The insulin / IGF axis is critically important for controlling gene transcription in the podocyte
Curation statements for this article:-
Curated by eLife
eLife Assessment
This valuable study used genetic and pharmacological manipulations of insulin/IGF signaling to address the role of insulin/IGF axis in the function of renal glomerular podocyte. Solid data are presented to demonstrate that co-inhibition of insulin/IGF signaling in podocytes led to aberrant splicing of mRNAs, which could contribute to the loss of podocytes in vitro and in vivo in mice. In light of the fact that IR/IGF-1R signaling are critically required for normal development and growth in multiple cells and organs, the lack of the assessment of developmental phenotype of podocytes in the mouse model limits the interpretation of the data.
[Editors' note: this paper was reviewed by Review Commons.]
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
Signalling to the podocyte via the structurally related insulin receptor (IR) or insulin-like growth factor 1 receptor (IGF1R) is important for podocyte function. This study sought to elucidate the compound role of the insulin/IGF1 axis in podocytes using transgenic mice and cell culture models deficient in both receptors.
Podocyte specific IR/IGF1R knockdown mice developed a severe kidney phenotype with albuminuria, glomerulosclerosis and renal failure with death occurring in some mice between 4 and 24 weeks. Simultaneous knockdown of both receptors in cultured podocytes resulted in >50% cell death by 7 days.
Proteomic analysis revealed a striking downregulation of spliceosome-related proteins in IR/IGF1R knockdown podocytes with long-read RNA sequence data indicating an increased fraction of transcripts with intron retention/premature termination codons in these cells. Furthermore, phospho-proteomic analysis revealed multiple insulin / IGF1 induced spliceosomal post-translational protein and kinase modifications suggesting dynamic control of this system.
This study underlines the critical importance of podocyte insulin/IGF signalling revealing a novel role for this extrinsic hormonal signalling axis in regulating gene transcription in this cell type.
Article activity feed
-

-

-

eLife Assessment
This valuable study used genetic and pharmacological manipulations of insulin/IGF signaling to address the role of insulin/IGF axis in the function of renal glomerular podocyte. Solid data are presented to demonstrate that co-inhibition of insulin/IGF signaling in podocytes led to aberrant splicing of mRNAs, which could contribute to the loss of podocytes in vitro and in vivo in mice. In light of the fact that IR/IGF-1R signaling are critically required for normal development and growth in multiple cells and organs, the lack of the assessment of developmental phenotype of podocytes in the mouse model limits the interpretation of the data.
[Editors' note: this paper was reviewed by Review Commons.]
-

Reviewer #1 (Public review):
Summary:
In this manuscript, the role of the insulin receptor and the insulin growth factor receptor was investigated in podocytes. Mice, where both receptors were deleted, developed glomerular dysfunction and developed proteinuria and glomerulrosclerosis over several months. Because of concerns about incomplete KO, the authors generated and studied podocyte cell lines where both receptors were deleted. Loss of both receptors was highly deleterious with greater than 50% cell death. To elucidate the mechanism of cell death, the authors performed global proteomics and found that spliceosome proteins were downregulated. They confirmed this directly by using long-read sequencing. These results suggest a novel role for insulin and IGF1R signaling in RNA splicing in podocytes.
This is primarily a descriptive study …
Reviewer #1 (Public review):
Summary:
In this manuscript, the role of the insulin receptor and the insulin growth factor receptor was investigated in podocytes. Mice, where both receptors were deleted, developed glomerular dysfunction and developed proteinuria and glomerulrosclerosis over several months. Because of concerns about incomplete KO, the authors generated and studied podocyte cell lines where both receptors were deleted. Loss of both receptors was highly deleterious with greater than 50% cell death. To elucidate the mechanism of cell death, the authors performed global proteomics and found that spliceosome proteins were downregulated. They confirmed this directly by using long-read sequencing. These results suggest a novel role for insulin and IGF1R signaling in RNA splicing in podocytes.
This is primarily a descriptive study and no technical concerns are raised. The mechanism of how insulin and IGF1 signaling regulates splicing is not directly addressed but implicates potentially the phosphorylation downstream of these receptors. In the revised manuscript, it is shown that the mouse KO is incomplete potentially explaining the slow onset of renal insufficiency. Direct measurement of GFR and serial serum creatinines might also enhance our understanding of progression of disease, proteinuria is a strong sign of renal injury. An attempt to rescue the phenotype by overexpression of SF3B4 would also be useful but may be masked by defects in other spliceosome genes. As insulin and IGF are regulators of metabolism, some assessment of metabolic parameters would be an optional add-on.
Significance:
With the GLP1 agonists providing renal protection, there is great interest in understanding the role of insulin and other incretins in kidney cell biology. It is already known that Insulin and IGFR signaling play important roles in other cells of the kidney. So, there is great interest in understanding these pathways in podocytes. The major advance is that these two pathways appear to have a role in RNA metabolism.
Latest comments:
The new reviewer raised two major points, whether the KO effect on splicing is specific to IGF1 and whether the interpretation could be developmental rather than due to splicing. The reviewer raises some important issues but the evidence to suggest that this is specific is data in the literature that IR/IGF signaling is already known to regulate splicing and that splicing defects were not detected in other models that they have analyzed. I agree with the reviewer (and authors) that the incomplete floxing of the genes is a major complication. The point that there could be a developmental defect with mice being born with fewer podocytes and perhaps the authors should caveat this point. The fact that they mice are born with normal function, that renal function can be maintained with up to 80% loss of podocytes suggest that they are likely born with a good number of podocytes and the dysfunction that occurs at 6 months is due to a process, induced by the loss of IR/IGF signaling that is detrimental to the podocyte.
-

Reviewer #2 (Public review):
Summary:
In this manuscript, submitted to Review Commons (journal agnostic), Coward and colleagues report on the role of insulin/IGF axis in podocyte gene transcription. They knocked out both the insulin and IGFR1 mice. Dual KO mice manifested a severe phenotype, with albuminuria, glomerulosclerosis, renal failure and death at 4-24 weeks.
Long read RNA sequencing was used to assess splicing events. Podocyte transcripts manifesting intron retention were identified. Dual knock-out podocytes manifested more transcripts with intron retention (18%) compared wild-type controls (18%), with an overlap between experiments of ~30%.
Transcript productivity was also assessed using FLAIR-mark-intron-retention software. Intron retention w seen in 18% of ciDKO podocyte transcripts compared to 14% of wild-type podocyte …
Reviewer #2 (Public review):
Summary:
In this manuscript, submitted to Review Commons (journal agnostic), Coward and colleagues report on the role of insulin/IGF axis in podocyte gene transcription. They knocked out both the insulin and IGFR1 mice. Dual KO mice manifested a severe phenotype, with albuminuria, glomerulosclerosis, renal failure and death at 4-24 weeks.
Long read RNA sequencing was used to assess splicing events. Podocyte transcripts manifesting intron retention were identified. Dual knock-out podocytes manifested more transcripts with intron retention (18%) compared wild-type controls (18%), with an overlap between experiments of ~30%.
Transcript productivity was also assessed using FLAIR-mark-intron-retention software. Intron retention w seen in 18% of ciDKO podocyte transcripts compared to 14% of wild-type podocyte transcripts (P=0.004), with an overlap between experiments of ~30% (indicating the variability of results with this method). Interestingly, ciDKO podocytes showed downregulation of proteins involved in spliceosome function and RNA processing, as suggested by LC/MS and confirmed by Western blot.
Pladienolide (a spliceosome inhibitor) was cytotoxic to HeLa cells and to mouse podocytes but no toxicity was seen in murine glomerular endothelial cells.
The manuscript is generally clear and well-written. Mouse work was approved in advance. The four figures are generally well-designed, bars/superimposed dot-plots.
Methods are generally well described.
Comments on previous version:
Coward and colleagues have done an excellent job of responding to all the reviewer comments.
-

Reviewer #4 (Public review):
Summary and background:
This report entitled "The insulin/IGF axis is critically important (for) controlling gene transcription in the podocyte" from Hurcombe et al is based on a mouse double knockdown of the IR and IGF1R and a parallel cultured mouse podocyte model. Insulin/IGF signaling system in mammals evolved as three gene reduplicated peptides (insulin, IGF-1, and IGF-2) and their two receptors IR and IGF1R that cross-react to variable extents with the peptides, are ubiquitously expressed, and signal through parallel pathways. The major downstream effect of insulin is to regulate glucose uptake and metabolism, while that of the IGF pathways is to regulate growth and cell cycling in part through mTORC1. The GH-IGF-1-IGF1R pathway regulates post-natal growth. IGF-2 signaling is thought to play a major …
Reviewer #4 (Public review):
Summary and background:
This report entitled "The insulin/IGF axis is critically important (for) controlling gene transcription in the podocyte" from Hurcombe et al is based on a mouse double knockdown of the IR and IGF1R and a parallel cultured mouse podocyte model. Insulin/IGF signaling system in mammals evolved as three gene reduplicated peptides (insulin, IGF-1, and IGF-2) and their two receptors IR and IGF1R that cross-react to variable extents with the peptides, are ubiquitously expressed, and signal through parallel pathways. The major downstream effect of insulin is to regulate glucose uptake and metabolism, while that of the IGF pathways is to regulate growth and cell cycling in part through mTORC1. The GH-IGF-1-IGF1R pathway regulates post-natal growth. IGF-2 signaling is thought to play a major role in regulating intrauterine growth and development, although IGF-2 is also present at high levels in post-natal life. Thus, one would anticipate that reducing IR/IGF1R signaling in any cell would slow growth and cell cycling by reducing growth factor and metabolic mTORC1-mediated and other processes including the splicing of RNA for protein synthesis.
Comments on revised version:
The second sentence of the Summary reads "This study sought to elucidate the compound role of the insulin/IGF1 axis in podocytes using transgenic mice and cell culture models deficient in both receptors." The study design and rationale for the proteosome analysis described is predicated on the finding that podocyte-specific knockdown of the IR/IGF-1R in mice is associated with development of proteinuria and reduced eGFR by 20months of life. Since the IR/IGF-1R are critically required for normal development and growth of all cells and organs, the obvious explanation for the observation would be that the model system results in defective podocyte development and deployment (caused by reduced IR/IGF-1) that, in turn, causes subsequent development of proteinuria and glomerulosclerosis (that may be much less dependent on a normal level of IR/IGF-1R expression). Thus, the experimental design does not allow a distinction between podocyte development and steady state function which are different biologic processes. The data provided does not examine podocyte status immediately after birth to confirm that podocyte number and size and structure is normal in mice that subsequently develop proteinuria and glomerulosclerosis. The response to the reviewer suggests that since this would require additional mice it has not been undertaken in order to reduce animal usage. This is not a valid argument, particularly when the investigators have not even used state-of-the-art methods to measure podocyte number, size and density in adult mice, key parameters that would be required to interpret their data. Counting podocyte nuclear number in glomerular cross-sections is simply an inadequate method, even if it is used and reported in other journals, and particularly where the examples given to justify its use can hardly be viewed as representing first rate science.
If the absence of studies that would answer the above questions, the investigators should add a sentence to the Discussion dealing with study limitations as follows. "The study design does not allow us to determine whether the primary effect of reduced IR/IGF-1R expression on the phenotype is during in utero and post-natal podocyte development and deployment, during periods of rapid growth when IGF-1 levels are highest, in steady state adult podocytes, or under all of the above conditions".
-

Author response:
The following is the authors’ response to the previous reviews
Public Reviews:
Reviewer #1 (Public review):
Summary:
In this manuscript, the role of the insulin receptor and the insulin growth factor receptor was investigated in podocytes. Mice, where both receptors were deleted, developed glomerular dysfunction and developed proteinuria and glomerulrosclerosis over several months. Because of concerns about incomplete KO, the authors generated and studied podocyte cell lines where both receptors were deleted. Loss of both receptors was highly deleterious with greater than 50% cell death. To elucidate the mechanism of cell death, the authors performed global proteomics and found that spliceosome proteins were downregulated. They confirmed this directly by using long-read sequencing. These results suggest a novel …
Author response:
The following is the authors’ response to the previous reviews
Public Reviews:
Reviewer #1 (Public review):
Summary:
In this manuscript, the role of the insulin receptor and the insulin growth factor receptor was investigated in podocytes. Mice, where both receptors were deleted, developed glomerular dysfunction and developed proteinuria and glomerulrosclerosis over several months. Because of concerns about incomplete KO, the authors generated and studied podocyte cell lines where both receptors were deleted. Loss of both receptors was highly deleterious with greater than 50% cell death. To elucidate the mechanism of cell death, the authors performed global proteomics and found that spliceosome proteins were downregulated. They confirmed this directly by using long-read sequencing. These results suggest a novel role for insulin and IGF1R signaling in RNA splicing in podocytes.
This is primarily a descriptive study and no technical concerns are raised. The mechanism of how insulin and IGF1 signaling regulates splicing is not directly addressed but implicates potentially the phosphorylation downstream of these receptors. In the revised manuscript, it is shown that the mouse KO is incomplete potentially explaining the slow onset of renal insufficiency. Direct measurement of GFR and serial serum creatinines might also enhance our understanding of progression of disease, proteinuria is a strong sign of renal injury. An attempt to rescue the phenotype by overexpression of SF3B4 would also be useful but may be masked by defects in other spliceosome genes. As insulin and IGF are regulators of metabolism, some assessment of metabolic parameters would be an optional add-on.
Significance:
With the GLP1 agonists providing renal protection, there is great interest in understanding the role of insulin and other incretins in kidney cell biology. It is already known that Insulin and IGFR signaling play important roles in other cells of the kidney. So, there is great interest in understanding these pathways in podocytes. The major advance is that these two pathways appear to have a role in RNA metabolism.
Comments on revised version:
I'm satisfied with the revised manuscript and the responses to my previous concerns.
Thank you.
Reviewer #2 (Public review):
Summary:
In this manuscript, submitted to Review Commons (journal agnostic), Coward and colleagues report on the role of insulin/IGF axis in podocyte gene transcription. They knocked out both the insulin and IGFR1 mice. Dual KO mice manifested a severe phenotype, with albuminuria, glomerulosclerosis, renal failure and death at 4-24 weeks.
Long read RNA sequencing was used to assess splicing events. Podocyte transcripts manifesting intron retention were identified. Dual knock-out podocytes manifested more transcripts with intron retention (18%) compared wild-type controls (18%), with an overlap between experiments of ~30%.
Transcript productivity was also assessed using FLAIR-mark-intron-retention software. Intron retention w seen in 18% of ciDKO podocyte transcripts compared to 14% of wild-type podocyte transcripts (P=0.004), with an overlap between experiments of ~30% (indicating the variability of results with this method). Interestingly, ciDKO podocytes showed downregulation of proteins involved in spliceosome function and RNA processing, as suggested by LC/MS and confirmed by Western blot.
Pladienolide (a spliceosome inhibitor) was cytotoxic to HeLa cells and to mouse podocytes but no toxicity was seen in murine glomerular endothelial cells.
The manuscript is generally clear and well-written. Mouse work was approved in advance. The four figures are generally well-designed, bars/superimposed dot-plots.
Methods are generally well described.
Comments on revised version:
Coward and colleagues have done an excellent job of responding to all the reviewer comments.
Thank you.
Reviewer #4 (Public review):
Summary and background:
This report entitled "The insulin/IGF axis is critically important (for) controlling gene transcription in the podocyte" from Hurcombe et al is based on a mouse double knockdown of the IR and IGF1R and a parallel cultured mouse podocyte model. Insulin/IGF signaling system in mammals evolved as three gene reduplicated peptides (insulin, IGF-1, and IGF-2) and their two receptors IR and IGF1R that cross-react to variable extents with the peptides, are ubiquitously expressed, and signal through parallel pathways. The major downstream effect of insulin is to regulate glucose uptake and metabolism, while that of the IGF pathways is to regulate growth and cell cycling in part through mTORC1. The GH-IGF-1-IGF1R pathway regulates post-natal growth. IGF-2 signaling is thought to play a major role in regulating intrauterine growth and development, although IGF-2 is also present at high levels in post-natal life. Thus, one would anticipate that reducing IR/IGF1R signaling in any cell would slow growth and cell cycling by reducing growth factor and metabolic mTORC1-mediated and other processes including the splicing of RNA for protein synthesis.
Thank you for this new extra review and assessing our paper with new suggestions (we addressed the previous suggestions to the satisfaction of other reviewers). Of note -regarding this introduction – the podocyte is a terminally differentiated cell and may have unique responses to insulin / IGF as it is accepted it does not generally proliferate (hence we consider understanding the actions of insulin / IGF and their receptors to be of interest). Indeed, we have recently shown a contrasting effect of IGF signalling in the podocyte. Partial suppression of the IGF1 receptor is beneficial in contrast to near complete suppression that results in mitochondrial dysfunction (PMID:38706850).
Mouse IR/IGF1R double knockdown model:
A double knockdown mouse model was generated by interbreeding mice with different genetic backgrounds carrying floxed sites for IR and IGF-1R to produce mixed background offspring with both floxed IR and IGF-1R genes. These mice were crossed so that the podocin promoter driven-Cre (that comes on at about embryonic day 12 bas podocytes are developing) would delete IR and IGF-1R genes. Since podocin is believed to be an absolutely podocyte-specific protein, this podocin promoter this is predicted to specifically knock down the IR and IGF1R genes only in podocytes. The weight and growth of double KO offspring was not different from controls, but some proportion of the double knockdown mice subsequently developed proteinuria by 6 months and 20% died, although no specific data is provided to identify the cause of the deaths since eGFR was not decreased. Surviving mice were evaluated at 6 months of age. The efficacy of knockdown was not demonstrated in the mouse model itself, although a temperature-sensitive cell line developed from these double knockdown mice showed that expression of IR and IGF-1R proteins in the Cre-treated cell line were both reduced by about 50% (no statistical analysis of this result provided).
In the knockout mice, proteinuria was significantly increased by 6 months, but not at earlier time points. Histologic analysis showed proteinaceous casts, glomerulosclerosis and interstitial fibrosis. Podocyte number was stated to be reduced by about 30% in double knockdown mice, although the method by which this was evaluated seems to have been by counting WT1 positive nuclei in glomerular cross-sections, an approach that is well-known not to be a reliable way of assessing true podocyte number. No information is provided about podocyte size, density or glomerular volume.
Comment: If IR/IGF1R deletion plays a significant role in normal podocyte function sufficient to cause proteinuria and glomerulosclerosis then the effect of reduced IR and IGF1R protein expression on podocyte function would have been expected to produce a phenotype before 6 months. A more likely scenario to explain the overall result is that deleting the IR and IGF1R genes at about embryonic day12 impacted podocyte development to a variable extent such that some mice developed fewer podocytes per glomerulus than other mice. As mice grow and their glomeruli and glomerular capillary area increases, those mice with fewer podocytes would not be able to completely cover the filtration surface with foot processes and would develop proteinuria and glomerulosclerosis. If reduced podocyte number per glomerulus is the proximate cause of the observed proteinuria, then modulation of the body and kidney growth rate by calorie restriction to slow growth (lower circulating IGF-1 levels) would be expected to be protective, while a high protein high calorie diet (higher circulating IGF-1 levels) or uni-nephrectomy to increase kidney growth rate would be expected to enhance proteinuria and glomerulosclerosis.
Thank you for these comments. In response to them:
(1) WT1 as a marker of podocyte number. We agree may not be the most accurate way of precisely measuring podocyte number but is widely accepted in the field (PMID:33655004 / PMID:38542564) and we think convincingly shows fewer podocytes at 6-months.
(2) Podocyte size and density was not measured. This was not the focus of the paper and the histology obviously showed a significant phenotype in several mice (Figs 1D-F). Of note we did objectively assess a glomeruloscleorosis index (Fig 1D). We took the approach to understand mechanism through non-biased proteomics and phospho-proteomics of conditionally immortalised podocytes in which we had convincingly knocked down the insulin and IGF1 receptors (Figure 2)
(3) You did not study the mice earlier to ascertain the developmental phenotype. We concede we did not do this but there was no significant proteinuria detected early in the mice so elected not to increase mouse numbers by studying them then (which we consider good practice for reduction, replacement and refinement). We suspect there would have been subtle changes in those mice that had significantly reduced simultaneous IR and IGF1R knockdown. It was precisely because of this that we generated a conditionally immortalised podocyte cell line with robust simultaneous knock-down of both receptors.
(4) You did not show significant insulin and IGF1 receptor knockdown in the conditionally immortalised cell line (reviewer states it was 50%). We clearly knocked both receptors down (insulin and IGF1R) in the podocyte line by >80% which was highly statistically significant (p<0.00001). Figure 2A. We agree this was crucial (and we made the cell line because of the variability in the mouse model).
The model as used may be more representative of a variable degree of podocyte depletion than an effect of impaired IR/IGF1R signaling. Therefore, although the phenotype may be ultimately attributable to the IR/IGF1R gene deletions the proteinuria and glomerulosclerotic phenotype itself was probably a consequence of defective podocyte development. Examining podocyte number, size, density and glomerular volume at earlier time points (4 weeks) would help to answer this question. Therefore, a more appropriate title would be "The insulin/IGF axis is critically important (for) normal podocyte development and deployment". In this context the effect of the knockdowns on splicing would make more sense.
Please see our response (above). We think our final conclusion that in the podocyte the insulin/IGF axis is important for spliceosome activity and control is valid. This is due to our findings (both total and phospho proteomics results) and considering recent other papers showing this axis can rapidly phosphorylate a variety of spliceosome proteins in different cell types (PMID:39939313 / PMID:32888406). All discussed in detail in the manuscript).
Cell culture studies. A cell line was generated using a temperature sensitive SV40 system that has been previously reported from this laboratory. A detailed analysis is provided to show that double knockout cells exhibited abnormal spliceosome activity. This forms the basis for the conclusion that "The insulin/IGF axis is critically important (for) controlling gene transcription in the podocyte". There are several concerns that weaken this conclusion.
(1) In the double knockdown cell culture system about 30% of cells were "lost" by 3 days and about 70% of cells were "lost" by 5days. The studies were done at the 3 day time point. It is not clear whether "lost" cells were in the process of dying, stress-induced detachment, or just growing more slowly than control due to reduced IR and IGF-1R signaling. These processes could have impacted splicing in a non-specific way independent of IR/IGF1R signaling itself.
(2) Can a single cell line derived from the double floxed mice be relied on to provide an unbiased picture of the effect of deleting IR and IGF-1R? Presumably, the transfection and selection process will select for cells that survive thereby including unknown biases, possibly related to spliceosome function. Is a single cell line adequate? These investigators have extensive experience with this type of analysis, but this question is not addressed in the discussion.
(3) To determine whether the effect is specific to reduced IR/IGFR signaling the deletion of IR and IGF-1R could be corrected by transfecting full length IR and IGF-1R cDNAs into the cells to restore normal IR/IGF1R signaling. If transfected cells with intact IR and IGF-1R expression and activity returns spliceosome activity to normal this would be evidence that receptors themselves play some role in spliceosome activity, as opposed to the downstream effect on growth limitation/stress on the cells.
(4) Other ways of testing whether the splicing effect is specifically due to reduced IR/IGF-1R signaling would be to (a) block IR and IGF1R receptors using available inhibitors, (b) remove or reduce insulin, IGF-1 and IGF-2 levels in the culture medium, (c) use low glucose and amino acid culture medium to slow growth rate independent of receptor function, (d) or block intra-cellular signaling via the IR and IGF-1R receptors through mTORC1 inhibition using rapamycin or other signaling targets.
(5) It would be useful to determine whether the cultured cells stressed in other ways (e.g. ischemia, toxins, etc.) also results in the same splicing abnormalities.
Point 1. 70% cell loss was observed at day 7 (not day 5). We found approximately 20% loss at day 3. We opted to go for this early date hypothesising the key detrimental processes would be clear then. This 3 day time point also ensures there has been enough time to allow for the expression of Cre recombinase, receptor gene excision and degradation of existing endogenous IR/IGF1R following lentiviral transduction. Interestingly we did not find a major “death or apoptosis” signal in our data then but agree it should be considered. We think this is a specific pathway as we have examined several other conditionally immortalised detrimental podocyte cell line previously using proteomics with a much more severe phenotype of cell death (E.g. podocyte GSK3 alpha/beta knockdown) and we detected NO spliceosome signal (PMID:30679422). Furthermore, there are now other podocyte proteomics “stress” studies that have been published in which there is proteinuria and significant cell loss / death that also do not show spliceosome dysfunction. These include studying the detailed proteosomal signature of podocytes stressed with Doxorubicin and Lipopolysaccharide endotoxin LPS in mice (PMID:32047005) and bradykinin stimulation of rat podocytes (PMID:32518694).
Point 2. Yes, we think it is valuable and reproducible. We generated a podocyte cell line from insulin receptor and IGF1 receptor homozygous floxed cells. Hence there is no selection bias in the cells when generating the line as both receptors are effectively intact. We then temporally “knocked down” the receptors with extrinsic lentiviral Cre.
Importantly we validated our cell line findings both back in the cells (with Western blotting) and in our transgenic receptor knockdown mice and found evidence of spliceosomal dysregulation (Figure 3E and 3F). Also as discussed above the spliceosome has been identified in other models in the insulin/IGF pathway.
Point 3. We don’t think the experiment of knocking down the receptors and then reconstituting them would prove this hypothesis. This is because if splicing abnormality was due to generalised cell dysfunction (which we do not think is the case in this situation) then putting the receptors back may simply restore cell health and the spliceosomal function (e.g. it does not prove it is via the receptors). Secondly, the process of transduction with multiple lentiviruses may be inherently stressful to the cell and there may be a high level of extrinsic receptor inserted which may also be confounding/detrimental. Finally, as discussed there are now several lines of evidence describing insulin / IGF signalling to spliceosomal proteins which we consider important (discussed in the paper in detail).
Point 4. We think modulating the receptors using the Cre-lox approach is the cleanest approach (with fewer off-target effects) to interrogate the insulin / IGF axis. It allows us to differentiate the cells by thermo-switching (which is crucial for this terminally differentiated cell) and then robustly knocking down both receptors simultaneously to investigate mechanism. We agree these supplementary approaches may give some extra information if their limitations (eg off target effects of inhibitors) are also taken into consideration.
Point 5. They do not. Please see response to point 1 above regarding GSK3, Doxorubicin, LPS and bradykinin challenge.
-

eLife Assessment
This valuable study used genetic and pharmacological manipulations of insulin/IGF signaling in renal glomerular podocytes to address the role of insulin/IGF axis in podocytes. Solid data are presented to demonstrate that co-inhibition of insulin/IGF signaling in podocytes led to aberrant splicing of mRNAs, which could contribute to the loss of podocytes in vitro and in vivo in mice. As it stands, the study lacks the assessment of developmental phenotype of podocytes in the mouse model.
[Editors' note: this paper was reviewed by Review Commons.]
-

Reviewer #1 (Public review):
Summary:
In this manuscript, the role of the insulin receptor and the insulin growth factor receptor was investigated in podocytes. Mice, where both receptors were deleted, developed glomerular dysfunction and developed proteinuria and glomerulrosclerosis over several months. Because of concerns about incomplete KO, the authors generated and studied podocyte cell lines where both receptors were deleted. Loss of both receptors was highly deleterious with greater than 50% cell death. To elucidate the mechanism of cell death, the authors performed global proteomics and found that spliceosome proteins were downregulated. They confirmed this directly by using long-read sequencing. These results suggest a novel role for insulin and IGF1R signaling in RNA splicing in podocytes.
This is primarily a descriptive study …
Reviewer #1 (Public review):
Summary:
In this manuscript, the role of the insulin receptor and the insulin growth factor receptor was investigated in podocytes. Mice, where both receptors were deleted, developed glomerular dysfunction and developed proteinuria and glomerulrosclerosis over several months. Because of concerns about incomplete KO, the authors generated and studied podocyte cell lines where both receptors were deleted. Loss of both receptors was highly deleterious with greater than 50% cell death. To elucidate the mechanism of cell death, the authors performed global proteomics and found that spliceosome proteins were downregulated. They confirmed this directly by using long-read sequencing. These results suggest a novel role for insulin and IGF1R signaling in RNA splicing in podocytes.
This is primarily a descriptive study and no technical concerns are raised. The mechanism of how insulin and IGF1 signaling regulates splicing is not directly addressed but implicates potentially the phosphorylation downstream of these receptors. In the revised manuscript, it is shown that the mouse KO is incomplete potentially explaining the slow onset of renal insufficiency. Direct measurement of GFR and serial serum creatinines might also enhance our understanding of progression of disease, proteinuria is a strong sign of renal injury. An attempt to rescue the phenotype by overexpression of SF3B4 would also be useful but may be masked by defects in other spliceosome genes. As insulin and IGF are regulators of metabolism, some assessment of metabolic parameters would be an optional add-on.
Significance:
With the GLP1 agonists providing renal protection, there is great interest in understanding the role of insulin and other incretins in kidney cell biology. It is already known that Insulin and IGFR signaling play important roles in other cells of the kidney. So, there is great interest in understanding these pathways in podocytes. The major advance is that these two pathways appear to have a role in RNA metabolism.
Comments on revised version:
I'm satisfied with the revised manuscript and the responses to my previous concerns.
-

Reviewer #2 (Public review):
Summary:
In this manuscript, submitted to Review Commons (journal agnostic), Coward and colleagues report on the role of insulin/IGF axis in podocyte gene transcription. They knocked out both the insulin and IGFR1 mice. Dual KO mice manifested a severe phenotype, with albuminuria, glomerulosclerosis, renal failure and death at 4-24 weeks.
Long read RNA sequencing was used to assess splicing events. Podocyte transcripts manifesting intron retention were identified. Dual knock-out podocytes manifested more transcripts with intron retention (18%) compared wild-type controls (18%), with an overlap between experiments of ~30%.
Transcript productivity was also assessed using FLAIR-mark-intron-retention software. Intron retention w seen in 18% of ciDKO podocyte transcripts compared to 14% of wild-type podocyte …
Reviewer #2 (Public review):
Summary:
In this manuscript, submitted to Review Commons (journal agnostic), Coward and colleagues report on the role of insulin/IGF axis in podocyte gene transcription. They knocked out both the insulin and IGFR1 mice. Dual KO mice manifested a severe phenotype, with albuminuria, glomerulosclerosis, renal failure and death at 4-24 weeks.
Long read RNA sequencing was used to assess splicing events. Podocyte transcripts manifesting intron retention were identified. Dual knock-out podocytes manifested more transcripts with intron retention (18%) compared wild-type controls (18%), with an overlap between experiments of ~30%.
Transcript productivity was also assessed using FLAIR-mark-intron-retention software. Intron retention w seen in 18% of ciDKO podocyte transcripts compared to 14% of wild-type podocyte transcripts (P=0.004), with an overlap between experiments of ~30% (indicating the variability of results with this method). Interestingly, ciDKO podocytes showed downregulation of proteins involved in spliceosome function and RNA processing, as suggested by LC/MS and confirmed by Western blot.
Pladienolide (a spliceosome inhibitor) was cytotoxic to HeLa cells and to mouse podocytes but no toxicity was seen in murine glomerular endothelial cells.
The manuscript is generally clear and well-written. Mouse work was approved in advance. The four figures are generally well-designed, bars/superimposed dot-plots.
Methods are generally well described.
Comments on revised version:
Coward and colleagues have done an excellent job of responding to all the reviewer comments.
-

Reviewer #4 (Public review):
Summary and background:
This report entitled "The insulin/IGF axis is critically important (for) controlling gene transcription in the podocyte" from Hurcombe et al is based on a mouse double knockdown of the IR and IGF1R and a parallel cultured mouse podocyte model. Insulin/IGF signaling system in mammals evolved as three gene reduplicated peptides (insulin, IGF-1, and IGF-2) and their two receptors IR and IGF1R that cross-react to variable extents with the peptides, are ubiquitously expressed, and signal through parallel pathways. The major downstream effect of insulin is to regulate glucose uptake and metabolism, while that of the IGF pathways is to regulate growth and cell cycling in part through mTORC1. The GH-IGF-1-IGF1R pathway regulates post-natal growth. IGF-2 signaling is thought to play a major …
Reviewer #4 (Public review):
Summary and background:
This report entitled "The insulin/IGF axis is critically important (for) controlling gene transcription in the podocyte" from Hurcombe et al is based on a mouse double knockdown of the IR and IGF1R and a parallel cultured mouse podocyte model. Insulin/IGF signaling system in mammals evolved as three gene reduplicated peptides (insulin, IGF-1, and IGF-2) and their two receptors IR and IGF1R that cross-react to variable extents with the peptides, are ubiquitously expressed, and signal through parallel pathways. The major downstream effect of insulin is to regulate glucose uptake and metabolism, while that of the IGF pathways is to regulate growth and cell cycling in part through mTORC1. The GH-IGF-1-IGF1R pathway regulates post-natal growth. IGF-2 signaling is thought to play a major role in regulating intrauterine growth and development, although IGF-2 is also present at high levels in post-natal life. Thus, one would anticipate that reducing IR/IGF1R signaling in any cell would slow growth and cell cycling by reducing growth factor and metabolic mTORC1-mediated and other processes including the splicing of RNA for protein synthesis.
Mouse IR/IGF1R double knockdown model:
A double knockdown mouse model was generated by interbreeding mice with different genetic backgrounds carrying floxed sites for IR and IGF-1R to produce mixed background offspring with both floxed IR and IGF-1R genes. These mice were crossed so that the podocin promoter driven-Cre (that comes on at about embryonic day 12 bas podocytes are developing) would delete IR and IGF-1R genes. Since podocin is believed to be an absolutely podocyte-specific protein, this podocin promoter this is predicted to specifically knock down the IR and IGF1R genes only in podocytes. The weight and growth of double KO offspring was not different from controls, but some proportion of the double knockdown mice subsequently developed proteinuria by 6 months and 20% died, although no specific data is provided to identify the cause of the deaths since eGFR was not decreased. Surviving mice were evaluated at 6 months of age. The efficacy of knockdown was not demonstrated in the mouse model itself, although a temperature-sensitive cell line developed from these double knockdown mice showed that expression of IR and IGF-1R proteins in the Cre-treated cell line were both reduced by about 50% (no statistical analysis of this result provided). In the knockout mice, proteinuria was significantly increased by 6 months, but not at earlier time points. Histologic analysis showed proteinaceous casts, glomerulosclerosis and interstitial fibrosis. Podocyte number was stated to be reduced by about 30% in double knockdown mice, although the method by which this was evaluated seems to have been by counting WT1 positive nuclei in glomerular cross-sections, an approach that is well-known not to be a reliable way of assessing true podocyte number. No information is provided about podocyte size, density or glomerular volume.
Comment: If IR/IGF1R deletion plays a significant role in normal podocyte function sufficient to cause proteinuria and glomerulosclerosis then the effect of reduced IR and IGF1R protein expression on podocyte function would have been expected to produce a phenotype before 6 months. A more likely scenario to explain the overall result is that deleting the IR and IGF1R genes at about embryonic day12 impacted podocyte development to a variable extent such that some mice developed fewer podocytes per glomerulus than other mice. As mice grow and their glomeruli and glomerular capillary area increases, those mice with fewer podocytes would not be able to completely cover the filtration surface with foot processes and would develop proteinuria and glomerulosclerosis. If reduced podocyte number per glomerulus is the proximate cause of the observed proteinuria, then modulation of the body and kidney growth rate by calorie restriction to slow growth (lower circulating IGF-1 levels) would be expected to be protective, while a high protein high calorie diet (higher circulating IGF-1 levels) or uni-nephrectomy to increase kidney growth rate would be expected to enhance proteinuria and glomerulosclerosis.
The model as used may be more representative of a variable degree of podocyte depletion than an effect of impaired IR/IGF1R signaling. Therefore, although the phenotype may be ultimately attributable to the IR/IGF1R gene deletions the proteinuria and glomerulosclerotic phenotype itself was probably a consequence of defective podocyte development. Examining podocyte number, size, density and glomerular volume at earlier time points (4 weeks) would help to answer this question. Therefore, a more appropriate title would be "The insulin/IGF axis is critically important (for) normal podocyte development and deployment". In this context the effect of the knockdowns on splicing would make more sense.
Cell culture studies. A cell line was generated using a temperature sensitive SV40 system that has been previously reported from this laboratory. A detailed analysis is provided to show that double knockout cells exhibited abnormal spliceosome activity. This forms the basis for the conclusion that "The insulin/IGF axis is critically important (for) controlling gene transcription in the podocyte". There are several concerns that weaken this conclusion.
(1) In the double knockdown cell culture system about 30% of cells were "lost" by 3 days and about 70% of cells were "lost" by 5days. The studies were done at the 3 day time point. It is not clear whether "lost" cells were in the process of dying, stress-induced detachment, or just growing more slowly than control due to reduced IR and IGF-1R signaling. These processes could have impacted splicing in a non-specific way independent of IR/IGF1R signaling itself.
(2) Can a single cell line derived from the double floxed mice be relied on to provide an unbiased picture of the effect of deleting IR and IGF-1R? Presumably, the transfection and selection process will select for cells that survive thereby including unknown biases, possibly related to spliceosome function. Is a single cell line adequate? These investigators have extensive experience with this type of analysis, but this question is not addressed in the discussion.
(3) To determine whether the effect is specific to reduced IR/IGFR signaling the deletion of IR and IGF-1R could be corrected by transfecting full length IR and IGF-1R cDNAs into the cells to restore normal IR/IGF1R signaling. If transfected cells with intact IR and IGF-1R expression and activity returns spliceosome activity to normal this would be evidence that receptors themselves play some role in spliceosome activity, as opposed to the downstream effect on growth limitation/stress on the cells.
(4) Other ways of testing whether the splicing effect is specifically due to reduced IR/IGF-1R signaling would be to (a) block IR and IGF1R receptors using available inhibitors, (b) remove or reduce insulin, IGF-1 and IGF-2 levels in the culture medium, (c) use low glucose and amino acid culture medium to slow growth rate independent of receptor function, (d) or block intra-cellular signaling via the IR and IGF-1R receptors through mTORC1 inhibition using rapamycin or other signaling targets.
(5) It would be useful to determine whether the cultured cells stressed in other ways (e.g. ischemia, toxins, etc.) also results in the same splicing abnormalities.
-

Author response:
The following is the authors’ response to the original reviews
Many thanks for your helpful and constructive comments for our work examining the effect of inhibiting both the insulin receptor (IR) and IGF1 receptor (IGF1R) in the podocyte. We are pleased to submit an updated manuscript addressing your concerns.
(1) A major concern was a lack of mechanistic insight into how deletion (or knock-down) of both receptors caused the spliceosomal phenotype (Reviewer 1 and Reviewer 3).
We now think this is due to the lack of a network of insulin/IGF phospho-signalling events to a variety of spliceosomal proteins and kinases. The reasons for this are as follows:
A. Since submitting our paper Turewicz et al have published a comprehensive phospho-proteomic paper examining the effects of 100nM insulin on human primary myotubes …
Author response:
The following is the authors’ response to the original reviews
Many thanks for your helpful and constructive comments for our work examining the effect of inhibiting both the insulin receptor (IR) and IGF1 receptor (IGF1R) in the podocyte. We are pleased to submit an updated manuscript addressing your concerns.
(1) A major concern was a lack of mechanistic insight into how deletion (or knock-down) of both receptors caused the spliceosomal phenotype (Reviewer 1 and Reviewer 3).
We now think this is due to the lack of a network of insulin/IGF phospho-signalling events to a variety of spliceosomal proteins and kinases. The reasons for this are as follows:
A. Since submitting our paper Turewicz et al have published a comprehensive phospho-proteomic paper examining the effects of 100nM insulin on human primary myotubes (DOI: 10.1038/s41467-025-56335-6). They discovered that multiple post-translational phosphorylation events occur in a variety of spliceosomal proteins at differing time points (1 minute to 60 minutes). Furthermore, they show that mRNA splicing is rapidly modified in response to insulin stimulation in their cells. This follows elegant work from Bastista et al who studied diabetic and non-diabetic iPSC derived human myositis and also detected a spliceosome phosphorylation signature (DOI: 10.1016/j.cmet.2020.08.007).
B. We have examined phospho-proteosome changes that occur in wild -type podocytes (expressing both the IR and IGF1R) compared to double (IR and IGF1R) knockout cells using phosho-proteomics. We have done this 3 days after inducing receptor knockdown, before major cell loss, and have stimulated the cells with either 10nM insulin or 100mg IGF1.
Interestingly, we detected several post-translational modifications (PTM) in our data set that are also present in Turewicz’s studies. Of note, 100nM insulin (as used by Turewicz) will signal through both the insulin and IGF1 receptor (and hybrid Insulin/IGF1 receptors) which is relevant to our studies.
Our work shows a cascade of phospho- signalling events affecting multiple components of the spliceosomal complex and evidence of kinase modulation (phosphorylation) (New Figure 7 and supplementary Figure 5). Also new results section in paper (lines 391-425 in track changes version). We acknowledge that we only studied a single time point after stimulation (10 minutes) and could have missed other PTM in the spliceosomal complex and other kinases. This is mentioned in our new limitations of study section (lines 595-606). This will be a focus of future work. We did not find major PTM differences when stimulating with either insulin or IGF1 in our studies and suspect that the doses of insulin (10nM) and IGF1 (100mg) used are still able to signal through cognate receptors.
Furthermore, we have examined the relative contributions of the insulin and IGF1 receptor in detail in the model (addressed in point 13 below).
(2) The phenotype of the mouse is only superficially addressed. The main issues are that the completeness of the mouse KO is never assessed nor is the completeness of the KO in cell lines. The absence of this data is a significant weakness. (Reviewer 1)
We apologise for not making this clear, but we did assess the level of receptor knockdown in both the animal and cell models. The in vivo model showed variable and non-complete levels of insulin receptor and IGF1 receptor podocyte knock down (shown in supplementary Figure 1C). This is why we made the in vitro floxed podocyte cell lines in which we could robustly knockdown both the IR and IGF1R. We show this using Western blotting (shown in Figure 2A). We agree that calling the models knockout is misleading and have changed all to knock down (KD) now.
(3) The mouse experiments would be improved if the serum creatinine’s were measured to provide some idea how severe the kidney injury is. (Reviewer 1)
There is variability in creatinine levels which is not uncommon in transgenic mouse models (probably partly due to variability in receptor knock down levels with cre-lox system). This is part of rationale of developing the robust double receptor knockout cell models where we robustly knocked out both receptors by >80%. We have added measured creatinine levels in a subset of mice in supplementary data (New Supplementary Figure 1E) and mention this in the text (lines 285-286). As some mice died we expect they may have developed acute kidney injury, but we did not serially measure the creatinine’s in every mouse over time. We could have assessed the GFR in a more sensitive way to look at differences. However, we consider the highly significant levels of albuminuria and histological damage observed in our models show a significant kidney phenotype.
(4) An attempt to rescue the phenotype by overexpression of SF3B4 would also be useful. If this didn't work, an explanation in the text would suffice. (Reviewer 1).
We did consider doing this but on reflection think it is very unlikely to rescue the phenotype as an array of different spliceosomal proteins quantitatively changed and were differentially phosphorylated / dephosphorylated throughout the complex (as we hope our revised work illustrates now). We think a single protein rescue is highly unlikely to work. We hope this is an appropriate explanation for this action. We have mentioned this in the text now in our discussion (lines 601-602).
(5) As insulin and IGF are regulators of metabolism, some assessment of metabolic parameters would be an optional add-on. (Reviewer 1).
Thank you for this suggestion. We did not extensively examine the metabolism of the mice however we did perform blood glucose measurement and weight which are included in the paper (Figure 1A and Figure 1B).
(6) The authors should caveat the cell experiments by discussing the ramifications of studying the 50% of the cells that survive vs the ones that died. (Reviewer 1).
We appreciate this and this was the rationale behind cells being studied after 3 days differentiation for total and phospho-proteomics before significant cell loss to avoid the issue of studying the 50% of cells that survive (which happened at 7 days). We have made this clearer in the manuscript. We also have added the data showing less cell death at 3 days in the cell model (New Supp Figure 2B).
(7) It would be helpful to say that tissue scoring was performed by an investigator masked to sample identity. (Reviewer 2)
We did this and have added to manuscript (line 113).
(8) Data are presented as mean/SEM. In general, mean/SD or median/IQR are preferred to allow the reader to evaluate the spread of the data. There may be exceptions where only SEM is reasonable. (Reviewer 2)
All graphs have now been changed to SD rather than SEM.
(9) It would be useful to for the reader to be told the number of over-lapping genes (with similar expression between mouse groups) and the results of a statistical test comparing WT and KO mice. The overlap of intron retention events between experimental repeats was about 30% in both knock-out podocytes. This seems low and I am curious to know whether this is typical for this method; a reference could be helpful. (Reviewer 2)
This is an excellent question. We had 30% overlap as the parameters used for analysis were very stringent. We suspect we could get more than 30% by being less stringent, which still be considered as similar events if requested. Our methods were based on FLAIR analysis (PMID: 32188845). We have added this reference to the manuscript (Line 242 & 680).
(10) With the GLP1 agonists providing renal protection, there is great interest in understanding the role of insulin and other incretins in kidney cell biology. It is already known that Insulin and IGFR signaling play important roles in other cells of the kidney. So, there is great interest in understanding these pathways in podocytes. The major advance is that these two pathways appear to have a role in RNA metabolism, the major limitations are the lack of information regarding the completeness of the KO's. If, for example, they can determine that in the mice, the KO is complete, that the GFR is relatively normal, then the phenotype they describe is relatively mild. (Reviewer 1)
Thank you. The receptor knock-out (KO) in the mice is highly unlikely to be complete (Please see comments above and Supplementary Figure 1C). There are many examples of “KO” animal models targeting other tissues showing that complete KO of these receptors seems difficult to achieve, particularly in reference to the IGF1 receptor. In the brain, which also contains terminally differentiated cells, barely 50% of IGF1R knockdown was achieved in the target cells (PMID:28595357). In ovarian granulosa cells (PMID:28407051) -several tissue specific drivers tried but couldn't achieve any better than 80%. The paper states that 10% of IGF1R is sufficient for function in these cells so they conclude that their knockdown animals are probably still responding to IGF1. Finally, in our recent IGF1R podocyte knockdown model we found Cre levels were important for excision of a single homozygous floxed gene (PMID: 38706850) hence we were not surprised that trying to excise two homozygous floxed genes (insulin receptor and IGF1 receptor) was challenging. This was the rationale for making the double receptor knockout cell lines to understand processes / biology in more detail. As stated earlier, we have changed our description of the mice and cell lines from knock-out to knock-down throughout the revised manuscript as this is more accurate.
(11) For the in vivo studies, the only information given is for mice at 24 weeks of age. There needs to be a full-time course of when the albuminuria was first seen and the rate of development. Also, GFR was not measured. Since the podocin-Cre utilized was not inducible, there should be a determination of whether there was a developmental defect in glomeruli or podocytes. Were there any differences in wither prenatal post-natal development or number of glomeruli? (Reviewer 3)
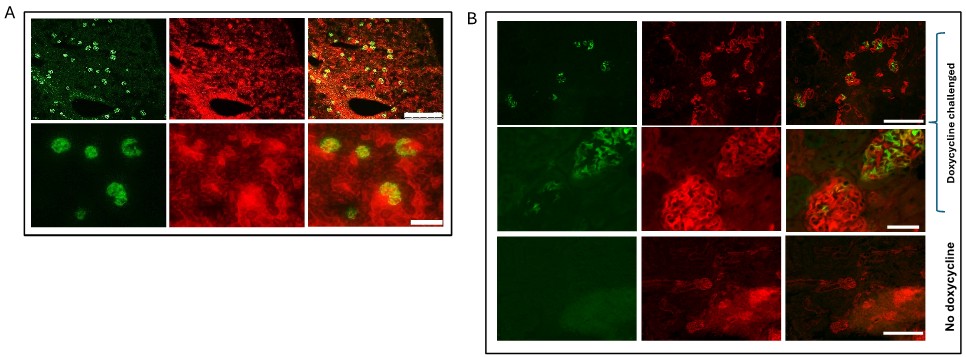
We have added further urinary Albumin:creatinine ratio (uACR) data at 12, 16 and 20 weeks to manuscript. We do not think there was a major developmental phenotype as albuminuria did not become significantly different until several months of age (new Supp Figure 1B). We did consider using a doxycycline inducible model but we know the excision efficiency is much less than the constitutive podocin-cre driven model Author response image 1. This would likely give a very mild (if any) phenotype when attempting to knockout both receptors and not reveal the biology adequately. We acknowledge the weaknesses of the animal model and this was the rationale for generating the cell models.
(12) Although the in vitro studies are of interest, there are no studies to determine if this is the underlying mechanism for the in vivo abnormalities seen in the mice. Cultured podocytes may not necessarily reflect what is occurring in podocytes in vivo. (Reviewer 3)
This is a good point. We have now immune-stained the DKD and WT mice for Sf3b4 (a spliceosomal change in our in vitro proteomics) and also find a significant reduction in this protein in podocytes of the DKD mice (New Figure 3F).
(13) Given that both receptors are deleted in the podocyte cell line, it is not clear if the spliceosome defect requires deletion of both receptors or if there is redundancy in the effect. The studies need to be repeated in podocyte cell lines with either IR or IGFR single deletions. (Reviewer 3)
We have now performed proteomics and phospho-proteomics in all 4 cell types (Wild-type, Insulin receptor knock down, IGF1R knockdown and double knockdown) at 3 days (New Figure 8 and supplementary Figure 6. Also new results section lines 425 to 450). This shows that both receptors contribute to the pathways (and hence there is a high level of compensation built into the system). For total proteins we detected that spliceosomal tri-snRNP was only reduced when both receptors were lacking but other proteins / pathways had an incremental effect of losing the insulin or IGF1 receptor. Likewise, the spliceosomal phospho-signaling events can go through either the insulin or igf1 receptors predominantly or through both. We think this reflects the complexity of this system and how evolutioatily it has developed in mammals to protect against its loss.
Finally in revision we have rewritten the discussion with a “limitations of the study” section and hopefully in an easier to read fashion for the readership.
Author response image 1.
(A) mT/mG reporter mouse crossed to constitutional podocin Cre heterozygous mouse. Illustrates podocyte specificity for Cre driver and excision Of reporter Figure shows GFP expression in Cre producing cells (top panel scale bar=250vm; bottom panel scale bar=50pm). Cre expression causes GFP to be switched on. (B) mT/mG reporter mouse crossed to podocin RtTA— tet-o-cre heterozygous mouse shows podocyte specificity for driver and approximately 60% excision. (top and bottom panels scale bar=250pm; middle panel scale bar=50pm). Doxycycline required for expression showing not leaky.
-

-

eLife Assessment
This study investigated the role of insulin receptor (IR) and insulin-like growth factor 1 receptor (IGF1R) in the renal glomerular podocytes by characterizing the mice with dual deletion of both receptors in vivo as well as the cultured murine podocytes with induced deletion of both receptors in vitro. The solid data presented in this paper demonstrated the critical requirement of both IR and IGF1R signaling in normal podocyte physiology in mice, albeit a more detailed characterization of the mouse model is desired. Interestingly, long-range sequencing revealed significant retention of introns in mRNAs, due to an altered spliceosome level resulted from the loss of IR and IGF1 signaling in cultured podocytes. This new finding suggests an essential role of IR and IGF1R signaling in regulating RNA metabolism in podocyte, …
eLife Assessment
This study investigated the role of insulin receptor (IR) and insulin-like growth factor 1 receptor (IGF1R) in the renal glomerular podocytes by characterizing the mice with dual deletion of both receptors in vivo as well as the cultured murine podocytes with induced deletion of both receptors in vitro. The solid data presented in this paper demonstrated the critical requirement of both IR and IGF1R signaling in normal podocyte physiology in mice, albeit a more detailed characterization of the mouse model is desired. Interestingly, long-range sequencing revealed significant retention of introns in mRNAs, due to an altered spliceosome level resulted from the loss of IR and IGF1 signaling in cultured podocytes. This new finding suggests an essential role of IR and IGF1R signaling in regulating RNA metabolism in podocyte, which provides useful information for the understanding of physiology and metabolism of podocytes. However, the underlying molecular mechanism for such a regulation is still unclear and awaits further studies.
[Editors' note: this paper was reviewed by Review Commons.]
-

Reviewer #1 (Public review):
Summary:
In this manuscript, the roles of the insulin receptor and the insulin growth factor receptor were investigated in podocytes. Mice in which both receptors were deleted developed glomerular dysfunction and developed proteinuria and glomerulrosclerosis over several months. Because of concerns about incomplete KO, the authors generated podocyte cell lines where both receptors were deleted. Loss of both receptors was highly deleterious with greater than 50% cell death. To elucidate the mechanism, the authors performed global proteomics and find that spliceosome proteins are down-regulated. They confirm this by using long-range sequencing. These results suggest a novel role for these pathways in podocytes.
This is primarily a descriptive study. The mechanism of how insulin and IGF1 signaling are linked to …
Reviewer #1 (Public review):
Summary:
In this manuscript, the roles of the insulin receptor and the insulin growth factor receptor were investigated in podocytes. Mice in which both receptors were deleted developed glomerular dysfunction and developed proteinuria and glomerulrosclerosis over several months. Because of concerns about incomplete KO, the authors generated podocyte cell lines where both receptors were deleted. Loss of both receptors was highly deleterious with greater than 50% cell death. To elucidate the mechanism, the authors performed global proteomics and find that spliceosome proteins are down-regulated. They confirm this by using long-range sequencing. These results suggest a novel role for these pathways in podocytes.
This is primarily a descriptive study. The mechanism of how insulin and IGF1 signaling are linked to the spliceosome is not addressed and the phenotype of the mice is only superficially explored. The main issues are that the completeness of the mouse KO is never assessed nor is the completeness of the KO in cell lines. The absence of this data is a significant weakness. The mouse experiments would be improved if the serum creatinines were measured to provide some idea about the severity of the kidney injury. An attempt to rescue the phenotype by overexpression of SF3B4 would also be useful. If this didn't rescue the phenotype, an explanation in the text would suffice. As insulin and IGF are regulators of metabolism, some assessment of metabolic parameters would be an optional add-on. Lastly, in the cell line experiments, the authors should discuss the caveats associated with studying the 50% of the cells that survive vs the ones that died.
Significance:
With the GLP1 agonists providing renal protection, there is great interest in understanding the role of insulin and other incretins in kidney cell biology. It is already known that Insulin and IGFR signaling play important roles in other cells of the kidney, therefore, there is great interest in understanding these pathways in podocytes. The major advance is that these two pathways appear to have a role in RNA metabolism, the major limitations are the lack of information regarding the completeness of the KO's. If, for example, they can determine that in the mice, the KO is complete, that the GFR is relatively normal, then the phenotype they describe is relatively mild.
Comments on revision plan:
I agree with the suggested experiments especially, the experiments to examine whether insulin/IGF1 signaling have effects on splicing proteins. An alternative experiment would be to ask whether rescue of IR or IGF1R would ameliorate the splicing effects.
-

Reviewer #2 (Public review):
Summary:
In this manuscript, submitted to Review Commons (journal agnostic), Coward and colleagues report on the role of insulin/IGF axis in podocyte gene transcription. They knocked out both the insulin and IGFR1 mice. Dual KO mice manifested a severe phenotype, with albuminuria, glomerulosclerosis, renal failure and death at 4-24 weeks.
Long read RNA sequencing was used to assess splicing events. Podocyte transcripts manifesting intron retention were identified. Dual knock-out podocytes manifested more transcripts with intron retention (18%) compared wild-type controls (18%), with an overlap between experiments of ~30%.
Transcript productivity was also assessed using FLAIR-mark-intron-retention software. Intron retention w seen in 18% of ciDKO podocyte transcripts compared to 14% of wild-type podocyte …
Reviewer #2 (Public review):
Summary:
In this manuscript, submitted to Review Commons (journal agnostic), Coward and colleagues report on the role of insulin/IGF axis in podocyte gene transcription. They knocked out both the insulin and IGFR1 mice. Dual KO mice manifested a severe phenotype, with albuminuria, glomerulosclerosis, renal failure and death at 4-24 weeks.
Long read RNA sequencing was used to assess splicing events. Podocyte transcripts manifesting intron retention were identified. Dual knock-out podocytes manifested more transcripts with intron retention (18%) compared wild-type controls (18%), with an overlap between experiments of ~30%.
Transcript productivity was also assessed using FLAIR-mark-intron-retention software. Intron retention w seen in 18% of ciDKO podocyte transcripts compared to 14% of wild-type podocyte transcripts (P=0.004), with an overlap between experiments of ~30% (indicating the variability of results with this method). Interestingly, ciDKO podocytes showed downregulation of proteins involved in spliceosome function and RNA processing, as suggested by LC/MS and confirmed by Western blot.
Pladienolide (a spliceosome inhibitor) was cytotoxic to HeLa cells and to mouse podocytes but no toxicity was seen in murine glomerular endothelial cells.
The manuscript is generally clear and well-written. Mouse work was approved in advance. The four figures are generally well-designed, with bars/superimposed dot-plots.
Methods are generally well described. It would be helpful to say that tissue scoring was performed by an investigator masked to sample identity.
Specific comments:
(1) Data are presented as mean/SEM. In general, mean/SD or median/IQR are preferred to allow the reader to evaluate the spread of the data. There may be exceptions where only SEM is reasonable.
(2) It would be useful to for the reader to be told the number of over-lapping genes (with similar expression between mouse groups) and the results of a statistical test comparing WT and KO mice. The overlap of intron retention events between experimental repeats was about 30% in both knock-out podocytes. This seems low and I am curious to know whether this is typical for typical for this method; a reference could be helpful.
(3) Please explain "adjusted p value of 0.01." It is not clear how was it adjusted. The number of differentially-expressed proteins between the two cell types was 4842.
Comments on revision plan:
The authors suggest additional experiments that should address my concerns and probably the other reviewers' concerns.
I encourage the authors to proceed with their proposed experiments and revisions.
-

Reviewer #3 (Public review):
Summary:
These investigators have previously shown important roles for either insulin receptor (IR) or insulin-like growth factor receptor (IGF1R) in glomerular podocyte function. They now have studied mice with deletion of both receptors and find significant podocyte dysfunction. They then made a podocyte cell line with inducible deletion of both receptors and find abnormalities in transcriptional efficiency with decreased expression of spliceosome proteins and increased transcripts with impaired splicing or premature termination.
The studies appear to be performed well and the manuscript is clearly written.
There are a number of potential issues and questions with these studies.
(1) For the in vivo studies, the only information given is for mice at 24 weeks of age. There needs to be a full time course of …
Reviewer #3 (Public review):
Summary:
These investigators have previously shown important roles for either insulin receptor (IR) or insulin-like growth factor receptor (IGF1R) in glomerular podocyte function. They now have studied mice with deletion of both receptors and find significant podocyte dysfunction. They then made a podocyte cell line with inducible deletion of both receptors and find abnormalities in transcriptional efficiency with decreased expression of spliceosome proteins and increased transcripts with impaired splicing or premature termination.
The studies appear to be performed well and the manuscript is clearly written.
There are a number of potential issues and questions with these studies.
(1) For the in vivo studies, the only information given is for mice at 24 weeks of age. There needs to be a full time course of when the albuminuria was first seen and the rate of development. Also, GFR was not measured. Since the podocin-Cre utilized was not inducible, there should be a determination of whether there was a developmental defect in glomeruli or podocytes. Were there any differences in wither prenatal post natal development or number of glomeruli?
(2) Although the in vitro studies are of interest, there are no studies to determine if this is the underlying mechanism for the in vivo abnormalities seen in the mice. Cultured podocytes may not necessarily reflect what is occurring in podocytes in vivo.
(3) Given that both receptors are deleted in the podocyte cell line, it is not clear if the spliceosome defect requires deletion of both receptors or if there is redundancy in the effect. The studies need to be repeated in podocyte cell lines with either IR or IGFR single deletions.
(4) There are no studies investigating signaling mechanisms mediating the spliceosome abnormalities.
Comments on revision plan:
I do not have any changes from my prior review. I applaud the authors for developing a plan to address the questions and concerns raised in my prior review.
-

Author response:
Evidence reducibility and clarity
Reviewer 1:
In this manuscript, the role of the insulin receptor and the insulin growth factor receptor was investigated in podocytes. Mice, were both receptors were deleted, developed glomerular dysfunction and developed proteinuria and glomerulosclerosis over several months. Because of concerns about incomplete KO, the authors generated podocyte cell lines where both receptors were deleted. Loss of both receptors was highly deleterious with greater than 50% cell death. To elucidate the mechanism, the authors performed global proteomics and find that spliceosome proteins are downregulated. They confirm this by using long-range sequencing. These results suggest a novel role for these pathways in podocytes.
Thank you
This is primarily a descriptive study and no technical concerns are …
Author response:
Evidence reducibility and clarity
Reviewer 1:
In this manuscript, the role of the insulin receptor and the insulin growth factor receptor was investigated in podocytes. Mice, were both receptors were deleted, developed glomerular dysfunction and developed proteinuria and glomerulosclerosis over several months. Because of concerns about incomplete KO, the authors generated podocyte cell lines where both receptors were deleted. Loss of both receptors was highly deleterious with greater than 50% cell death. To elucidate the mechanism, the authors performed global proteomics and find that spliceosome proteins are downregulated. They confirm this by using long-range sequencing. These results suggest a novel role for these pathways in podocytes.
Thank you
This is primarily a descriptive study and no technical concerns are raised. The mechanism of how insulin and IGF1 signaling are linked to the spiceosome is not addresed.
We do not think the paper is descriptive as we used non-biased phospho and total proteomics in the DKO cells to uncover the alterations in the spliceosome (that have not been previously described) that were detrimental. However, we are happy to look further into the underlying mechanism.
We would propose:
(1) Stimulating/inhibiting insulin/IGF signalling pathways in the Wild-type and DKO knockout cells and check expression levels and/or phosphorylation status of splice factors (including those in Figure 3E) and those revealed by phospho-proteomic data; a variety of inhibitors of insulin/IGF1 pathways could also be used along the pathways that are shown in Fig 2.
(2) Looking at the RNaseq data bioinformatically in more detail – the introns/exons that move up or down are targets of the splice factors involved; most splice factors binding sequences are known, so it should be possible to ask bioinformatically – from the sequences around the splice sites of the exons and introns that move in the DKO, which splice factors binding sites are seen most frequently? To uncover splice factors/RNA-binding proteins (RBPs) that are involved in the insulin signaling we will use a software named MATT which was specifically designed to look for RNA-binding motifs (PMID 30010778). In brief, using the long-sequencing data, we will test 250 nt sequences flanking the splice sites of all regulated splicing events (intronic and exonic) against all RNA- binding proteins in the CISBP-RNA database (PMID 23846655) using MATT. This will result in a list of RBPs potentially involved in the insulin signaling. We will validate these by activating insulin signaling (similar to Figures 2 B,C) and probe whether the RBPs are activated (e.g. phosphorylated or change in expression) or we will manipulate expression of the candidate RBPs and measure how they affect the insulin signaling.
(3) Examining the phospho and total proteomic data for IGF1R and Insulin receptor knockout alone podocytes (which we have already generated) and analysing these in more detail and include this data set to elucidate the relative importance of both receptors to spliceosome function.
The phenotype of the mouse is only superficially addressed. The main issues are that the completeness of the mouse KO is never assessed nor is the completeness of the KO in cell lines. The absence of this data is a significant weakness.
We apologise for not making clear but we did assess the level of receptor knockdown in the animal and cell models. The in vivo model showed variable and non-complete levels of insulin receptor and IGF1 receptor podocyte knock down (shown in supplementary figure 1B). This is why we made the in vitro floxed podocyte cell lines in which we could robustly knockdown both the insulin receptor and IGF1 receptor (shown in Figure 2A)
The mouse experiments would be improved if the serum creatinines were measured to provide some idea how severe the kidney injury is.
We can address this:
We have further urinary Albumin:creatinine ratio (uACR) data at 12, 16 and 20 weeks. We also have more blood tests of renal function that can be added. There is variability in creatinine levels which is not uncommon in transgenic mouse models (probably partly due to variability in receptor knock down with cre-lox system). This is part of rationale of developing the robust double receptor knockout cell models where we knocked out both receptors by >80%.
An attempt to rescue the phenotype by overexpression of SF3B4 would also be useful. If this didn't work, an explanation in the text would suffice.
We would consider over express SF3BF4 in the Wild type and DKO cells and assess the effects on spliceosome if deemed necessary. However, we think it is unlikely to rescue the phenotype as so many other spliceosome components are downregulated in the DKO cells.
As insulin and IGF are regulators of metabolism, some assessment of metabolic parameters would be an optional add-on.
We have some detail on this and can add to the manuscript. However it is not extensive as not a major driver of this work.
Lastly, the authors should caveat the cell experiments by discussing the ramifications of studying the 50% of the cells that survive vs the ones that died.
Thank you, we appreciate this and this was the rationale behind cells being studied after 2 days differentiation before significant cell loss in order to avoid the issue of studying the 50% of cells that survive.
Reviewer 2:
In this manuscript, submitted to Review Commons (journal agnostic), Coward and colleagues report on the role of insulin/IGF axis in podocyte gene transcription. They knocked out both the insulin and IGFR1 mice. Dual KO mice manifested a severe phenotype, with albuminuria, glomerulosclerosis, renal failure and death at 4-24 weeks.
Long read RNA sequencing was used to assess splicing events. Podocyte transcripts manifesting intron retention were identified. Dual knock-out podocytes manifested more transcripts with intron retention (18%) compared wild-type controls (18%), with an overlap between experiments of ~30%.
Transcript productivity was also assessed using FLAIR-mark-intron-retention software. Intron retention w seen in 18% of ciDKO podocyte transcripts compared to 14% of wild-type podocyte transcripts (P=0.004), with an overlap between experiments of ~30% (indicating the variability of results with this method). Interestingly, ciDKO podocytes showed downregulation of proteins involved in spliceosome function and RNA processing, as suggested by LC/MS and confirmed by Western blot.
Pladienolide (a spliceosome inhibitor) was cytotoxic to HeLa cells and to mouse podocytes but no toxicity was seen in murine glomerular endothelial cells.
Specific comments.The manuscript is generally clear and well-written. Mouse work was approved in advance. The six figures are generally well-designed, bars/superimposed dot-plots.
Thank you
Evaluation.
Methods are generally well described. It would be helpful to say that tissue scoring was performed by an investigator masked to sample identity.
We did this and will add this information to the methods/figure legend.
Specific comments.
(1) Data are presented as mean/SEM. In general, mean/SD or median/IQR are preferred to allow the reader to evaluate the spread of the data. There may be exceptions where only SEM is reasonable.
Graphs can be changed to SD rather than SEM.
(2) It would be useful to for the reader to be told the number of over-lapping genes (with similar expression between mouse groups) and the results of a statistical test comparing WT and KO mice. The overlap of intron retention events between experimental repeats was about 30% in both knock-out podocytes. This seems low and I am curious to know whether this is typical for typical for this method; a reference could be helpful.
This is an excellent question. We had 30% overlap as the parameters used for analysis were very stringent. We suspect we could get more than 30% by being less stringent, which still be considered as similar events if requested. Our methods were based on FLAIR analysis (PMID: 32188845)
(3) Please explain "adjusted p value of 0.01." It is not clear how was it adjusted. The number of differentially-expressed proteins between the two cell types was 4842.
We used the Benjamini-Hochberg method to adjust our data. We think the reviewer is referring to the transcriptomic data and not the proteomic data.
Minor comments
Page numbers in the text would help the reviewer communicate more effectively with the author.
We will do this
Reviewer 3:
These investigators have previously shown important roles for either insulin receptor (IR) or insulin-like growth factor receptor (IGF1R) in glomerular podocyte function. They now have studied mice with deletion of both receptors and find significant podocyte dysfunction. They then made a podocyte cell line with inducible deletion of both receptors and find abnormalities in transcriptional efficiency with decreased expression of spliceosome proteins and increased transcripts with impaired splicing or premature termination.
The studies appear to be performed well and the manuscript is clearly written.
Thank you
Referees cross-commenting
I am in agreement with Reviewer 1 that the studies are overly descriptive and do not provide sufficient mechanism and the lack of more investigation of the in vivo model is a significant weakness.
Please see our responses to reviewer 1 above.
Significance
Reviewer 1:
With the GLP1 agonists providing renal protection, there is great interest in understanding the role of insulin and other incretins in kidney cell biology. It is already known that Insulin and IGFR signaling play important roles in other cells of the kidney. So, there is great interest in understanding these pathways in podocytes. The major advance is that these two pathways appear to have a role in RNA metabolism, the major limitations are the lack of information regarding the completeness of the KO's. If, for example, they can determine that in the mice, the KO is complete, that the GFR is relatively normal, then the phenotype they describe is relatively mild.
Thank you. The receptor KO in the mice is unlikely to be complete (Please see comments above and Supplementary Figure 1b). There are many examples of KO models targeting other tissues showing that complete KO of these receptors seems difficult to achieve , particularly in reference to the IGF1 receptor. In the brain (which is also terminally differentiated cells PMID:28595357 (barely 50% iof IGF1R knockdown was achieved in the target cells). Ovarian granulosa cells PMID:28407051 -several tissue specific drivers tried but couldn't achieve any better than 80%. The paper states that 10% of IGF1R is sufficient for function in these cells so they conclude that their knockdown animals are probably still responding to IGF1. Finally, in our recent IGF1R podocyte knockdown model we found Cre levels were important for excision of a single floxed gene (PMID: 38706850) hence we were not surprised that trying to excise two floxed genes (insulin receptor and IGF1 receptor) was challenging. This is the rationale for making the double receptor knockout cell lines to understand process / biology in more detail.
Reviewer 2:
The manuscript is generally clear and well-written. Mouse work was approved in advance. The figures are generally well-designed, bars/superimposed dot-plots.
Evaluation.
Methods are generally well described. It would be helpful to say that tissue scoring was performed by an investigator masked to sample identity.
Thank you we will do this.
Reviewer 3:
There are a number of potential issues and questions with these studies.
(1) For the in vivo studies, the only information given is for mice at 24 weeks of age. There needs to be a full time course of when the albuminuria was first seen and the rate of development. Also, GFR was not measured. Since the podocin-Cre utilized was not inducible, there should be a determination of whether there was a developmental defect in glomeruli or podocytes. Were there any differences in wither prenatal post natal development or number of glomeruli?
Thank you we will add in further phenotyping data. We do not think there was a major developmental phenotype as albuminuria did not become significantly different until several months of age. We could have used a doxycycline inducible model but we know the excision efficiency is much less than the podocin-cre driven model SUPP FIGURE 1. This would likely give a very mild (if any) phenotype and not reveal the biology adequately.
(2) Although the in vitro studies are of interest, there are no studies to determine if this is the underlying mechanism for the in vivo abnormalities seen in the mice. Cultured podocytes may not necessarily reflect what is occurring in podocytes in vivo.
Thank you for this we are happy to employ Immunohistochemistry (IHC) and immunofluorescence (IF) using spliceosome antibodies on tissue sections from DKO and control mice to examine spliceosome changes. However, as the DKO results in podocyte loss, there may not be that many DKO podocytes still present in the tissue sections. This will be taken into consideration.
(3) Given that both receptors are deleted in the podocyte cell line, it is not clear if the spliceosome defect requires deletion of both receptors or if there is redundancy in the effect. The studies need to be repeated in podocyte cell lines with either IR or IGFR single deletions.
Thank you. We have full total and phospho-proteomic data sets from single insulin receptor and IGF1 receptor knockout cell lines that we will investigate for this point.
(4) There are not studies investigating signaling mechanisms mediating the spliceosome abnormalities.
Thank you as outlined as above to reviewer 1 point 1 we are very happy to investigate insulin / IGF signalling pathways in more detail.
-

-