a different model if participants performed more trials. We have added the following
Supplementary: “Given there were 151 trials and 10 left/right probe trials for each experimental condition, it is possible that completing more trials may have lead to different involuntary visuomotor feedback responses. Therefore, we analysed the in voluntary visuomotor feedback responses over the course of each experimental condition. Visually, involuntary visuomotor feedback responses in neither Experiment 1 (Fig. S8) nor Experiment 2 (Fig. S9) show any consistent learning (see Fig. S10 for statistical analysis). Therefore, it appears participants rapidly formed a partner model based on knowledge of their movement goal to modify their involuntary visuomotor feedback responses.”
Supplementary: “Supplementary Fig. S10 shows the involuntary visuomotor feedback responses in the first half (A,C) and second half (B,D) for each experimental condition. In Experiment 1, we observed the same statistical results in the first half and second half of trials as the analysis of all trials. That is, we observed a significant interaction between self-target and partner target in the first half (F[1,47] = 37.09, p < 0.001) and second half (F[1,47] = 48.68, p < 0.001) of trials. Follow-up mean comparisons showed the same significant trends as our analysis of all trials in the main manuscript (see Fig. S10A-B).”
Supplementary: “In Experiment 2, we observed the same statistical results in the first half and second half of trials as the analysis of all trials. That is, we observed a significant interaction between self-target and partner target in the first half (F[1,47] = 9.42, p = 0.004) and second half (F[1,47] = 17.40, p < 0.001) of trials. Follow-up mean comparisons showed the same significant trends as our analysis of all trials in the main manuscript (Fig. S10C-D).”
Supplementary: “Showing the same involuntary visuomotor feedback response trends across the experimental conditions for the first half, second half, and all trials suggests that the sensorimotor system quickly formed a model of a partner and considered their costs to modify rapid motor responses.”
We have also added to the discussion:
“Finally, we also considered whether time to target [1,2] (Supplementary D), participant forward hand position (Supplementary E), or learning [4] (Supplementary G) influenced feedback responses, but found that none impacted the observed differences between experimental conditions nor changed our interpretation.”
(4) The authors should also discuss some of the prior work which is very relevant to the tasks studied: (Knill, Bondata & Chhabra, 2011, J Neuroscience). There may also be other papers that use this task for visuomotor feedback responses and therefore, should be included.
We have included the Knill 2011 paper and also Cross 2019 in our discussion:
“This modification of feedback responses based on a relevant/irrelevant task goal has also been shown in response to visual perturbations [7,8].”
(5) Lines 301-303: The terms ’relevant’ and ’irrelevant’ here describe different concepts than the ones used in this study. I suggest making a distinction to avoid confusion for the reader.
We thank the reviewer for pointing out that this is confusing. We’ve made the following changes to improve the clarity:
“Further, Franklin and colleagues (2008) designed a visual perturbation to be relevant or irrelevant when reaching to the same target, showing greater involuntary visuomotor feedback responses to a relevant visual perturbation compared to an irrelevant visual perturbation [9].”
(6) Line 459: The reaching movement was quite slow (25cm in about 1.2 seconds). Is this needed to ensure that both participants can complete the movements, given potentially very different start times? Please comment as this is different than many previous studies.
Participants needed to stabilize the cursor for 500ms in their target within a time constraint of 1400 - 1600 ms. Therefore, they had to reach the target between 900 - 1100 ms (before stabilizing). Additionally, participants did not perform self-initiated movements, but were required to begin their movement as soon as they heard an audible tone. Given that reaction times are ~200ms, participants had ~700 - 900 ms to reach the target, which aligns with previous research (Franklin et al. (2008), Franklin et al. (2012), Nashed et al. (2012)). We have clarified the time constraints of the task in our Methods:
“They therefore had 700 - 900 ms to first reach the target, since humans generally have response times ~200 ms, and they needed to stabilize within the target for 500 ms (i.e., 1400 - 200 - 500 = 700 ms and 1600 - 200 - 500 = 900 ms). Movement times of 700 - 900 ms are thus consistent with previous human reaching studies [4,9,10].”
(7) Reference [25] is incomplete
Thank you for catching this.
And thank you for the thoughtful and clear review. We feel it has greatly improved the quality and clarity of our manuscript!
Reviewer #2 (Public review):
Summary
Sullivan and colleagues studied the fast, involuntary, sensorimotor feedback control in interpersonal coordination. Using a cleverly designed joint-reaching experiment that separately manipulated the accuracy demands for a pair of participants, they demonstrated that the rapid visuomotor feedback response of a human participant to a sudden visual perturbation is modulated by his/her partner’s control policy and cost. The behavioral results are well-matched with the predictions of the optimal feedback control framework implemented with the dynamic game theory model. Overall, the study provides an important and novel set of results on the fast, involuntary feedback response in human motor control, in the context of interpersonal coordination.
We thank the reviewer for the kind words!
Review:
Sullivan and colleagues investigated whether fast, involuntary sensorimotor feedback control is modulated by the partner’s state (e.g., cost and control policy) during interpersonal coordination. They asked a pair of participants to make a reaching movement to control a cursor and hit a target, where the cursor’s position was a combination of each participant’s hand position. To examine fast visuomotor feedback response, the authors applied a sudden shift in either the cursor (experiment 1) or the target (experiment 2) position in the middle of movement. To test the involvement of partner’s information in the feedback response, they independently manipulated the accuracy demand for each participant by varying the lateral length of the target (i.e., a wider/narrower target has a lower/higher demand for correction when movement is perturbed). Because participants could also see their partner’s target, they could theoretically take this information (e.g., whether their partner would correct, whether their correction would help their partner, etc.) into account when responding to the sudden visual shift. Computationally, the task structure can be handled using dynamic game theory, and the partner’s feedback control policy and cost function are integrated into the optimal feedback control framework. As predicted by the model, the authors demonstrated that the rapid visuomotor feedback response to a sudden visual perturbation is modulated by the partner’s control policy and cost. When their partner’s target was narrow, they made rapid feedback corrections even when their own target was wide (no need for correction), suggesting integration of their partner’s cost function. Similarly, they made corrections to a lesser degree when both targets were narrower than when the partner’s target was wider, suggesting that the feedback correction takes the partner’s correction (i.e., feedback control policy) into account.
The strength of the current paper lies in the combination of clever behavioral experiments that independently manipulate each participant’s accuracy demand and a sophisticated computational approach that integrates optimal feedback control and dynamic game theory. Both the experimental design and data analysis sound good. While the main claim is well-supported by the results, the only current weakness is the lack of discussion of limitations and an alternative explanation. Adding these points will further strengthen the paper.
Reviewer #2 (Recommendations for the authors):
(1) While the current version is already well-written, it would be helpful for readers to further discuss the relationship between the current study and some potentially relevant studies, such as Braun et al. (2009), Ganesh et al. (2014), and Takagi et al. (2017) (2019).
Thank you for pointing out these papers that we missed, which we now cite appropriately in light of our own work. In particular, we have added the following to our discussion, including Braun et al. (2009) and Takagi et al. (2017) (2019). However, Beckers et al. (2020) showed conflicting results from Ganesh et al. (2014), and since these works are about learning, we feel it is outside the scope of our work.
“Further, others have shown that the sensorimotor system modifies movement selection according to game-theoretic predictions, [11] and that the sensorimotor system modifies movements using an estimate of the joint goal during human-human interactions [12,13].”
(2) For an alternative interpretation of the results, one could consider, for instance, that the target’s visual appearance could have served as a contextual cue for learning different movement gains in the lateral direction (e.g., whether the partner corrects the shift might be approximated as a gain change). Although less likely, this alternative account could be tested by simulation and would strengthen the argument.
This a thoughtful comment, also brought up by Reviewer 1. Here we provide our previous response that addresses this concern. While it is possible that the change in the visuomotor feedback responses could be just from a scaling factor. This hypothesis could explain the difference between two conditions, but would fail to explain differences between two other conditions. Specifically, this hypothesis could explain a decrease in involuntary visuomotor feedback responses between partner-irrelevant/self-relevant and partner-relevant/self-relevant. Critically, this hypothesis could not explain the difference between partner-irrelevant/self-irrelevant and partner-relevant/self-irrelevant. That is, there is no reason to scale a response to correct for a partner’s relevant target when your own target is irrelevant. However, our finding that there is a greater involuntary visuomotor feedback response in partner-relevant/self-irrelevant compared to partner irrelevant/self-irrelevant is predicted by the notion that humans form a representation of others and consider their movement costs.
We have added a paragraph in the discussion to justify our hypothesis over the scaling factor hypothesis.
“Our hypothesis that the sensorimotor system uses a representation of a partner and considers the partner’s costs to modify involuntary visuomotor feedback responses can parsimoniously explain all of our experimental findings. There are a few alternative hypotheses that could explain a subset of results. One alternative hypothesis is that participants simply learned the hand to center cursor mapping in each experimental condition. That is, instead of using a model of their partner, participants simply adapted to the dynamics of the center cursor. However, this hypothesis would not predict an increased involuntary visuomotor feedback response in the partner-relevant/self-irrelevant condition compared to the partner-irrelevant/self-irrelevant condition. If participants did not form a model of their partner nor consider their partner’s costs, then they would not display an increased feedback response when they had an irrelevant target and their partner’s target was relevant. An increased feedback response to help a partner achieve their goal is captured by our hypothesis that the sensorimotor system uses a representation of a partner and considers the partner’s costs to modify involuntary visuomotor feedback responses.”
(3) Another (maybe unlikely) alternative interpretation is that the targets’ visual appearances might have been confusing. One might find that the closed square is common to both targets for the “Partner Relevant Self Irrelevant” and the “Partner Relevant Self Relevant”, and that this might have elicited the response to perturbation in “Partner Relevant Self Irrelevant”. Related to this point, it would be informative to describe how the “cooperative” fast feedback response developed over the course of the experiment, for instance, by comparing behaviors across experimental blocks.
We have partitioned this question into two responses, relating to visual appearance of the targets and the development (i.e., learning) of visuomotor feedback responses over the course of the experiments.
(1) Participants confused by visual appearance of the targets.
We were also concerned that participants might be confused by the targets, and therefore confirmed with participants after the experiment that they correctly understood that the light grey filled rectangle was their own target and the dark grey hollow rectangle was their partners. Furthermore, in the partner-relevant/self-irrelevant, partner-irrelevant/self-relevant, and partner-relevant/self-relevant conditions, there is a small square target in each of the conditions. However, we found that the partner-irrelevant/self-relevant and partner-relevant/self-relevant conditions both elicited significantly greater involuntary visuomotor feedback responses than the partner-relevant/self-irrelevant condition. Thus, participants involuntary visuomotor feedback responses suggest that they correctly formed different representations based on an accurate understanding of the self vs partner target. The other reviewer had related comments about the visual stimuli, which we also address within the discussion.
“Another alternative hypothesis would be that the sensorimotor system was responding only to the relevant target displayed on the screen. Again, this hypothesis would only explain a subset of our results. In particular, this relevant target hypothesis cannot explain the observed differences between the partner-relevant/self-irrelevant and partner-irrelevant/self-relevant conditions in both Experiments 1 and 2.”
(2) Comparing feedback responses over time
We have included the visuomotor feedback responses over each experimental condition in Supplementary G. Notably, we did not find any learning effect, suggesting that the sensorimotor system quickly developed a model of a partner’s behaviour and used that model to modify feedback responses. We have also added a paragraph on learning to our discussion.
We’ve addressed how learning did not play a role in this study:
“Finally, we also considered whether time to target [1,2] (Supplementary D), participant forward hand position (Supplementary E), or learning [4] (Supplementary G-H) influenced feedback responses, but found that none impacted the observed differences between experimental conditions nor changed our interpretation.”
Supplementary: “Given there were 151 trials and 10 left/right probe trials for each experimental condition, it is possible that completing more trials may have lead to different in voluntary visuomotor feedback responses. Therefore, we analysed the in voluntary visuomotor feedback responses over the course of each experimental condition. Visually, involuntary visuomotor feedback responses in neither Experiment 1 (Fig. S8) nor Experiment 2 (Fig. S9) show any consistent learning (see Fig. S10 for statistical analysis). Therefore, it appears participants rapidly formed a partner model based on knowledge of their movement goal to modify their involuntary visuomotor feedback responses.”
Supplementary: “Supplementary Fig. S10 shows the involuntary visuomotor feedback responses in the first half (A,C) and second half (B,D) for each experimental condition. In Experiment 1, we observed the same statistical results in the first half and second half of trials as the analysis of all trials. That is, we observed a significant interaction between self-target and partner target in the first half (F[1,47] = 37.09, p < 0.001) and second half (F[1,47] = 48.68, p < 0.001) of trials. Follow-up mean comparisons showed the same significant trends as our analysis of all trials in the main manuscript (see Fig. S10A-B).”
Supplementary: “Supplementary Fig. S10 shows the involuntary visuomotor feedback responses in the first half (A,C) and second half (B,D) for each experimental condition. In Experiment 1, we observed the same statistical results in the first half and second half of trials as the analysis of all trials. That is, we observed a significant interaction between self-target and partner target in the first half (F[1,47] = 37.09, p < 0.001) and second half (F[1,47] = 48.68, p <0.001) of trials. Follow-up mean comparisons showed the same significant trends as our analysis of all trials in the main manuscript (see Fig. S10A-B).”
Supplementary: “Showing the same involuntary visuomotor feedback response trends across the experimental conditions for the first half, second half, and all trials suggests that the sensorimotor system used a model of a partner based on their goals and considered their costs to modify rapid motor responses.”
(4) It looks slightly counter intuitive (and therefore interesting) that the participant shows some amount of fast feedback responses in the “Partner Relevant Self Irrelevant” condition, since they were instructed to only consider the self-target. Based on the results, the authors suggest an altruistic feature of the motor system (lines 333-340). It would be helpful to clarify the basis for this interpretation, whether it is formally derived from the game-theoretic framework or represents a more conceptual interpretation. Providing additional explanation that translates the game-theoretic reasoning into more accessible, intuitive terms would help readers better understand and evaluate this claim.
We are glad the reviewer also finds this result interesting. The reviewer raises an important point that there needs to be a more clear explanation for why we believe this result was found. We have made the following changes to the discussion:
“Furthermore, this result is predicted by our dynamic game theory models that include the partner’s costs in the self cost function. In other words, a dynamic game theory model that selects feedback gains to minimize both the self and partner cost reflects an altruistic control policy.”
(5) Please check whether all references are displayed correctly. Some of them (e.g., 25, 65) seemed not correctly shown in the References section.
We have fixed the citation.
We thank the reviewer for providing a clear and insightful review. Their comments have significantly improved the manuscript.
References
(1) Česonis, J., & Franklin, D. W. (2020). Time-to-Target Simplifies Optimal Control of Visuomotor Feedback Responses. eneuro, 7 (2), ENEURO.0514–19.2020.
(2) Česonis, J., & Franklin, D. W. (2022). Contextual Cues Are Not Unique for Motor Learning: Task-dependant Switching of Feedback Controllers. PLOS Computational Biology, 18 (6), ed. by Haith, A. M.: e1010192.
(3) Crevecoeur, F., Kurtzer, I., Bourke, T., & Scott, S. H. (2013). Feedback Responses Rapidly Scale with the Urgency to Correct for External Perturbations. Journal of Neurophysiology, 110 (6), 1323–1332.
(4) Franklin, S., Wolpert, D. M., & Franklin, D. W. (2012). Visuomotor Feedback Gains Upregulate during the Learning of Novel Dynamics. Journal of Neurophysiology, 108 (2), 467–478.
(5) Liu, Y., Leib, R., Dudley, W., Shafti, A., Faisal, A. A., & Franklin, D. W. (2025). Partner-Sourced Haptic Feedback Rather than Environmental Inputs Drives Coordination Improvement in Human Dyadic Collaboration. Scientific Reports, 15 (1), 40347.
(6) Franklin, D. W., Reichenbach, A., Franklin, S., & Diedrichsen, J. (2016). Temporal Evolution of Spatial Computations for Visuomotor Control. The Journal of Neuroscience, 36 (8), 2329–2341.
(7) Knill, D. C., Bondada, A., & Chhabra, M. (2011). Flexible, Task-Dependent Use of Sensory Feedback to Control Hand Movements. The Journal of Neuroscience, 31 (4), 1219–1237.
(8) Cross, K. P., Cluff, T., Takei, T., & Scott, S. H. (2019). Visual Feedback Processing of the Limb Involves Two Distinct Phases. The Journal of Neuroscience, 39 (34), 6751–6765.
(9) Franklin, D. W., & Wolpert, D. M. (2008). Specificity of Reflex Adaptation for Task-Relevant Variability. The Journal of Neuroscience, 28 (52), 14165–14175.
(10) Nashed, J. Y., Crevecoeur, F., & Scott, S. H. (2012). Influence of the Behavioral Goal and Environmental Obstacles on Rapid Feedback Responses. Journal of Neurophysiology, 108 (4), 999–1009.
(11) Braun, D. A., Ortega, P. A., & Wolpert, D. M. (2009). Nash Equilibria in Multi-Agent Motor Interactions. PLoS Computational Biology, 5 (8), ed. by Friston, K. J.: e1000468.
(10) Takagi, A., Ganesh, G., Yoshioka, T., Kawato, M., & Burdet, E. (2017). Physically Interacting Individuals Estimate the Partner’s Goal to Enhance Their Movements. Nature Human Behaviour, 1 (3), 0054.
(11) Takagi, A., Hirashima, M., Nozaki, D., & Burdet, E. (2019). Individuals Physically Interacting in a Group Rapidly Coordinate Their Movement by Estimating the Collective Goal. eLife, 8 , e41328.
-

eLife Assessment
This important study combines a two-person joint hand-reaching paradigm with game-theoretical modeling to examine whether, and how, one's reflexive visuomotor responses are modulated by a partner's control policy and cost structure. The study provides a solid and novel set of behavioral findings suggesting that involuntary visuomotor feedback is indeed modulated in the context of interpersonal coordination. The work will be of interest to cognitive scientists studying the motoric and social aspects of action control.
-

Reviewer #1 (Public review):
Summary:
Sullivan and colleagues examined the modulation of reflexive visuomotor responses during collaboration between pairs of participants performing a joint reaching movement to a target. In their experiments, the players jointly controlled a cursor that they had to move towards narrow or wide targets. In each experimental block, each participant had a different type of target they had to move the joint cursor to. During the experiment, the authors used lateral perturbation of the cursor to test participants' fast feedback responses to the different target types. The authors suggest participants integrate the target type and related cost of their partner into their own movements, which suggests that visuomotor gains are affected by the partner's task.
Strengths:
The topic of the manuscript is very …
Reviewer #1 (Public review):
Summary:
Sullivan and colleagues examined the modulation of reflexive visuomotor responses during collaboration between pairs of participants performing a joint reaching movement to a target. In their experiments, the players jointly controlled a cursor that they had to move towards narrow or wide targets. In each experimental block, each participant had a different type of target they had to move the joint cursor to. During the experiment, the authors used lateral perturbation of the cursor to test participants' fast feedback responses to the different target types. The authors suggest participants integrate the target type and related cost of their partner into their own movements, which suggests that visuomotor gains are affected by the partner's task.
Strengths:
The topic of the manuscript is very interesting, and the authors are using well-established methodology to test their hypothesis. They combine experimental studies with optimal control models to further support their work. Overall, the manuscript is very timely and shows important findings - that the feedback responses reflect both our and our partner's tasks.
Weaknesses:
However, in the current version of the manuscript, I believe the results could also be interpreted differently, which suggests that the authors should provide further support for their hypothesis and conclusions.
Major Comments:
(1) Results of the relevant conditions:
In addition to the authors' explanation regarding the results, it is also possible that the results represent a simple modulation of the reflexive response to a scaled version of cursor movement. That is, when the cursor is partially controlled by a partner, which also contributes to reducing movement error, it can also be interpreted by the sensorimotor system as a scaling of hand-to-cursor movement. In this case, the reflexes are modulated according to a scaling factor (how much do I need to move to bring the cursor to the target). I believe that a single-agent simulation of an OFC model with a scaling factor in the lateral direction can generate the same predictions as those presented by the authors in this study. In other words, maybe the controller has learned about the nature of the perturbation in each specific context, that in some conditions I need to control strongly, whereas in others I do not (without having any model of the partner). I suggest that the authors demonstrate how they can distinguish their interpretation of the results from other explanations.
(2) The effect of the partner target:
The authors presented both self and partner targets together. While the effect of each target type, presented separately, is known, it is unclear how presenting both simultaneously affects individual response. That is, does a small target with a background of the wide target affect the reflexive response in the case of a single participant moving? The results of Experiment 2, comparing the case of partner- and self-relevant targets versus partner-irrelevant and self-relevant targets, may suggest that the system acted based on the relevant target, regardless of the presence and instructions regarding the self-target.
(3) Experiment instructions:
It is unclear what the general instructions were for the participants and whether the instructions provided set the proposed weighted cost, which could be altered with different instructions.
(4) Some work has shown that the gain of visuomotor feedback responses reflects the time to target and that this is updated online after a perturbation (Cesonis & Franklin, 2020, eNeuro; Cesonis and Franklin, 2021, NBDT; also related to Crevecoeur et al., 2013, J Neurophysiol). These models would predict different feedback gains depending on the distance remaining to the target for the participant and the time to correct for the jump, which is directly affected by the small or large targets. Could this time be used to target instead of explaining the results? I don't believe that this is the case, but the authors should try to rule out other interpretations. This is maybe a minor point, but perhaps more important is the location (& time remaining) for each participant at the time of the jump. It appears from the figures that this might be affected by the condition (given the change in movement lengths - see Figure 3 B & C). If this is the case, then could some of the feedback gain be related to these parameters and not the model of the partner, as suggested? Some evidence to rule this out would be a good addition to the paper - perhaps the distance of each partner at the time of the perturbation, for example. In addition, please analyze the synchrony of the two partners' movements.
-

Reviewer #2 (Public review):
Summary:
Sullivan and colleagues studied the fast, involuntary, sensorimotor feedback control in interpersonal coordination. Using a cleverly designed joint-reaching experiment that separately manipulated the accuracy demands for a pair of participants, they demonstrated that the rapid visuomotor feedback response of a human participant to a sudden visual perturbation is modulated by his/her partner's control policy and cost. The behavioral results are well-matched with the predictions of the optimal feedback control framework implemented with the dynamic game theory model. Overall, the study provides an important and novel set of results on the fast, involuntary feedback response in human motor control, in the context of interpersonal coordination.
Review:
Sullivan and colleagues investigated whether fast, …
Reviewer #2 (Public review):
Summary:
Sullivan and colleagues studied the fast, involuntary, sensorimotor feedback control in interpersonal coordination. Using a cleverly designed joint-reaching experiment that separately manipulated the accuracy demands for a pair of participants, they demonstrated that the rapid visuomotor feedback response of a human participant to a sudden visual perturbation is modulated by his/her partner's control policy and cost. The behavioral results are well-matched with the predictions of the optimal feedback control framework implemented with the dynamic game theory model. Overall, the study provides an important and novel set of results on the fast, involuntary feedback response in human motor control, in the context of interpersonal coordination.
Review:
Sullivan and colleagues investigated whether fast, involuntary sensorimotor feedback control is modulated by the partner's state (e.g., cost and control policy) during interpersonal coordination. They asked a pair of participants to make a reaching movement to control a cursor and hit a target, where the cursor's position was a combination of each participant's hand position. To examine fast visuomotor feedback response, the authors applied a sudden shift in either the cursor (experiment 1) or the target (experiment 2) position in the middle of movement. To test the involvement of partner's information in the feedback response, they independently manipulated the accuracy demand for each participant by varying the lateral length of the target (i.e., a wider/narrower target has a lower/higher demand for correction when movement is perturbed). Because participants could also see their partner's target, they could theoretically take this information (e.g., whether their partner would correct, whether their correction would help their partner, etc.) into account when responding to the sudden visual shift. Computationally, the task structure can be handled using dynamic game theory, and the partner's feedback control policy and cost function are integrated into the optimal feedback control framework. As predicted by the model, the authors demonstrated that the rapid visuomotor feedback response to a sudden visual perturbation is modulated by the partner's control policy and cost. When their partner's target was narrow, they made rapid feedback corrections even when their own target was wide (no need for correction), suggesting integration of their partner's cost function. Similarly, they made corrections to a lesser degree when both targets were narrower than when the partner's target was wider, suggesting that the feedback correction takes the partner's correction (i.e., feedback control policy) into account.
The strength of the current paper lies in the combination of clever behavioral experiments that independently manipulate each participant's accuracy demand and a sophisticated computational approach that integrates optimal feedback control and dynamic game theory. Both the experimental design and data analysis sound good. While the main claim is well-supported by the results, the only current weakness is the lack of discussion of limitations and an alternative explanation. Adding these points will further strengthen the paper.
-

-