Mettl5 coordinates protein production and degradation of PERIOD to regulate sleep in Drosophila
Curation statements for this article:-
Curated by eLife
eLife Assessment
The authors present useful findings demonstrating that the RNA modification enzyme Mettl5 regulates sleep in Drosophila. Through transcriptome- and proteome-wide analyses, the authors identified downstream targets affected in heterozygous mutants and proposed that Mettl5 regulates the translation and degradation of clock genes to maintain normal sleep function. Through additional analyses, the authors provided solid evidence that Mettl5 regulates translation and degradation of clock genes to maintain normal sleep cycle. The mechanistic details of Mettl5 is unclear and requires further support.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
Sleep plays a critical role in animal physiology, primarily governed by the brain, and its disruption is prevalent in various brain disorders. Mettl5 is associated with intellectual disability (ID), which often includes sleep disturbances. However, the mechanism underlying these sleep disruptions in ID remains poorly understood. In this study, we investigated the sleep phenotypes resulting from Drosophila Mettl5 mutations. Rescue experiments revealed that Mettl5 functions predominantly within neurons and glia marked by Mettl5-Gal4 to regulate sleep. Previous work established that Mettl5 forms a complex with Trmt112 to influence rRNA methylation. Notably, a mutation in Trmt112 mutation recapitulated these sleep disturbances, implicating translational regulation by the Mettl5/Trmt112 complex. Subsequent RNA-seq and Ribo-seq analyses of Mettl51bp mutants uncovered downstream effects, including altered expression of proteasome components and clock genes. Rescue experiments confirmed that the net increase in PERIOD protein underlies the sleep phenotype. This study illuminates the interplay between ribosome function, clock genes, and the proteasome in sleep regulation, highlighting the integrated roles of protein synthesis and degradation. These findings could potentially provide an example for in vivo study of rRNA methylation function, expand our understanding of protein homeostasis in sleep and offer insights into the sleep phenotypes associated with ID.
Article activity feed
-

eLife Assessment
The authors present useful findings demonstrating that the RNA modification enzyme Mettl5 regulates sleep in Drosophila. Through transcriptome- and proteome-wide analyses, the authors identified downstream targets affected in heterozygous mutants and proposed that Mettl5 regulates the translation and degradation of clock genes to maintain normal sleep function. Through additional analyses, the authors provided solid evidence that Mettl5 regulates translation and degradation of clock genes to maintain normal sleep cycle. The mechanistic details of Mettl5 is unclear and requires further support.
-

Reviewer #1 (Public review):
Here, the authors attempted to test whether the function of Mettl5 in sleep regulation was conserved in drosophila, and if so, by which molecular mechanisms. To do so they performed sleep analysis, as well as RNA-seq and ribo-seq in order to identify the downstream targets. They found that the loss of one copy of Mettl5 affects sleep, and that its catalytic activity is important for this function. Transcriptional and proteomic analyses show that multiple pathways were altered, including the clock signaling pathway and the proteasome. Based on these changes the authors propose that Mettl5 modulate sleep through regulation of the clock genes, both at the level of their production and degradation, possibly by altering the usage of Aspartate codon.
Comments on revised version:
The authors satisfactorily …
Reviewer #1 (Public review):
Here, the authors attempted to test whether the function of Mettl5 in sleep regulation was conserved in drosophila, and if so, by which molecular mechanisms. To do so they performed sleep analysis, as well as RNA-seq and ribo-seq in order to identify the downstream targets. They found that the loss of one copy of Mettl5 affects sleep, and that its catalytic activity is important for this function. Transcriptional and proteomic analyses show that multiple pathways were altered, including the clock signaling pathway and the proteasome. Based on these changes the authors propose that Mettl5 modulate sleep through regulation of the clock genes, both at the level of their production and degradation, possibly by altering the usage of Aspartate codon.
Comments on revised version:
The authors satisfactorily addressed my comments, even though the precise mechanism by which Mettl5 regulates translation of clock genes remains to be firmly demonstrated.
-

Reviewer #3 (Public review):
Xiaoyu Wu and colleagues examined a potential role in sleep of a Drosophila ribosomal RNA methyltransferase, mettl5. Based on sleep defects reported in CRISPR generated mutants, the authors performed both RNA-seq and Ribo-seq analyses of head tissue from mutants and compared to control animals collected at the same time point. A major conclusion was that the mutant showed altered expression of circadian clock genes, and that the altered expression of the period gene in particular accounted for the sleep defect reported in the mettl5 mutant. In this revision, the authors have added a more thorough analysis of clock gene expression and show that PER protein levels are increased relative to wild type animals a specific times of day, indicating increased stability of the protein. Given that PER inhibits its own …
Reviewer #3 (Public review):
Xiaoyu Wu and colleagues examined a potential role in sleep of a Drosophila ribosomal RNA methyltransferase, mettl5. Based on sleep defects reported in CRISPR generated mutants, the authors performed both RNA-seq and Ribo-seq analyses of head tissue from mutants and compared to control animals collected at the same time point. A major conclusion was that the mutant showed altered expression of circadian clock genes, and that the altered expression of the period gene in particular accounted for the sleep defect reported in the mettl5 mutant. In this revision, the authors have added a more thorough analysis of clock gene expression and show that PER protein levels are increased relative to wild type animals a specific times of day, indicating increased stability of the protein. Given that PER inhibits its own transcription, the per RNA is low in the mutants. Efforts toward a more detailed understanding of how clock gene expression was altered in the mutants, as well as other clarification of sleep phenotypes throughout is appreciated. As noted above, a strength of this work is its relevance to a human developmental disorder as well as the transcriptomic and ribosomal profiling of the mutant. However, there still remain some minor weaknesses in the manuscript. This reviewer is not in agreement with the interpretation of the epigenetic experiments. Specifically, co-expression of Clk[jrk] or per[01] with the mettl5 mutant recovered the nighttime sleep phenotype, but was additive to the daytime sleep phenotype such that double mutants showed higher sleep. This effect should be acknowledged and discussed. Overall, this is an interesting paper that indicates a molecular link between mettl5 and the circadian clock in regulation of sleep.
-

Author response:
The following is the authors’ response to the original reviews.
Public Reviews:
Reviewer #1 (Public review):
Summary:
Here the authors attempted to test whether the function of Mettl5 in sleep regulation was conserved in drosophila, and if so, by which molecular mechanisms. To do so they performed sleep analysis, as well as RNA-seq and ribo-seq in order to identify the downstream targets. They found that the loss of one copy of Mettl5 affects sleep and that its catalytic activity is important for this function. Transcriptional and proteomic analyses show that multiple pathways were altered, including the clock signaling pathway and the proteasome. Based on these changes the authors propose that Mettl5 modulate sleep through regulation of the clock genes, both at the level of their production and degradation.
Strengths:
Author response:
The following is the authors’ response to the original reviews.
Public Reviews:
Reviewer #1 (Public review):
Summary:
Here the authors attempted to test whether the function of Mettl5 in sleep regulation was conserved in drosophila, and if so, by which molecular mechanisms. To do so they performed sleep analysis, as well as RNA-seq and ribo-seq in order to identify the downstream targets. They found that the loss of one copy of Mettl5 affects sleep and that its catalytic activity is important for this function. Transcriptional and proteomic analyses show that multiple pathways were altered, including the clock signaling pathway and the proteasome. Based on these changes the authors propose that Mettl5 modulate sleep through regulation of the clock genes, both at the level of their production and degradation.
Strengths:
The phenotypical consequence of the loss of one copy of Mettl5 on sleep function is clear and well-documented.
Weaknesses:
The imaging and molecular parts are less convincing.
- The colocalization of Mettl5 with glial and neuronal cells is not very clear
We truly appreciate your suggestion. We repeated the staining experiments. To ensure better results, we tried another antibody of ELAV (mouse) and optimized the experimental conditions. This result has been included in the Figure S1 of the revised version.
- The section on gene ontology analysis is long and confusing
The session is revised for clarity. To get a better flow of logic, we deleted the paragraph which describing the details of Figure S6.
- Among all the pathways affected the focus on proteosome sounds like cherry picking. And there is no experiment demonstrating its impact in the Mettl5 phenotype
Thank you for the comments. The changes of period oppositely at transcriptional versus translational levels puzzled us a while until we found the ubiquitin pathway components changes. The regulation of Period protein degradation by ubiquitin-proteasome pathway has been well documented (Grima et al., 2002; Ko et al., 2002; Chiu et al., 2008). In addition, previous reports indicated that N6 methyladenosine (m6A) regulates ubiquitin proteasome pathway in skeletal muscle physiology (Sun et al., 2023). This information has been included in the revised manuscript in the last paragraph under the title: Mettl5 regulates the clock gene regulatory loop.
Indeed, we haven’t found a proper way to manipulate proteasome levels in genetic tests. Proteasome is a large protein complex which is composed of many subunits. Enhancing the its activity by overexpressing its components was not applicable. Moreover, proteasome has important function during many biological processed. Disrupting its function by simply MG132 treatment which we tried results in lots of side effects.
In this study, we also noticed the codon usage alteration caused by mettl5 mutant. Please refer to the answers to the following question for details. Previous reports also found the regulation of mettl5 on translation in other systems (Rong et al, 2020; Peng et al., 2022). Based on these analyses, it is possible that both the regulation on translation and protein degradation contributed the period protein upregulation found in mettl5 mutant. This idea has been included in the Discussion session of the revised manuscript.
References
Sun J, Zhou H, Chen Z, et al. Altered m6A RNA methylation governs denervation-induced muscle atrophy by regulating ubiquitin proteasome pathway. J Transl Med. 2023;21(1):845. Published 2023 Nov 23. doi:10.1186/s12967-023-04694-3
Grima, B. et al. The F-box protein slimb controls the levels of clock proteins period and timeless. Nature 420, 178–182 (2002).
Ko, H. W., Jiang, J. & Edery, I. Role for Slimb in the degradation of Drosophila period protein phosphorylated by doubletime. Nature 420, 673–678 (2002).
Chiu, J. C., Vanselow, J. T., Kramer, A. & Edery, I. The phosphooccupancy of an atypical SLIMB-binding site on PERIOD that is phosphorylated by DOUBLETIME controls the pace of the clock. Genes Dev. 22, 1758–1772 (2008).
- The ribo seq shows some changes at the level of translation efficiency but there is no connection with the Mettl5 phenotypes. In other words, how the increased usage of some codons impact clock signalling. Are the genes enriched for these codons?
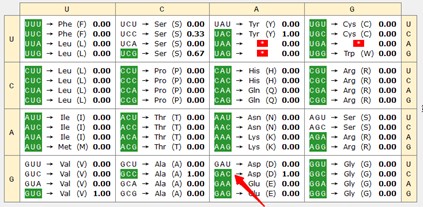
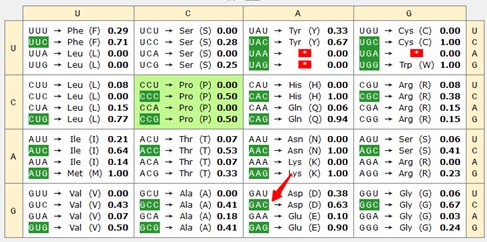
Thank you for raising this point. In our analysis, we observed an increased usage of the codons for Asp in the Mettl5 mutant. Prior work has reported a possible connection between codon usage and per protein activity. In the report, a per version with optimized codon cannot rescue circadian rhythmicity caused by per mutant, in contrast to WT version (Fu J et al. 2016). Further study indicated that dPER protein levels were also elevated in the mutant flies, suggesting a role for codon optimization in enhancing dPER expression (Figure 2B in Fu J et al. 2016). Consistent with this, we analyzed the region of codon optimization in Fu J et al. 2016. The result indicated that that GAC has a relatively high usage rate in these regions (indicated in the following two Author response image charts by the red arrow), suggesting that the Mettl5 mutation may influence per protein accumulation through altered GAC usage. Further experiments are needed to confirm this possibility. We included these details in the second last paragraph of the Discussion session.
Author response image 1.
15-21
SDSAYSN
Author response image 2.
43-316
SSGSSGYGGKPSTQASSSDMIIKRNKEKSRKKKKPKCIALATATTVSLEGTEESPLPANGGCEKVLQELQDTQQLGEPLVVTETQLSEQLLETEQNEDQNKSEQLAQFPLPTPIVTTLSPGIGPGHDCVGGASGGAVAGGCSVVGAGTDKTSELIPGKLESAGTKPSQERPKEESFCCVISMHDGIVLYTTPSISDVLGFPRDMWLGRSFIDFVHHKDRATFASQITTGIPIAESRGCMPKDARSTFCVMLRRYRGLNSGGFGVIGRAVNYEPF
Fu J, Murphy KA, Zhou M, Li YH, Lam VH, Tabuloc CA, Chiu JC, Liu Y. Codon usage affects the structure and function of the Drosophila circadian clock protein PERIOD. Genes Dev. 2016 Aug 1;30(15):1761-75.
- A few papers already demonstrated the role of Mettl5 in translation, even at the structural level (Rong et al, Cell reports 2020) and this was not commented by the authors. In Peng et al, 2022 the authors show that the m6A bridges the 18S rRNA with RPL24. Is this conserved in Drosophila?
Thanks for the reminder. We discussed and cited these papers in the revised version.
Rong B, Zhang Q, Wan J, et al. Ribosome 18S m6A Methyltransferase METTL5 Promotes Translation Initiation and Breast Cancer Cell Growth. Cell Rep. 2020;33(12):108544. doi:10.1016/j.celrep.2020.108544
Peng H, Chen B, Wei W, et al. N6-methyladenosine (m6A) in 18S rRNA promotes fatty acid metabolism and oncogenic transformation. Nat Metab. 2022;4(8):1041-1054. doi:10.1038/s42255-022-00622-9
- The text will require strong editing and the authors should check and review extensively for improvements to the use of English.
Thanks. The text of the paper are thoroughly revised.
Conclusion
Despite the effort to identify the underlying molecular defects following the loss of Mettl5 the authors felt short in doing so. Some of the results are over-interpreted and more experiments will be needed to understand how Mettl5 controls the translation of its targets. References to previous works was poorly commented.
Thanks for your suggestion. We have incorporated the references mentioned above. However, our efforts have thus far fallen short of elucidating a precise picture of METTL5's functional mechanism. To address this, the limitations of the current study have been discussed more thoroughly in the revised main text.
Reviewer #2 (Public review):
Summary:
The authors define the m6A methyltransferase Mettl5 as a novel sleep-regulatory gene that contributes to specific aspects of Drosophila sleep behaviors (i.e., sleep drive and arousal at early night; sleep homeostasis) and propose the possible implication of Mettl5-dependent clocks in this process. The model was primarily based on the assessment of sleep changes upon genetic/transgenic manipulations of Mettl5 expression (including CRISPR-deletion allele); differentially expressed genes between wild-type vs. Mettl5 mutant; and interaction effects of Mettl5 and clock genes on sleep. These findings exemplify how a subclass of m6A modifications (i.e., Mettl5-dependent m6A) and possible epi-transcriptomic control of gene expression could impact animal behaviors.
Strengths:
Comprehensive DEG analyses between control and Mettl5 mutant flies reveal the landscape of Mettl5-dependent gene regulation at both transcriptome and translatome levels. The molecular/genetic features underlying Mettl5-dependent gene expression may provide important clues to molecular substrates for circadian clocks, sleep, and other physiology relevant to Mettl5 function in Drosophila.
Weaknesses:
While these findings indicate the potential implication of Mettl5-dependent gene regulation in circadian clocks and sleep, several key data require substantial improvement and rigor of experimental design and data interpretation for fair conclusions. Weaknesses of this study and possible complications in the original observations include but are not limited to:
(1) Genetic backgrounds in Mettl5 mutants: the heterozygosity of Mettl5 deletion causes sleep suppression at early night and long-period rhythms in circadian behaviors. The transgenic rescue using Gal4/UAS may support the specificity of the Mettl5 effects on sleep. However, it does not necessarily exclude the possibility that the Mettl5 deletion stocks somehow acquired long-period mutation allelic to other clock genes. Additional genetic/transgenic models of Mettl5 (e.g., homozygous or trans-heterozygous mutants of independent Mettl5 alleles; Mettl5 RNAi etc.) can address the background issue and determine 1) whether sleep suppression tightly correlates with long-period rhythms in Mettl5 mutants; and 2) whether Mettl5 effects are actually mapped to circadian pacemaker neurons (e.g., PDF- or tim-positive neurons) to affect circadian behaviors, clock gene expression, and synaptic plasticity in a cell-autonomous manner and thereby regulate sleep. Unfortunately, most experiments in the current study rely on a single genetic model (i.e., Mettl5 heterozygous mutant).
We believe that the multiple rescue experiments presented in Figure 1H-L and Figure 2H-L have effectively addressed the background concern. To further confirm this, we have subsequently repeated sleep and circadian rhythm assays using RNAi lines, aiming to further eliminate any remaining concerns in this regard. It appears to replicate the reduced sleep phenotype seen at night. This result has been included in the Figure S1. It is true that we have not specifically addressed whether the effects of Mettl5 are mapped to circadian pacemaker neurons in this study. We acknowledge this as a limitation and appreciate the importance of this question. Further investigations focusing on circadian pacemaker neurons, such as PDF- or tim-positive neurons, would be necessary to clarify the precise role of Mettl5 in regulating circadian behaviors and related molecular mechanisms.
(2) Gene expression and synaptic plasticity: gene expression profiles and the synaptic plasticity should be assessed by multiple time-point analyses since 1) they display high-amplitude oscillations over the 24-h window and 2) any phase-delaying mutation (e.g., Mettl5 deletion) could significantly affect their circadian changes. The current study performed a single time-point assessment of circadian clock/synaptic gene expression, misleading the conclusion for Mettl5 effects. Considering long-period rhythms in Mettl5 mutant clocks, transcriptome/translatome profiles in Mettl5 cannot distinguish between direct vs. indirect targets of Mettl5 (i.e., gene regulation by the loss of Mettl5-dependent m6A vs. by the delayed circadian phase in Mettl5 mutants).
In the revised version, we provided data collected at multiple time points. Specifically, we reexamined the per expression at both transcriptional and translational levels at different timepoints. The corresponding results were incorporated in Figure 4 D-F. We also dissected fly brains from UAS-DenMark, UAS-syt.eGFP/+; pdf-GAL4/+ and UAS-DenMark, UAS-syt.eGFP/+; pdf-GAL4/Mettl51bp at these four time points to quantify the synaptic structures of PDF neurons. The result has been included in revised Figure 6.
(3) The text description for gene expression profiling and Mettl5-dependent gene regulation was very detailed, yet there is a huge gap between gene expression profiling and sleep/behavioral analyses. The model in Figure 5 should be better addressed and validated.
Thank you for your suggestion. We added data to better confirm the expression changes of PER protein at different time points. Indeed, what you mention is the weak point of this paper. We did analysis thoroughly during the revision process.
The opposing changes in Period at the transcriptional versus translational levels puzzled us for some time until we identified alterations in the ubiquitin pathway components. The regulation of Period protein degradation by the ubiquitin-proteasome pathway is well-documented (Grima et al., 2002; Ko et al., 2002; Chiu et al., 2008). Additionally, previous studies have shown that N6-methyladenosine (m6A) modulates the ubiquitin-proteasome pathway in skeletal muscle physiology (Sun et al., 2023). We have incorporated this information into the revised manuscript in the last paragraph under the section titled: Clock gene regulatory loop regulating circadian rhythm was affected by Mettl51bp
Indeed, we have not yet identified an effective method to manipulate proteasome levels in genetic tests. The proteasome is a large protein complex composed of numerous subunits, making it impractical to enhance its activity simply by overexpressing individual components. Furthermore, the proteasome plays a critical role in many biological processes. Disrupting its function—such as through MG132 treatment, which we attempted—leads to significant off-target effects.
Sun J, Zhou H, Chen Z, et al. Altered m6A RNA methylation governs denervation-induced muscle atrophy by regulating ubiquitin proteasome pathway. J Transl Med. 2023;21(1):845. Published 2023 Nov 23. doi:10.1186/s12967-023-04694-3
Grima, B. et al. The F-box protein slimb controls the levels of clock proteins period and timeless. Nature 420, 178–182 (2002).
Ko, H. W., Jiang, J. & Edery, I. Role for Slimb in the degradation of Drosophila period protein phosphorylated by doubletime. Nature 420, 673–678 (2002).
Chiu, J. C., Vanselow, J. T., Kramer, A. & Edery, I. The phosphooccupancy of an atypical SLIMB-binding site on PERIOD that is phosphorylated by DOUBLETIME controls the pace of the clock. Genes Dev. 22, 1758–1772 (2008).
Reviewer #3 (Public review):
Xiaoyu Wu and colleagues examined the potential role in sleep of a Drosophila ribosomal RNA methyltransferase, mettl5. Based on sleep defects reported in CRISPR generated mutants, the authors performed both RNA-seq and Ribo-seq analyses of head tissue from mutants and compared to control animals collected at the same time point. While these data were subjected to a thorough analysis, it was difficult to understand the relative direction of differential expression between the two genotypes. In any case, a major conclusion was that the mutant showed altered expression of circadian clock genes, and that the altered expression of the period gene in particular accounted for the sleep defect reported in the mettl5 mutant. As noted above, a strength of this work is its relevance to a human developmental disorder as well as the transcriptomic and ribosomal profiling of the mutant. However, there are numerous weaknesses in the manuscript, most of which stem from misinterpretation of the findings, some methodological approaches, and also a lack of method detail provided. The authors seemed to have missed a major phenotype associated with the mettl5 mutant, which is that it caused a significant increase in period length, which was apparent even in a light: dark cycle. Thus the effect of the mutant on clock gene expression more likely contributed to this phenotype than any associated with changes in sleep behavior.
Recommendations for the authors:
Reviewer #1 (Recommendations for the authors):
Some of the questions that the authors should address are the following ones:
How does Mettl5 control the translation of the clock genes ? Why the level of some genes are specifically increased or decreased? What is the relation with the effect on uORF and dORF, overlapping and non overlapping ones? The observation of these defects is interesting but how they occurs and how they impact clock signaling is missing.
Thank you for your suggestion. This is the weak point of this paper. We did analysis thoroughly during the revision process.
The opposing changes in Period at the transcriptional versus translational levels puzzled us for some time until we identified alterations in the ubiquitin pathway components. The regulation of Period protein degradation by the ubiquitin-proteasome pathway is well-documented (Grima et al., 2002; Ko et al., 2002; Chiu et al., 2008). Additionally, previous studies have shown that N6-methyladenosine (m6A) modulates the ubiquitin-proteasome pathway in skeletal muscle physiology (Sun et al., 2023). We have incorporated this information into the revised manuscript in the last paragraph under the section titled: Clock gene regulatory loop regulating circadian rhythm was affected by Mettl51bp.
Indeed, we have not yet identified an effective method to manipulate proteasome levels in genetic tests. The proteasome is a large protein complex composed of numerous subunits, making it impractical to enhance its activity simply by overexpressing individual components. Furthermore, the proteasome plays a critical role in many biological processes. Disrupting its function—such as through MG132 treatment, which we attempted—leads to significant off-target effects.
In this study, we also observed codon usage alterations caused by the mettl5 mutant. For details, please refer to our responses to 4th question of the weakness session above. Previous studies have reported mettl5's role in translational regulation in other systems (Rong et al., 2020; Peng et al., 2022). Based on these findings, we propose that both translational regulation and protein degradation may contribute to the upregulation of Period protein in the mettl5 mutant. This hypothesis has been included in the Discussion section of the revised manuscript.
“The mechanism by which METTL5 regulates translation warrants further investigation. Previous studies have demonstrated that METTL5 influences translation (Rong et al., 2020; Peng et al., 2022), but whether the mechanisms identified here are conserved across other systems remains an intriguing question. In our analysis, we observed increased usage of aspartate (Asp) codons in Mettl5 mutants. Notably, prior work has linked codon usage to PER protein function—specifically, a codon-optimized version of PER failed to rescue circadian rhythmicity in per mutant flies, unlike the wild-type version (Fu et al., 2016). Further analysis revealed that PER protein levels were elevated in these mutants, suggesting that codon optimization enhances PER expression (Figure 2B in Fu et al., 2016). Strikingly, when we examined the codon-optimized region from Fu et al. (2016), we found that GAC (Asp) was highly enriched, raising the possibility that Mettl5 mutation affects PER protein accumulation by altering GAC codon usage. Additional experiments will be needed to validate this hypothesis. Furthermore, we detected changes in upstream open reading frames (uORFs) in Mettl5 mutants, but their relationship to translational regulation requires further exploration.”
References
Sun J, Zhou H, Chen Z, et al. Altered m6A RNA methylation governs denervation-induced muscle atrophy by regulating ubiquitin proteasome pathway. J Transl Med. 2023;21(1):845. Published 2023 Nov 23. doi:10.1186/s12967-023-04694-3
Grima, B. et al. The F-box protein slimb controls the levels of clock proteins period and timeless. Nature 420, 178–182 (2002).
Ko, H. W., Jiang, J. & Edery, I. Role for Slimb in the degradation of Drosophila period protein phosphorylated by doubletime. Nature 420, 673–678 (2002).
Chiu, J. C., Vanselow, J. T., Kramer, A. & Edery, I. The phosphooccupancy of an atypical SLIMB-binding site on PERIOD that is phosphorylated by DOUBLETIME controls the pace of the clock. Genes Dev. 22, 1758–1772 (2008).
Rong B, Zhang Q, Wan J, et al. Ribosome 18S m6A Methyltransferase METTL5 Promotes Translation Initiation and Breast Cancer Cell Growth. Cell Rep. 2020;33(12):108544. doi:10.1016/j.celrep.2020.108544
Peng H, Chen B, Wei W, et al. N6-methyladenosine (m6A) in 18S rRNA promotes fatty acid metabolism and oncogenic transformation. Nat Metab. 2022;4(8):1041-1054. doi:10.1038/s42255-022-00622-9
Fu J, Murphy KA, Zhou M, Li YH, Lam VH, Tabuloc CA, Chiu JC, Liu Y. Codon usage affects the structure and function of the Drosophila circadian clock protein PERIOD. Genes Dev. 2016 Aug 1;30(15):1761-75.
Reviewer #2 (Recommendations for the authors):
Please find my comments to improve the quality of your manuscript.
Major comments
(1) The quality of text writing in English needs to be at publishable levels. It is not a trivial problem, but it literally impairs the readability of your work. So please have professionals edit your manuscript text appropriately.
We have carefully revised the language throughout the manuscript during the revision process.
(2) Fig 1O: please include the total sleep profile and other analyses for rebound sleep phenotypes in control vs. Mettl5 to better validate that both genotypes were comparably sleep-deprived, but the latter shows less sleep rebound.
Thank you for your suggestion, The other reviewer also suggested to reanalyze the sleep rebound data. We did the analysis according to the following reference. We included data sleep profiles of both genotypes in original Fig 1O. Total sleep profile and other analyses for rebound sleep phenotypes are included in the revised panel. As shown in this revised panel (now Figure 1K, L), both genotypes were comparably sleep-deprived.
Cirelli C, Bushey D, Hill S, Huber R, Kreber R, Ganetzky B, Tononi G. 2005. Reduced sleep in Drosophila Shaker mutants. Nature 434:1087-92.
(3) Line 90: the authors did not actually address this critical question. Additional Gal4 mapping (e.g., Mettl5 rescue or Mettl5 RNAi) will determine which cells/neural circuits are important for Mettl5-dependent sleep.
This sentence has been revised into “The observed expression pattern of Mettl5 further supports its sleep regulatory function.”
(4) Fig 1H-L; Fig 2H-L: the authors should check if overexpression of wild-type or mutant Mettl5 in control backgrounds could affect nighttime sleep to better define the transgenic effects among overexpression, rescue, and dominant-negative.
Thank you for the comment. We added the overexpression phenotypes in the revised version.
(5) Lines 225-226. Fig S11: The neural projections from PDF-expressing neurons should be better imaged and quantified. Current images can visualize PDF projections onto the optic lobe but not others (e.g., dorsal, POT), so the conclusion is not validated.
Thank you for the suggestion. We acknowledge the limitation in the current images of PDF-expressing neuronal projections. We included new, higher-resolution images to better visualize and quantify the neural projections, including the dorsal and POT regions, to ensure the conclusion is well-supported.
(6) Lines 230-232: per RNA/PER protein expression oscillates daily, so the authors should perform time-point experiments to conclude Mettl5 effects on clock gene expression, including per.
Thank you for the insightful comment. We performed experiments in the Mettl5 mutant background at four time points to analyze PER protein expression using both RT-PCR and Western blot (anti-PER). The updated results have been included in Figure 4D-F.
(7) Lines 235-238: the authors should note that Mettl5 effects on sleep in Clk or per mutant backgrounds are actually opposite to those in w1118/control one. Mettl5 deletion promotes daytime or nighttime sleep in Clk or per mutants, respectively. Any explanation?
We are trying to use epistasis analysis to determine which gene is upstream here. Epistasis (or epistatic effect) in genetics refers to the interaction between different genes where the expression of one gene (the epistatic gene) masks or modifies the expression of another gene (the hypostatic gene). The epistatic gene (masking gene) usually functions downstream in the pathway because its effect overrides the output of the hypostatic gene. The double mutant showed the similar phenotype as downstream genes. Thus, Clk or per functions downstream of Mettl5.
(8) Fig 6: The dorsal PDF projections actually show time-dependent plasticity. Results from the single time-point are not conclusive.
Thank you for the insightful comment. we further dissected fly brains from UAS-DenMark, UAS-syt.eGFP/+; pdf-GAL4/+ and UAS-DenMark, UAS-syt.eGFP/+; pdf-GAL4/Mettl51bp at these four time points to analyze the morphology of PDF neurons. The results have been included in figure 6.
Minor comments
(1) Please avoid simple bar graphs in the data presentation-include individual data points or use a different graph showing the distribution of raw data (e.g., violin plot, box plot, etc.).
Thank you for the suggestion. In the revised version of the manuscript, we have included individual data points, violin plots, and box plots to present the data, effectively showing both the distribution and differences in the raw data.
(2) Line 19: "Clock" indicates the gene name or general terminology such as "circadian clock". Please clarify it and revise the font accordingly.
This has been revised into“clock”
(3) The overall flow in the Abstract/Summary is somewhat challenging for a general audience to follow.
We have revised the text, especially the overall flow in the Abstract/Summary.
(4) Fonts for the names of genes and gene products (i.e., mRNA, protein) should be appropriately corrected throughout the manuscript.
We have checked the text and made changes where necessary.
(5) Methods: the authors should provide detailed information on the methods. For instance, there is little description of how they generate Mettl5 deletions (e.g., sgRNA/target sequence). Also, they should clarify whether they test heterozygous vs. homozygous mutants of Mettl5 deletions in each experiment since the genotype description in the figure appears mixed-up (e.g., Fig 1B vs. Fig 1I-L).
Thank you for pointing this out. In the updated version, we provided detailed information about the strains used, including the sgRNA/target sequences for generating Mettl5 deletions. Regarding the genotypes, Figure 1B represents homozygous mutants, while Figures 1I-L represent heterozygous mutants. This distinction has been clarified in the figure legends, and the genotype notation for Figures 1I-L will be revised for consistency and clarity.
(6) Fig 1: the figure panels should be re-arranged based on the order of their text description (i.e., Fig 1H-L should go after Fig 1M-O).
Thank you for the suggestion. In the revised version, we rearranged the figure panels so that Figures 1H-L appear after Figures 1M-O, following the order of their description in the text.
(7) Sleep education in Trmt112 RNAi looks different from that in Mettl5 mutant het. Any explanation?
The functional divergence between Trmt112 and Mettl5 may also contribute to the observed sleep phenotype. While Trmt112 and Mettl5 share some downstream targets, they each regulate many unique genes, some of which could influence sleep. Sleep is a highly sensitive trait that can be modulated by numerous genetic factors. Previous studies have also suggested that sleep behaves more like a quantitative trait, reflecting the combined effects of multiple genes (Mackay and Huang, 2018).
Mackay TFC, Huang W. Charting the genotype-phenotype map: lessons from the Drosophila melanogaster Genetic Reference Panel. Wiley Interdiscip Rev Dev Biol. 2018;7(1):10.1002/wdev.289. doi:10.1002/wdev.289
Reviewer #3 (Recommendations for the authors):
A detailed critique is provided below. Generally, the authors can greatly improve this manuscript if they focus more rigorously on the circadian phenotype associated with the Mettl5 mutant, which could be the basis for the apparent sleep phenotype.
(1) Please provide more information as to how each of the mettl5 mutants were generated. This information should include, specifically, the gRNA sequences, plasmids generated for the 5' and 3' arms, and anything related to the CRISPR approach for generating the mutants. Was any sequencing done to verify the CRISPR alleles, or was this limited to the analysis of mettl5 expression and behavior? Please indicate where the qPCR primers (used in Fig 1B) are located relative to the mutant loci. The figure legend is also incomplete in that there is no reference to the boxed area in Fig 1A.
In the updated version, we have provided detailed information about the how each of the mettl5 mutants were generated. The sequence was verified by sequencing following PCR. The following references to the boxed area were added in the revised version.
Reference
Iyer LM, Zhang D, Aravind L. Adenine methylation in eukaryotes: Apprehending the complex evolutionary history and functional potential of an epigenetic modification. Bioessays. 2016 Jan;38(1):27-40. doi: 10.1002/bies.201500104.
(2) As noted, I am not in agreement with the interpretation of findings for the sleep defect reported in the mettl5[1b]/+ mutants. There is a clear increase in morning sleep in the mutants that may not have reached significance by lumping the data in 12h increments (Fig1C-E). Were the overall 24h sleep values between the mutants and controls the same? The sleep profile appears to be shifted, such that nighttime sleep onset in the mutants occurs much later than wild type, and daytime waking is also much later, all pointing to a long period phenotype, which is very strongly supported by the data in Table 1, as well as the RNA- and ribo-seq data. The implications for this leading to sleep disturbances in humans is very exciting. An additional suggestion to the authors here is to report the nighttime sleep latency values (time to onset of the first sleep bout after lights off).
We appreciate your insightful observation. As shown in Table 1, the Mettl51bp/+ mutant exhibits a robust long-period phenotype, with circadian rhythms significantly extended to 28.3 ± 0.4 hours compared to the wild-type's 23.9 ± 0.05 hours. This prolonged period perfectly aligns with the observed behavioral phenotypes, including delayed nighttime sleep onset, later daytime waking, and the overall shift in sleep profile. This is indeed quite similar to previous report on Period3 variant (Zhang et al., 2016). We agree that the prolonged circadian period contributes to the observed sleep phenotype. However, since total sleep time was significantly reduced in the mutant, we cannot attribute the phenotype solely to period lengthening. Furthermore, our 24-hour PER expression analysis in mettl5 mutants revealed elevated PER protein levels at ZT1 and ZT18, while ZT6 and ZT12 showed no significant changes, with no apparent phase shift. These findings collectively suggest that the phenotype primarily results from PER protein stabilization and accumulation.
Importantly, genetic rescue experiments restoring wild-type Mettl5 function (UAS-Mettl5/Mettl5-Gal4; Figure 1 and Table 1) completely normalized the circadian period to 24 ± 0.02 hours, providing compelling evidence that these phenotypes specifically result from loss of Mettl5 function. Together with the sleep architecture data, these findings establish Mettl5 as a crucial regulator of circadian rhythms, with important implications for understanding human sleep disorders. To further substantiate these observations, we have now included quantitative nighttime sleep latency measurements in the revised manuscript to better document the delayed sleep onset in mutants (Figure S1G).
We have discussed this in the third paragraph of the Discussion session and included the reference in the revised manuscript.
Zhang L, Hirano A, Hsu PK, et al. A PERIOD3 variant causes a circadian phenotype and is associated with a seasonal mood trait. Proc Natl Acad Sci U S A. 2016;113(11):E1536-E1544. doi:10.1073/pnas.1600039113.
(3) The description for how circadian behavior was measured and analyzed (Table 1) is missing from the methods section.
We have included a detailed description of the methods used to measure and analyze circadian behavior, as presented in Table 1, in the revised methods “Sleep behavior assays” section.
(4) Please explain what the "awake %" values reported in Figs 1G, 1L, Fig 2G, and 2L, Fig 4G and 4M are. Is this simply the number of flies that are awake at a given time point? This does not provide useful information beyond what is already reported for the sleep profiling in other parts of these figures. If it is an arousal threshold assay, as shown in supplementary Fig 1H, please indicate this. The description for "sleep arousal" in the methods (lines 368-371) is also concerning. If most of the mutant flies are already awake at ZT 14, then I would expect that this assay would not work at this time of day. A more suitable time point would be ZT 19, or later, when the mutants are falling asleep. Moreover, calculating the number of flies awakened as long as 5 minutes after a stimulus pulse cannot be distinguished from a spontaneous awakening, and so is not really a metric of arousal threshold. The number of sleeping flies awakened by the stimulus should be calculated within, at most, one minute afterward.
Thank you for your suggestion. Regarding the 'awake %' metric, it indicates that at specific time points (e.g., ZT14), the percentage of awake fruit fly population at that moment. In the revised version, we further clarify the definition and significance of 'awake %'. Additionally, we have reevaluated the time points for the arousal threshold assay, selecting a more appropriate time (e.g., ZT19) to better reflect the sleep state of the mutants. Based on your suggestion, we calculate the number of flies awakened within one minute after the stimulus to ensure a more accurate measurement of arousal threshold. This has been included in the revised Figure 1M.
(5) Fig1M-O is problematic. First, is it possible that expression of Mettl5 mRNA fluctuates with time-of-day and is not affected by sleep loss? There are no undisturbed controls collected at equivalent time points. The method used for quantifying sleep rebound in Fig 1O (lines 365-367) does not make sense, as negative values would be expected. Moreover, since the Mettl5 mutants show high sleep amounts in the morning and very low sleep amounts from ZT 12-18, this analysis would be severely confounded. Also, the sleep deprivation applied would not produce equivalent amounts of sleep loss as compared to wild type controls, so this also needs to be corrected. The authors should consider consulting Cirelli et al (2005, DOI: 10.1038/nature03486 ) as an approach for quantifying sleep homeostasis in a short-sleeping mutant. Please also show the sleep profiling in the mutants for these experiments.
Thank you for your valuable suggestions. Regarding the possibility that Mettl5 mRNA expression fluctuates with circadian rhythms rather than being affected by sleep deprivation, we acknowledge that collecting undisturbed control samples at equivalent time points would provide critical insights. In the revised version, we included undisturbed controls to distinguish between circadian-driven fluctuations and the effects of sleep deprivation on Mettl5 expression.
For the quantification of sleep rebound in Figure 1O, we agree that the current method may not fully capture the dynamics of sleep recovery, especially in Mettl5 mutants, where sleep patterns differ significantly from wild-type. We have referred to the method proposed by Cirelli et al. paper for quantifying sleep homeostasis in short-sleeping mutants, ensuring a more accurate evaluation of sleep rebound. The results have been included in Figure 1K-L of the revised version.
(6) Fig 3B and C (minor) - while the volcano plots are clear, it is not clear whether "down" or "up" means for the mutant relative to wild type or the other way around? Please clarify. In Fig 3P, the legend indicates a depiction of the "top 5 pathway associated genes", but it seems there are 10 pathways depicted. Which of these are the "top 5"?
In the volcano plots (Fig. 3B and 3C), “up” and “down” refer to genes that the mutant relative to the wild-type strain. In Fig. 3P, the legend was mislabeled as “top 5” pathway-associated genes. In fact, we displayed the top 10 pathway-associated genes. We apologize for the confusion and will correct both the figure legend and the corresponding text in our revised manuscript.
(7) Fig 4 D-E, and F,G do not have sufficient information to draw the conclusion that Per mRNA/protein expression is increased in the Mettl5 mutant. Since both mRNA protein of this gene oscillates significantly throughout the day, it is still possible that the single time point shown in this figure might indicate a disruption in cycling rather than overall expression level. Please first indicate what time of day the tissue was collected, second, consider adding more time points to both assays. For the first part of this figure, A and B, per and Clock gene expression are expected to be in different phases, and so this aspect is not unexpected. However, it is notable that it is reversed in the mutant vs wild type. Again, an alternate interpretation of this finding that the authors have not considered is a change in period duration of gene cycling.
Thank you for your suggestion. For the PER WB experiments, we have included multiple time points in the revised version to more comprehensively evaluate PER expression in the Mettl5 mutant and better understand its circadian rhythm changes. We appreciate your observation regarding the potential changes in the period duration of gene cycling. This has been discussed in the 3rd paragraph of the Discussion session of the revised version.
(8) The data shown in Figs 4H-M does not support the conclusion that "Clock and Per genes were downstream of Mettl5" (line 236-237). The daytime sleep phenotype, in particular, appears additive between both circadian genes and mutant because the morning sleep of the double mutant is much higher than either mutant by itself. Statistical comparisons between the double mutant and each clock mutant are also noticeably missing. These data are difficult to interpret. One potential explanation is that Mettl5 alters gene expression of non-circadian genes, and that the phenotypes become additive when both clock and Mettl5 genes are missing. A full molecular analysis of clock gene cycling in the Mettl5 mutant may help improve understanding of the relationship between the circadian clock Mettl5 gene expression. It may also be worthwhile checking whether Mettl5 gene expression itself shows a daily oscillation.
Thank you for your suggestion. In the revised version, we have included four additional time points to analyze the oscillatory expression of Per and Clock in the Mettl5 mutant, providing a more comprehensive understanding of their circadian rhythm changes. In Figs 4H-M, we are trying to use epistasis analysis to determine which gene is upstream here. Epistasis (or epistatic effect) in genetics refers to the interaction between different genes where the expression of one gene (the epistatic gene) masks or modifies the expression of another gene (the hypostatic gene). The epistatic gene (masking gene) usually functions downstream in the pathway because its effect overrides the output of the hypostatic gene. The double mutant showed the similar phenotype as downstream genes. Thus, Clk or per functions downstream of Mettl5. Statistical comparisons between the double mutant and each clock mutant are added.
(9) In Fig 6, what time of day were the flies collected? PDF terminal morphology is known to change throughout the day; this is another piece of data that could indicate a defect in circadian function rather than a chronic change in synaptic morphology.
The flies were collected around ZT14. We included additional dissection time points in future experiments. Differences between the control and Mettl5 mutants are observed consistently across multiple time points, suggesting that Mettl5 has an impact on synaptic plasticity.
Minor:
There are letter indicators, presumably for statistical comparisons, depicted in Figs 1 and 2 (panels I-L), but no explanation as to what these mean in the figure legends.
We have added notes in the revised version.
What is the purpose of the boxed regions shown in Fig S1A-F? There is no explanation of these in the figure legend nor in the text.
The boxed regions highlight the significant co-localization of two proteins. We have included this explanation in the figure legend in the revised version.
The statement (lines 310-311) that per and clock genes "exhibit more pronounced sleep rebound after sleep deprivation" is inaccurate. The article cited for this (Shaw et al 2002) showed that it was female mutants of the cycle gene which showed prolonged sleep rebound; other clock mutants were normal.
Thank you for pointing out this. We revised the statement accordingly.
Overall, the manuscript may benefit from editing or writing assistance to improve the language. There were many incomplete sentences, grammatical errors, etc.
We have carefully refined the language throughout the manuscript during the revision process.
-

-

-

eLife Assessment
In this manuscript, the authors present useful findings demonstrating that the RNA modification enzyme Mettl5 regulates sleep in Drosophila. Through transcriptome- and proteome-wide analyses, the authors identified downstream targets affected in heterozygous mutants and proposed that Mettl5 regulates the translation and degradation of clock genes to maintain normal sleep function. However, the mechanisms by which Mettl5 achieves these functions, and whether they are direct or indirect, remain incomplete and would benefit from further analysis.
-

Reviewer #1 (Public review):
Summary:
Here the authors attempted to test whether the function of Mettl5 in sleep regulation was conserved in drosophila, and if so, by which molecular mechanisms. To do so they performed sleep analysis, as well as RNA-seq and ribo-seq in order to identify the downstream targets. They found that the loss of one copy of Mettl5 affects sleep and that its catalytic activity is important for this function. Transcriptional and proteomic analyses show that multiple pathways were altered, including the clock signaling pathway and the proteasome. Based on these changes the authors propose that Mettl5 modulate sleep through regulation of the clock genes, both at the level of their production and degradation.
Strengths:
The phenotypical consequence of the loss of one copy of Mettl5 on sleep function is clear and …
Reviewer #1 (Public review):
Summary:
Here the authors attempted to test whether the function of Mettl5 in sleep regulation was conserved in drosophila, and if so, by which molecular mechanisms. To do so they performed sleep analysis, as well as RNA-seq and ribo-seq in order to identify the downstream targets. They found that the loss of one copy of Mettl5 affects sleep and that its catalytic activity is important for this function. Transcriptional and proteomic analyses show that multiple pathways were altered, including the clock signaling pathway and the proteasome. Based on these changes the authors propose that Mettl5 modulate sleep through regulation of the clock genes, both at the level of their production and degradation.
Strengths:
The phenotypical consequence of the loss of one copy of Mettl5 on sleep function is clear and well-documented.
Weaknesses:
The imaging and molecular parts are less convincing.
- The colocalization of Mettl5 with glial and neuronal cells is not very clear
- The section on gene ontology analysis is long and confusing
- Among all the pathways affected the focus on proteosome sounds like cherry picking. And there is no experiment demonstrating its impact in the Mettl5 phenotype
- The ribo seq shows some changes at the level of translation efficiency but there is no connection with the Mettl5 phenotypes. In other words, how the increased usage of some codons impact clock signalling. Are the genes enriched for these codons?
- A few papers already demonstrated the role of Mettl5 in translation, even at the structural level (Rong et al, Cell reports 2020) and this was not commented by the authors. In Peng et al, 2022 the authors show that the m6A bridges the 18S rRNA with RPL24. Is this conserved in Drosophila?
- The text will require strong editing and the authors should check and review extensively for improvements to the use of English.Conclusion
Despite the effort to identify the underlying molecular defects following the loss of Mettl5 the authors felt short in doing so. Some of the results are over-interpreted and more experiments will be needed to understand how Mettl5 controls the translation of its targets. References to previous works was poorly commented.
-

Reviewer #2 (Public review):
Summary:
The authors define the m6A methyltransferase Mettl5 as a novel sleep-regulatory gene that contributes to specific aspects of Drosophila sleep behaviors (i.e., sleep drive and arousal at early night; sleep homeostasis) and propose the possible implication of Mettl5-dependent clocks in this process. The model was primarily based on the assessment of sleep changes upon genetic/transgenic manipulations of Mettl5 expression (including CRISPR-deletion allele); differentially expressed genes between wild-type vs. Mettl5 mutant; and interaction effects of Mettl5 and clock genes on sleep. These findings exemplify how a subclass of m6A modifications (i.e., Mettl5-dependent m6A) and possible epi-transcriptomic control of gene expression could impact animal behaviors.
Strengths:
Comprehensive DEG analyses …
Reviewer #2 (Public review):
Summary:
The authors define the m6A methyltransferase Mettl5 as a novel sleep-regulatory gene that contributes to specific aspects of Drosophila sleep behaviors (i.e., sleep drive and arousal at early night; sleep homeostasis) and propose the possible implication of Mettl5-dependent clocks in this process. The model was primarily based on the assessment of sleep changes upon genetic/transgenic manipulations of Mettl5 expression (including CRISPR-deletion allele); differentially expressed genes between wild-type vs. Mettl5 mutant; and interaction effects of Mettl5 and clock genes on sleep. These findings exemplify how a subclass of m6A modifications (i.e., Mettl5-dependent m6A) and possible epi-transcriptomic control of gene expression could impact animal behaviors.
Strengths:
Comprehensive DEG analyses between control and Mettl5 mutant flies reveal the landscape of Mettl5-dependent gene regulation at both transcriptome and translatome levels. The molecular/genetic features underlying Mettl5-dependent gene expression may provide important clues to molecular substrates for circadian clocks, sleep, and other physiology relevant to Mettl5 function in Drosophila.
Weaknesses:
While these findings indicate the potential implication of Mettl5-dependent gene regulation in circadian clocks and sleep, several key data require substantial improvement and rigor of experimental design and data interpretation for fair conclusions. Weaknesses of this study and possible complications in the original observations include but are not limited to:
(1) Genetic backgrounds in Mettl5 mutants: the heterozygosity of Mettl5 deletion causes sleep suppression at early night and long-period rhythms in circadian behaviors. The transgenic rescue using Gal4/UAS may support the specificity of the Mettl5 effects on sleep. However, it does not necessarily exclude the possibility that the Mettl5 deletion stocks somehow acquired long-period mutation allelic to other clock genes. Additional genetic/transgenic models of Mettl5 (e.g., homozygous or trans-heterozygous mutants of independent Mettl5 alleles; Mettl5 RNAi etc.) can address the background issue and determine 1) whether sleep suppression tightly correlates with long-period rhythms in Mettl5 mutants; and 2) whether Mettl5 effects are actually mapped to circadian pacemaker neurons (e.g., PDF- or tim-positive neurons) to affect circadian behaviors, clock gene expression, and synaptic plasticity in a cell-autonomous manner and thereby regulate sleep. Unfortunately, most experiments in the current study rely on a single genetic model (i.e., Mettl5 heterozygous mutant).
(2) Gene expression and synaptic plasticity: gene expression profiles and the synaptic plasticity should be assessed by multiple time-point analyses since 1) they display high-amplitude oscillations over the 24-h window and 2) any phase-delaying mutation (e.g., Mettl5 deletion) could significantly affect their circadian changes. The current study performed a single time-point assessment of circadian clock/synaptic gene expression, misleading the conclusion for Mettl5 effects. Considering long-period rhythms in Mettl5 mutant clocks, transcriptome/translatome profiles in Mettl5 cannot distinguish between direct vs. indirect targets of Mettl5 (i.e., gene regulation by the loss of Mettl5-dependent m6A vs. by the delayed circadian phase in Mettl5 mutants).
(3) The text description for gene expression profiling and Mettl5-dependent gene regulation was very detailed, yet there is a huge gap between gene expression profiling and sleep/behavioral analyses. The model in Figure 5 should be better addressed and validated.
-

Reviewer #3 (Public review):
Xiaoyu Wu and colleagues examined the potential role in sleep of a Drosophila ribosomal RNA methyltransferase, mettl5. Based on sleep defects reported in CRISPR generated mutants, the authors performed both RNA-seq and Ribo-seq analyses of head tissue from mutants and compared to control animals collected at the same time point. While these data were subjected to a thorough analysis, it was difficult to understand the relative direction of differential expression between the two genotypes. In any case, a major conclusion was that the mutant showed altered expression of circadian clock genes, and that the altered expression of the period gene in particular accounted for the sleep defect reported in the mettl5 mutant. As noted above, a strength of this work is its relevance to a human developmental disorder as …
Reviewer #3 (Public review):
Xiaoyu Wu and colleagues examined the potential role in sleep of a Drosophila ribosomal RNA methyltransferase, mettl5. Based on sleep defects reported in CRISPR generated mutants, the authors performed both RNA-seq and Ribo-seq analyses of head tissue from mutants and compared to control animals collected at the same time point. While these data were subjected to a thorough analysis, it was difficult to understand the relative direction of differential expression between the two genotypes. In any case, a major conclusion was that the mutant showed altered expression of circadian clock genes, and that the altered expression of the period gene in particular accounted for the sleep defect reported in the mettl5 mutant. As noted above, a strength of this work is its relevance to a human developmental disorder as well as the transcriptomic and ribosomal profiling of the mutant. However, there are numerous weaknesses in the manuscript, most of which stem from misinterpretation of the findings, some methodological approaches, and also a lack of method detail provided. The authors seemed to have missed a major phenotype associated with the mettl5 mutant, which is that it caused a significant increase in period length, which was apparent even in a light: dark cycle. Thus the effect of the mutant on clock gene expression more likely contributed to this phenotype than any associated with changes in sleep behavior.
-

-