Hierarchical cortical plasticity in congenital sight impairment
Curation statements for this article:-
Curated by eLife
eLife Assessment
This important study examines the stability and compensatory plasticity in the retinotopic mapping in patients with congenital achromatopsia. It provides convincing evidence for a stable mapping of the visual field in V1, alongside changes of the readout from V1 into V3, which shows revised receptive field location and size. This paper would be of interest to scientists studying the visual system, brain plasticity, and development.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
A robust learning system balances adaptability to new experiences with stability of its foundational architecture. To investigate how the human brain implements this we used a new approach to study plasticity and stability across hierarchical processing stages in visual cortex. We compare the rod system of individuals born with rod-only photoreceptor inputs (achromatopsia) to the typically developed rod system, allowing us to dissociate impacts of life-long versus transient responses to altered input. Cortical input stages (V1) exhibited high stability, with input-deprived cortex showing no retinotopic remapping and exhibiting structural hallmarks of deprivation. However, plasticity manifested as reorganised read-out of these inputs by higher-order cortex, in a pattern that could compensate for the lower resolution of a rod-only system and its lack of high-density foveal input. We propose that these hierarchical dynamics robustly optimize processing of available input and could reflect a broader principle of brain organisation with important implications for emerging sight-rescue therapies.
Article activity feed
-

-

-

eLife Assessment
This important study examines the stability and compensatory plasticity in the retinotopic mapping in patients with congenital achromatopsia. It provides convincing evidence for a stable mapping of the visual field in V1, alongside changes of the readout from V1 into V3, which shows revised receptive field location and size. This paper would be of interest to scientists studying the visual system, brain plasticity, and development.
-

Reviewer #1 (Public review):
Summary:
This paper examines plasticity in early cortical (V1-V3) areas in an impressively large number of rod monochromats (individuals with achromatopia). The paper examines three things:
(1) Cortical thickness. It is now well established that early complete blindness leads to increases in cortical thickness. This paper shows increased thickness confined to the foveal projection zone within achromats. This paper replicates work by Molz (2022) and Lowndes (2021), but the detailed mapping of cortical thickness as a function of eccentricity and the inclusion of higher retinotopic areas is particularly elegant.
(2) Failure to show largescale reorganization of early visual areas using retinotopic mapping. This is a replication of a very recent study of Molz et al. but I believe, given anatomical variability, …
Reviewer #1 (Public review):
Summary:
This paper examines plasticity in early cortical (V1-V3) areas in an impressively large number of rod monochromats (individuals with achromatopia). The paper examines three things:
(1) Cortical thickness. It is now well established that early complete blindness leads to increases in cortical thickness. This paper shows increased thickness confined to the foveal projection zone within achromats. This paper replicates work by Molz (2022) and Lowndes (2021), but the detailed mapping of cortical thickness as a function of eccentricity and the inclusion of higher retinotopic areas is particularly elegant.
(2) Failure to show largescale reorganization of early visual areas using retinotopic mapping. This is a replication of a very recent study of Molz et al. but I believe, given anatomical variability, the larger n in this study, and how susceptible pRF findings are to small changes in procedure, this replication is also of interest.
(3) Connective field modelling, examining the connections between V3-V1. The paper finds changes in the pattern of connections, and smaller connective fields in individuals with achromatopsia than normally sighted controls, and suggests that these reflect compensatory plasticity, with V3 compensating for the lower resolution V1 signal in individuals with achromatopsia.
This is a carefully done study (both in terms of data collection and analysis) that is an impressive amount of work.
*Effects of eye-movements
The authors have carried out the eye-movement analyses I asked of them. Unfortunately, in 4 individuals they couldn't calibrate the eyetracker (it's impressive they managed in 10). I think this means that 4 of 13 (since a different participant was excluded from head motion) individuals weren't included in correlation analyses. Limiting the correlation analysis to individuals with better fixation has obvious issues. I'd recommend redoing (or additionally including) stats using non-parametric measures while classifying these 4 as having fixation instability of 3 (i.e. greater instability than the participant with the worst fixation who was successfully calibrated).
*Interpreting pRFs
The paper would be strengthened by a little more explicit clarity about what pRFs represent and how that affects their interpretation of their findings as plasticity vs. non-plasticity (I know the authors are aware of this, but I think it would be helpful for readers who are less experienced in pRFs). In the introduction it would be helpful to point out that pRFs represent the collective response of a large population of neurons, and as a result pRF estimates can vary depending on which population of neurons that stimulus drives.
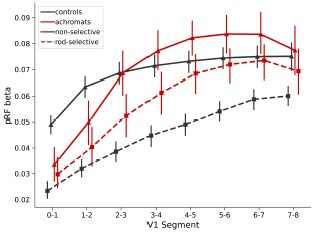
For example, imagine for the sake of argument that rods only project to V1 neurons with larger receptive fields. If one measured pRFs in a control observer under phototopic vs. scotopic conditions one would see smaller pRFs in the photopic conditions. This wouldn't represent 'plasticity' - it would represent the fact that the firing neurons contributing to the pRF signal are a slightly different population because of a change in the stimulus content. This is of course exactly what you see in 2C. And indeed, the authors make this identical point ". In the non-selective condition, the smaller pRFs in controls are in line with the higher spatial resolution of the
cone system, which is not active in the achromat group." But this point would be clearer if more of the conceptual underpinnings were made explicit in the introduction (or at this point in the paper).Shifts in which population of neurons drive your pRFs can explain main of the more puzzling results in the paper without detracting from your main conclusions. For example, in 2D, I don't think it's differences in S/N driving your results (pRFs are at least theoretically meant to be robust to S/N). If smaller RFs 'drop out' under low luminance and these smaller RFs also tend to be more central, then one would expect the control results of 1D. And I think a similar argument might even be made for the smaller difference in the rod monochromats.
It would be possible to make the point of Figure 4B more simply if Figure 4B was replaced by additional Panels in Figure 2 simply showing V3 pRF sizes/eccentricity distributions. That would make the point that you don't see the same expansion in pRF sizes in V3 in a way that is just as clear, and is closer to the data.
*Interpreting cRFs
Similarly, I think the paper would be improved with more clarity about the underlying signal in CF modeling. Once again, I appreciate that the authors are familiar with this, but it will help the reader in interpretation. (And I do believe thinking carefully about this may alter your interpretations). CF receptive fields 'find' the region in V1 that best predict the V3 signal in a given voxel. In resting state this likely represents a combination of:
(1) visually driven signal - correlations that may or may not reflect connectivity but represent the fact that regions that represent the same region of visual space will be active at the same time.
(2) global bilaterally symmetrical signal consisting of enhanced correlations between iso-eccentric regions (Raemaekers et al., 2014), which may arise from vasculature that symmetrically stems from the posterior cerebral artery (Tong et al., 2013; Tong and Frederick, 2014).
(3) intrinsic neural fluctuations that are more strongly correlated between connected neurons. These are likely quite weak compared to the other contributions.
I think if you ignore 2, (which is not likely to differ between rod mono and controls) and model 1 and 3, you might well see shifts in CFs towards the boundary of the scotoma - essentially the CF's location will be biased towards the region of V1 that has stronger correlations - which = the region which has a visual signal.
I do find convincing the argument that you don't see the same shift in controls in the rod-selective condition. So I think the results of 4A are fine. But a little more clarity about 'what's under the hood' in CF modeling would be nice.
*Interpreting the relationship between pRFs and cRFs
So there's something here that confuses me. We are all agreed that V3 pRF sizes are similar across RM and control. V1 pRFs are larger in RM. It feels intuitive that smaller CFs would compensate but I can't make it make sense to myself when I think it through. Each pRF represents a combination of receptive field location scatter and bandwidth. You want to argue that eccentricity mapping looks pretty normal, so there's no reason to think increased rf scatter, and I can believe that (though I do think this assumption should be discussed explictly).
So far I think we agree.
But let's think about what drives a CF during visual stimulation ... Specifically lets think about 'the pRF of the CF' (the region of visual space represented by the cluster of voxels in the CF). If pRFs for individual voxels in V1 are big, then the pRF for the CF is also going to be large. But we know that pRFs for V3 are normal size. So, the V3 CF will 'find' a smaller number of voxels in V1, in order to try to find the 'correct sized' CF pRF. Note that this explanation is very similar to yours. But doesn't require ANY 'intrinsic' connectivity. It's really just assuming the whole thing is driven by the visual signal and the CF size is determined by the ratio of the pRF sizes in V3 vs. V1.
One possible solution would be to regress out the visual stimulus and redo this analysis based on the residuals.
-

Reviewer #3 (Public review):
Summary:
This study addresses a long-standing question in visual neuroscience concerning how the human visual system balances stability and plasticity when sensory input is altered from early in life. Using achromatopsia as a model of lifelong cone deprivation, the authors examine whether early visual cortex undergoes retinotopic reorganization to compensate for the absence of foveal cone input, or whether canonical retinotopic organization is largely preserved. By combining fMRI-based population receptive field (pRF) mapping with connective field (CF) modelling, the authors characterize changes across multiple hierarchical stages of visual processing.
The main findings indicate that primary visual cortex (V1) shows no systematic remapping of the foveal projection zone, whereas extrastriate cortex, …
Reviewer #3 (Public review):
Summary:
This study addresses a long-standing question in visual neuroscience concerning how the human visual system balances stability and plasticity when sensory input is altered from early in life. Using achromatopsia as a model of lifelong cone deprivation, the authors examine whether early visual cortex undergoes retinotopic reorganization to compensate for the absence of foveal cone input, or whether canonical retinotopic organization is largely preserved. By combining fMRI-based population receptive field (pRF) mapping with connective field (CF) modelling, the authors characterize changes across multiple hierarchical stages of visual processing.
The main findings indicate that primary visual cortex (V1) shows no systematic remapping of the foveal projection zone, whereas extrastriate cortex, particularly V3, exhibits altered patterns of sampling from V1. The authors interpret these results as evidence for hierarchical adaptation, whereby downstream readout mechanisms adjust to make more efficient use of degraded rod-mediated input while preserving early-stage retinotopic organization.
Strengths:
A major strength of this work is the use of silent substitution to generate rod-selective stimuli. This approach enables a principled comparison between achromats and typically sighted controls by isolating rod-driven responses in both groups. In doing so, the study overcomes a key limitation of prior work, where differences in cortical organization could often be confounded by differences in photoreceptor class rather than reflecting neural reorganization per se. The inclusion of a rod-driven baseline in controls provides an important reference for distinguishing long-term adaptation from transient or stimulus-driven effects.
Another notable strength is the integration of CF modelling alongside conventional pRF mapping. While pRF analyses alone suggest enlarged receptive fields in V1, consistent with reduced spatial resolution, the CF analysis offers a more mechanistic account by revealing changes in how V3 samples information from the V1 surface. This multi-level modelling approach moves beyond descriptive accounts of cortical map structure and provides a framework for interpreting how downstream areas may adjust their integration strategies under conditions of altered input.
Weaknesses:
Although the study is methodologically strong, the central claims regarding stability and compensatory plasticity require clearer conceptual framing and stronger empirical support. Stability is primarily defined as the absence of large-scale retinotopic remapping in V1, yet the presence of significantly enlarged V1 pRFs indicates substantial tuning-level plasticity at the input stage; distinguishing topographic stability from functional reorganization would therefore strengthen the interpretation. Moreover, the proposed compensatory mechanism raises a signal-processing concern, as reduced downstream sampling (smaller CFs in V3) cannot restore spatial information lost due to coarse upstream representations, and may instead limit integration. The mechanistic link between altered CF properties and normalization of extrastriate pRFs is not directly tested, as group differences are not shown to covary across individuals or visual field locations. Finally, the interpretation of these changes as compensatory implies functional benefit, yet no behavioral or performance measures are provided to establish that the observed reorganization preserves or enhances visual function, leaving open whether these effects reflect adaptive optimization or passive downstream consequences of altered input.
-

Author response:
The following is the authors’ response to the original reviews.
Public Reviews:
Reviewer #1 (Public review):
Summary:
This paper examines plasticity in early cortical (V1-V3) areas in an impressively large number of rod monochromats (individuals with achromatopia). The paper examines three things:
(1) Cortical thickness. It is now well established that early complete blindness leads to increases in cortical thickness. This paper shows increased thickness confined to the foveal projection zone within achromats. This paper replicates the work by Molz (2022) and Lowndes (2021), but the detailed mapping of cortical thickness as a function of eccentricity and the inclusion of higher visual areas is particularly elegant.
(2) Failure to show largescale reorganization of early visual areas using retinotopic mapping. This is a …
Author response:
The following is the authors’ response to the original reviews.
Public Reviews:
Reviewer #1 (Public review):
Summary:
This paper examines plasticity in early cortical (V1-V3) areas in an impressively large number of rod monochromats (individuals with achromatopia). The paper examines three things:
(1) Cortical thickness. It is now well established that early complete blindness leads to increases in cortical thickness. This paper shows increased thickness confined to the foveal projection zone within achromats. This paper replicates the work by Molz (2022) and Lowndes (2021), but the detailed mapping of cortical thickness as a function of eccentricity and the inclusion of higher visual areas is particularly elegant.
(2) Failure to show largescale reorganization of early visual areas using retinotopic mapping. This is a replication of a very recent study by Molz et al. but I believe, given anatomical variability (and the very large n in this study) and how susceptible pRF findings are to small changes in procedure, this replication is also of interest.
(3) Connective field modelling, examining the connections between V3-V1. The paper finds changes in the pattern of connections, and smaller connective fields in individuals with achromatopsia than normally sighted controls, and suggests that these reflect compensatory plasticity, with V3 compensating for the lower resolution V1 signal in individuals with achromatopsia.
Strengths:
This is a carefully done study (both in terms of data collection and analysis) that is an impressive amount of work. I have a number of methodological comments but I hope they will be considered as constructive engagement - this work is highly technical with a large number of factors to consider.
Weaknesses:
(1) Effects of eye-movements
I have some concerns with how the effects of eye-movements are being examined. There are two main reasons the authors give for excluding eye-movements as a factor in their results. Both explanations have limitations.
(a) The first is that R2 values are similar across groups in the foveal confluence. This is fine as far as it goes, but R2 values are going to be low in that region. So this shows that eyemovements don't affect coverage (the number of voxels that generate a reliable pRF), but doesn't show that eye-movements aren't impacting their other measures.
We agree with the reviewer that eye movements could affect pRF measures. We have now also included data for all participants where we were able to obtain eye tracking measures and directly tested this relationship. Relevant results are copied below.
Recap of results: 1) as expected gaze was less stable in achromats than controls, 2) achromats with more stable gaze did not show more activation in the scotoma projections zone, which we might have observed if fixation instability masks signals in this region 3) Gaze instability was not correlated with pRF size and eccentricity across V1 in achromats. We note that the relationship between nystagmus and visual sampling is complex - patients experience a stable image and may sample only during a specific phase of the eye movement. It is therefore not inherently clear if and how nystagmus affects pRF size.
Relevant Manuscript text incorporating these analyses is copied below.
To quantify eye movement, we used the following methods added to the manuscript:
“Fixation stability
Participants’ gaze was tracked throughout all pRF mapping runs. Collecting reliable gaze data from individuals with nystagmus is a challenge because out of the box calibration procedures mostly fail without stable fixation. To account for this, we implemented a post-hoc custom calibration procedure (Tailor et al., 2021). The eye-tracker was first precalibrated on a typically sighted individual. Then, before every other run, we collected gaze data from a 5-point fixation task (at fixation and above, below, left, and right of fixation at 5 eccentricity). This data allowed us to subsequently map the patient's recorded gaze coordinates to their precise locations on the screen. In 10 out of the 14 achromats we acquired reliable enough data to assess fixation stability.
Calibration data processing: We first removed the first 0.5 seconds for each fixation location to allow for fixation to arrive on the target. We then performed (a) blink removal, (b) filtered out time points with eye movement velocity outliers (±2SD), and (c) filtered out any positions >3SDs to the left or right of the mean fixation location, and >1SD above or below. We took the median of the remaining gaze measurements as an approximate fixation estimate. The resulting 5 median fixation locations were used to fit an affine transformation that remapped the recorded gaze positions into screen space.
Quantifying fixation stability: after applying the transformation of the post-hoc calibration, data was filtered for blinks and extreme velocities (<2SD). For each functional run, fixation instability was measured as the standard deviation of gaze x-positions across 1second windows. Measures were then averaged across the two run repeats.”
We report the resulting new fixation data results as follows:
Results (coverage section):
“Another potential confound in our findings is fixation instability. In pRF mapping, which is usually conducted under photopic (cone-dominant) conditions, unstable fixation can cause a signal drop in the foveal projection zone. As expected due to nystagmus, the achromatopsia group showed higher fixation instability compared to controls (rodselective: t(9.08)=-3.19, p=0.01; non-selective: t<sub<(9.41)=-4.88, p<0.001 degrees-offreedom corrected for unequal-variance; see Supplement Figure S2a). However, several lines of evidence suggest this instability cannot fully account for the lack of "filling in" in achromats. First, within the achromat group, we found no correlation between fixation stability and coverage (rod-selective: spearman-r(8) = -0.36, p=0.31; non-selective spearman-r(8)=0.07,p=0.85); Individuals with more stable, control-like fixation did not show more signal inside the scotoma (see Supplement 2). Second, in adults with achromatopsia, typically with less severe nystagmus (Kohl et al., 1993), two recent studies also found absence of filling in (Anderson et al., 2024; Molz et al., 2023).
So, while we cannot fully exclude nystagmus masking foveal signals in the cortex of some patients, this converging evidence from structural and functional MRI measures across different studies and groups, strongly suggests that the deprived cortex does not substantially ‘fill in’ with peripheral rod inputs in achromatopsia.”
Results (pRF size + eccentricity):
“Larger pRFs indicate that neuronal populations in achromats’ V1 cortex, combine information across larger areas in visual space than in typically sighted controls. This could reflect true neural tuning differences as well as be driven by larger eye movement. However, fixation instability in achromats do not significantly correlate with pRF size in our sample (rod-selective: spearman-r(8) = -0.41, p=0.24; non-selective spearman-r(8)=0.37,p=0.29)
It has been shown that fitting artefacts around scotoma edges, can give rise to similar outward eccentricity shifts (Binda et al., 2013). However, when accounting for fitting artefacts around the foveal scotoma edge by modelling the rod-free zone during pRF fitting, pRF size and eccentricity differences remain unchanged (see Supplement 3). Finally, we found no significant correlations between gaze stability and the eccentricity shift (rod-selective: spearman-r(8) = 0.58, p=0.08; non-selective spearman-r(8)=0.09,p=0.8, Supplement 4D)
Together, these analyses reveal subtle differences in how V1 of achromats responds to rod signals outside the foveal zone, which are consistent with results from other studies (Molz et al. 2023, Anderson et al. 2024). While we found no direct evidence that these are being driven by confounding factors such as eye-movements or fitting artefacts, more work is needed to understand the underlying processes that give rise to these shifts.”
The following text has been added to Supplement 2
“As expected, achromats showed significant higher fixation instability compared to controls (as reported in the main text). We found no significant correlation between fixation instability and either coverage, pRF size, eccentricity in achromats. Results of Spearman R correlations in both rod- and non-selective conditions are reported in the figure. We note that the relationship between nystagmus and visual sampling is complex- patients experience a stable image and may sample only during specific eyemovement phases. It is therefore not fully clear if and how nystagmus should give rise to altered pRFs.”
(b) The authors don't see a clear relationship between coverage and fixation stability. This seems to rest on a few ad hoc examples. (What happens if one plots mean fixation deviation vs. coverage (and sets the individuals who could not be calibrated as the highest value of calibrated fixation deviation. Does a relationship then emerge?).
In any case, I wouldn't expect coverage to be particularly susceptible to eye-movements. If a voxel in the cortex entirely projects to the scotoma then it should be robustly silent. The effects of eye-movements will be to distort the size and eccentricity estimates of voxels that are not entirely silent.
There are many places in the paper where eye-movements might be playing an important role.
Examples include the larger pRF sizes observed in achromats. Are those related to fixation instability?
We thank the reviewer for their comment. As detailed in our previous response, we have now extracted fixation instability data from additional patients and have expanded our discussion of its potential effects throughout the manuscript.
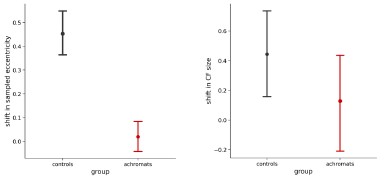
Given that fixation instability is expected to increase pRF size by a fixed amount, that would explain why ratios are close to 1 in V3 (Figure 4).
We agree with the reviewer’s point, that the ratio change on its own is not strong evidence of compensation, this analysis was meant to complement the CF result. The plot in Figure 4 is intended to reconcile the connective field (CF) and pRF results. Its purpose is to illustrate that even though larger pRFs in achromats might seem counterintuitive alongside their smaller V3 CF sizes, the pRF data do not contradict the CF findings but they are in fact consistent with one another. We also agree that there are alternative explanations for the differences in pRF size, such as fixation stability, and we have now added this point to the text.
Results (CF size):
“To understand how this finer cortical sampling in V3 (smaller connective fields) impacts visual processing, we consider its effect on population receptive fields (pRFs). In V1, pRF sizes in achromats were significantly larger than in controls for both stimulus conditions, indicating coarser spatial tuning at the cortical input stage (Figure 4C, left). By selectively sampling from a smaller area of the V1 surface (smaller CFs), V3 can effectively compensate for this coarser input. If so, this process should result in a relative normalisation of pRF size in V3 compared to V1 (Figure 4C, right).
To test this prediction, we plotted the ratio of pRF sizes between achromats and controls, where a value of 1 indicates parity between the groups (Figure 4B). As our compensatory connective field hypothesis predicts, the ratio was closer to 1 in V3 than in V1 across both stimulus conditions, confirming the pRF size difference was significantly reduced at the higher cortical stage. Together this shows converging evidence across the two models (pRF and CF) of hierarchical refinement as a possible compensatory mechanism, where V3's altered connectivity helps to normalize the processing of degraded sensory input from V1.”
Discussion:
“The hierarchical reorganisation observed in V3 is unlikely to be driven by fixation instability. Connective field (CF) estimates are robust to eye movements (Tangtartharakul et al., 2023), because they are anchored to V1 inputs rather than absolute screen position. Considered alone, the pRF results could alternatively be explained by eye movements introducing a fixed size offset that affects smaller V1 pRFs more strongly than those in V3. While we found no evidence for this relationship between pRF size and gaze measures in our patients, we cannot fully rule out the possibility. Nevertheless, the internal consistency between the CF and pRF measures provides a more parsimonious account; that sampling across the hierarchy accounts for coarser tuning at the input stage.”
(2) Topography
The claim of no change in topography is a little confusing given that you do see a change in eccentricity mapping in achromats.
Either this result is real, in which case there *is* a change in topography, albeit subtle, or it's an artifact.
Perhaps these results need a little bit of additional scrutiny.
One reason for concern is that you see different functions relating eccentricity to V1 segments depending on the stimulus. That almost certainly reflects biases in the modelling, not reorganization - the curves of Figure 2D are exactly what Binda et al. predict.
Another reason for concern is that I'm very surprised that you see so little effect of including/not including the scotoma - the differences seem more like what I'd expect from simply repeating the same code twice. (The quickest sanity check is just to increase the size of the estimated scotoma to be even bigger?).
We thank the reviewer for their comment. We have double-checked our scotoma modelling, confirming its correct implementation. The results of the scotoma modelling are not identical to the full one, just similar (see below).
Previous studies on “artificial scotomas” (such as the one reported by Binda et al.) have shown mixed results. While Binda and colleagues found that modelling artificial scotomas normalised pRF shifts, others found no effect (Haak et al. 2012, Prabhakaran et al. 2020). Notably, the rodfree zone in achromatopsia is considerably smaller (~0.5° radius) than most tested artificial scotomas. Moreover, it is unclear whether scotoma modelling is beneficial in clinical populations as artificial scotomas (screen-based masking) are not equivalent to retinal scotomas from inactive photoreceptors. A recent achromatopsia study (Anderson et al. 2024) also found no change in pRF estimates with scotoma modelling.
In our scotoma analyses, we found meaningful differences only in the non-selective condition in controls where cones in the rod-free zone are stimulated - which would be the main expected effect of this modelling exercise (see below). In all other conditions (rod-selective in controls, both conditions in achromats), only rods are stimulated, we found no difference in coverage, eccentricity or pRF size when modelling the scotoma likely because the foveal signal is weak/absent, and did not contribute much to pRF estimates in the unmasked analyses.
This means we cannot account for the eccentricity shift as an edge effect with this scotoma model – but we remain cautious about interpreting it as real. This is because first, as we mention in the paper, in the non-selective condition, which has a higher signal-to-noise ratio, the eccentricity estimates in achromats match those of the control group's rod system. Second, it is still possible that the observed shift is an artefact of modelling that was not accounted for by the approach of scotoma modelling.
Our claim of "no change in topography" specifically referred to the absence of "filling-in" as measured by cortical coverage - the percentage of activated tissue regardless of fitted parameters. However, to avoid confusing given the eccentricity and pRF size results we now rephrased our claim.
Abstract:
“Cortical input stages (V1) exhibited high stability, with input-deprived cortex showing no retinotopic remapping and exhibiting structural hallmarks of deprivation.”
Results (pRF eccentricity):
“It has been shown that fitting artefacts around scotoma edges, can give rise to similar outward eccentricity shifts (Binda et al., 2013). However, when accounting for fitting artefacts around the foveal scotoma edge by modelling the rod-free zone during pRF fitting, pRF size and eccentricity differences remain unchanged (see Supplement 3). Finally, we found no significant correlations between gaze stability and the eccentricity shift (rod-selective: spearman-r(8) = 0.58, p=0.08; non-selective spearman-r(8)=0.09,p=0.8, Supplement 4D)
Together, these analyses reveal subtle differences in how V1 of achromats responds to rod signals outside the foveal zone, which are consistent with results from other studies (Molz et al. 2023, Anderson et al. 2024). While we found no direct evidence that these are being driven by confounding factors such as eye movements or fitting artefacts, more work is needed to understand the underlying processes that give rise to these shifts.”
To better illustrate the effect of scotoma modelling text has been added to Supplement 3:
“Studies on artificial scotomas, where part of the visual field is masked, suggest that pRF estimates of eccentricity and size can be biased by fitting scotoma-edge artefacts, and that these can be mitigated by modelling the scotoma in the pRF fitting procedure (e.g., Binda et al. 2013).
We therefore repeated the pRF modelling procedure with the rod-scotoma being modelled as a black oval mask (1.25°x0.9°) over the stimulus aperture model. As expected, a visible difference between the two models is only apparent in the nonselective condition in controls where the cones in the rod-free zone are being stimulated. In all the other conditions (rod-selective in controls, and both stimulation conditions in achromats) only the rods are stimulated, therefore the masked stimulus still matches the retinal activation, and no major differences can be observed. Performing the same statistical tests applied to the full model in the main text yields equivalent results of equivalent coverage in the rod-selective condition, with equivalent coverage across groups(t(47) = 0.78, p=0.43, BF10=0.31) and controls show a higher coverage in the non-selective stimulation condition compared to achromats (Mann U(52)=141, p<0.01; unequal variance, reverted to non-parametric).
This consistency in pRF properties when modelling the rod scotoma, is in line with previous results from scotoma modelling; While Binda and colleagues found that this normalised pRF shifts, others found no effect (Haak et al. 2012, Prabhakaran et al. 2020). Notably, the rod-free zone in achromatopsia is considerably smaller (~0.5° radius) than most tested artificial scotomas, and as artificial scotomas (screen-based masking) are not equivalent to retinal scotomas from inactive photoreceptors, it is unclear how artificial scotoma findings generalise to clinical populations. Our results are in line with a recent achromatopsia study (Anderson et al. 2024) which also found no change in pRF estimates with scotoma modelling.”
I'd also look at voxels that pass an R2>0.2 threshold for both the non-selective and selective stimulus. Are the pRF sizes the same for both stimuli? Are the eccentricity estimates? If not, that's another clear warning sign.
Comparable results were obtained when using higher R2 thresholds. These results are now included in Supplement 6.
(3) Connective field modelling
Let's imagine a voxel on the edge of the scotoma. It will tend to have a connective field that borders the scotoma, and will be reduced in size (since it will likely exclude the cortical region of V1 that is solely driven by resting state activity). This predicts your rod monochromat data. The interesting question is why this doesn't happen for controls. One possibility is that there is topdown 'predictive' activity that smooths out the border of the scotoma (there's some hint of that in the data), e.g., Masuda and Wandell.
One thing that concerns me is that the smaller connective fields don't make sense intuitively. When there is a visual stimulus, connective fields are predominantly driven by the visual signal. In achromats, there is a large swath of cortex (between 1-2.5 degrees) which shows relatively flat tuning as regards eccentricity. The curves for controls are much steeper, See Figure 2b. This predicts that visually driven connective fields should be larger for achromats. So, what's going on?
The reviewer raises interesting points about the interpretation of our connective field results. The possibility of differential top-down modulation between controls and achromats is intriguing, however it is not supported by the data, if top-down modulation is activating foveal V1 in controls then we shouldn’t see a drop in the amount of significant vertices sampling from the fovea in the rod-selective condition compared to the non-selective, but in fact we do see quite a large drop in the amount of significant vertices in that area in the rod-selective condition. Therefore, at the moment we do not think there is strong basis to assume our data could be explained by achromats lacking top-down predictive activity in the scotoma area that is present in controls.
Regarding the concern about smaller CFs seeming counterintuitive given the flat eccentricity tuning in achromats' V1: we believe there is not a straightforward prediction from pRF properties to CF sizes. The relationship between V1 pRF characteristics and V3 CF sampling is complex and not well-established in the literature, and the two can be decoupled to some degree. For instance, in our data, controls show flat V1 pRF sizes in the rod-selective condition (similar to achromats), yet their V3 CF sizes maintain the typical eccentricity-dependent increase seen in the non-selective condition. This suggests that CF size patterns don't simply mirror V1 pRF properties or visual stimuli responses.
Importantly, CF modelling fundamentally differs from pRF analysis in how it might be affected by scotomas. Unlike pRF analysis where a scotoma creates a "silent" region in visual space, in CF modelling the deprived cortex remains physically present and continues generating neural signals (albeit not visually-driven ones). If V3-V1 connectivity were anatomically fixed, V3 would continue sampling from deprived V1 regions even if they do not produce visual-driven signals. A change in this sampling pattern, as we see in our data, is therefore evidence for plasticity.
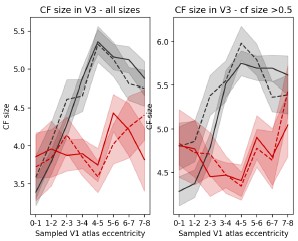
Our data support this interpretation. First, in achromats, the CF size pattern observed cannot be easily explained by scotoma-edge artefacts. V3 vertices sampling from the immediate vicinity of the scotoma (1°-3°) show CF sizes comparable to controls. The effect is only significant further away from the scotoma (4°-6°).
Second, to assess how the presence of a scotoma affects CF measure we can compare the two conditions in the controls, since the rod-selective condition has a scotoma present and the nonselective condition does not. For this purpose, we performed an additional analysis, quantifying on a vertex-by-vertex level the differences in CF fitted parameters between the two stimulation conditions across V1. See results below. In achromats there are no systematic shifts between the stimulation conditions, as expected as both are rod-driven. In controls, this analysis reveals only subtle shifts (~0.45° in the rod-selective condition). CF size has also changed slightly although not significantly different from that observed in achromats. These shifts are much smaller than the CF size and eccentricity differences between controls and achromats, so we consider it unlikely that our findings are driven by scotoma artefacts.
Author response image 1.
Results (CF size):
“The significant CF size differences are unlikely to be a model-fitting bias around a scotoma edge, as V3 vertices sampling from the immediate vicinity of the scotoma (1°3°) show CF sizes comparable to controls. The significant reduction in CF size occurs only further in the periphery (4°-6°), in regions that are primarily stimulus-driven.
To understand how this finer cortical sampling in V3 (smaller connective fields) impacts visual processing, we consider its effect on population receptive fields (pRFs). In V1, pRF sizes in achromats were significantly larger than in controls for both stimulus conditions, indicating coarser spatial tuning at the cortical input stage (Figure 4C, left). By selectively sampling from a smaller area of the V1 surface (smaller CFs), V3 can effectively compensate for this coarser input. If so, this process should result in a relative normalisation of pRF size in V3 compared to V1 (Figure 4C, right).
To test this prediction, we plotted the ratio of pRF sizes between achromats and controls, where a value of 1 indicates parity between the groups (Figure 4B). As our compensatory connective field hypothesis predicts, the ratio was closer to 1 in V3 than in V1 across both stimulus conditions, confirming the pRF size difference was significantly reduced at the higher cortical stage. Together this shows converging evidence across the two models (pRF and CF) of hierarchical refinement as a possible compensatory mechanism, where V3's altered connectivity helps to normalize the processing of degraded sensory input from V1.”
Discussion (added paragraph):
“The hierarchical reorganisation observed in V3 is unlikely to be driven by fixation instability. Connective field (CF) estimates are robust to eye movements (Tangtartharakul et al., 2023), because they are anchored to V1 inputs rather than absolute screen position. Considered alone, the pRF results could alternatively be explained by eye movements introducing a fixed size offset that affects smaller V1 pRFs more strongly than those in V3. While we found no evidence for this relationship between pRF size and gaze measures in our patients, we cannot fully rule out the possibility. Nevertheless, the internal consistency between the CF and pRF measures provides a more parsimonious account; that sampling across the hierarchy accounts for coarser tuning at the input stage.”
The beta parameter is not described (and I believe it can alter connective field sizes).
In Author response image 2, we plot the beta parameter of the pRF modelling in V1 with no R2 filtering, error bars are 95% CIs:
Author response image 2.
The reviewer did not specify how beta might alter connective field sizes. We assume he meant that as in pRF mapping, the slope of activity from deprived to non-deprived cortex will artefactually create a CF model fit with smaller CF sizes. To test this, we calculated the slope of beta values between 0° and 3° in each participant in the rod-selective condition, as this range includes the scotoma and the area at the edge of the scotoma. We then used the slope as a covariate in an ANCOVA when comparing the CF sizes across groups in each sampled V1 segment. Accounting for the beta slope of V1 did not change the reported results. This analysis still shows smaller CF sizes in V3 in the rod-selective conditions between 4°-6° eccentricity – these differences remain significant (p<0.001 for 4°-5° and p<0.05 for 5°-6° when comparing achromats vs controls).
Similarly, it's possible to get very small connective fields, but there wasn't a minimum size described in the thresholding.
CF sizes were fit with a grid fit. Possible values were [0.5,1,2,3,4,5,7,10]. Therefore, the minimum size is 0.5. Filtering out the smallest connective field sizes does not change the results:
Author response image 3.
I might be missing something obvious, but I'm just deeply confused as to how the visual maps and the connectome maps can provide contradictory results given that the connectome maps are predominantly determined by the visual signal. Some intuition would be helpful.
We agree that this appears counterintuitive, and now added further clarification. The two models (pRF and CF) fundamentally differ in what they measure and how they relate to visual processing. V1 pRF sizes reflect the relationship between neural activity and visual stimuli - essentially how much of a visual stimulus drives a voxel's response - while V3 CF sizes reflect how V3 samples from the V1 cortical surface, indicating how many V1 voxels contribute to a V3 voxel's activity.
The measures constrain each other, as a V3 voxel's pRF size is expected to match the pooling of its connected V1 inputs. But they can be decoupled: A V3 voxel could sample from a small area of V1 cortex (a small CF in mm) that happens to represent a large area of visual space if those V1 voxels have large pRFs. The aim of Figure 4B is to clarify that the measures are consistent with one another even though they diverge in direction. In achromats, where V1 voxels have larger pRFs (coarser spatial resolution), V3 appears to compensate by sampling more selectively from V1 via smaller CF sizes. Theoretically, this should reduce the pRF size difference between controls and patients in V3, a prediction that our data supports.
Results (CF size):
“To understand how this finer cortical sampling in V3 (smaller connective fields) impacts visual processing, we consider its effect on population receptive fields (pRFs). In V1, pRF sizes in achromats were significantly larger than in controls for both stimulus conditions, indicating coarser spatial tuning at the cortical input stage (Figure 4C, left). By selectively sampling from a smaller area of the V1 surface (smaller CFs), V3 can effectively compensate for this coarser input. If so, this process should result in a relative normalisation of pRF size in V3 compared to V1 (Figure 4C, right).
To test this prediction, we plotted the ratio of pRF sizes between achromats and controls, where a value of 1 indicates parity between the groups (Figure 4B). As our compensatory connective field hypothesis predicts, the ratio was closer to 1 in V3 than in V1 across both stimulus conditions, confirming the pRF size difference was significantly reduced at the higher cortical stage. Together this shows converging evidence across the two models (pRF and CF) of hierarchical refinement as a possible compensatory mechanism, where V3's altered connectivity helps to normalize the processing of degraded sensory input from V1.”
Discussion (added paragraph):
“The hierarchical reorganisation observed in V3 is unlikely to be driven by fixation instability. Connective field (CF) estimates are robust to eye movements (Tangtartharakul et al., 2023), because they are anchored to V1 inputs rather than absolute screen position. Considered alone, the pRF results could alternatively be explained by eye movements introducing a fixed size offset that affects smaller V1 pRFs more strongly than those in V3. While we found no evidence for this relationship between pRF size and gaze measures in our patients, we cannot fully rule out the possibility. Nevertheless, the internal consistency between the CF and pRF measures provides a more parsimonious account; that sampling across the hierarchy accounts for coarser tuning at the input stage.”
Some analyses might also help provide the reader with insight. For example, doing analyses separately on V3 voxels that project entirely to scotoma regions, project entirely to stimulusdriven regions, and V3 voxels that project to 'mixed' regions.
We agree that it is important to plot the connective field dynamics across the scotoma region.
In Figure 4A we split the V3 vertices based on the V1 area they sample from. Therefore the 0°-1° would be considered as mainly sampling from the “scotoma” region and the higher the eccentricity is, the less “scotoma” it includes. The V3 vertices that have a significantly smaller CF size compared to controls are those sampling from mostly if not entirely stimulusdriven regions 4°-5° and 5°-6°. We are not sure how further binning the data by within, across and outside scotoma would be more informative.
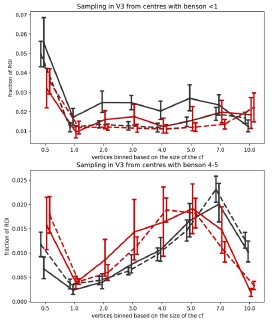
However, in Author response image 4, we plot in more details the distribution of CF sizes sampling from a V1 segment clearly inside and clearly outside the scotoma. The top figure shows the CF size distribution of V3 vertices that sample from a V1 0°-1° segment, where V1 is deprived of input due to the rod scotoma. In achromats, there is a clear drop in vertices with a very small (0.5) CF size. The bottom figure shows the distribution of V3 vertices that sample from the V1 4°-5° segment which falls outside the scotoma and shows a significant difference in CF size across the groups. Here in achromats you can see a drop in larger V3 CF sizes sampling from the V1 region, and an increase in smaller ones (note that this further addresses a previous concern that connective field differences across groups are solely driven by very small CFs).
Author response image 4.
Following the reviewer’s comment we have added the following statement in the results section discussing CF size:
“The significant CF size differences are unlikely to be a model-fitting bias around a scotoma edge, as V3 vertices sampling from the immediate vicinity of the scotoma (1°3°) show CF sizes comparable to controls. The significant reduction in CF size occurs only further in the periphery (4°-6°), in regions that are primarily stimulus-driven.”
The finding that pRF sizes are larger in achromats by a constant factor as a function of eccentricity is what differences in eye-movements would predict. It would be worth examining the relationship between pRF sizes and fixation stability.
We found no relationship between fixation stability and pRF size in V1, although as we explain in response to an earlier point, this does not fully exclude the reviewers alterative explanation, which we now add to the discussion.
Discussion:
“The hierarchical reorganisation observed in V3 is unlikely to be driven by fixation instability. Connective field (CF) estimates are robust to eye movements (Tangtartharakul et al., 2023), because they are anchored to V1 inputs rather than absolute screen position. Considered alone, the pRF results could alternatively be explained by eye movements introducing a fixed size offset that affects smaller V1 pRFs more strongly than those in V3. While we found no evidence for this relationship between pRF size and gaze measures in our patients, we cannot fully rule out the possibility. Nevertheless, the internal consistency between the CF and pRF measures provides a more parsimonious account; that sampling across the hierarchy accounts for coarser tuning at the input stage.”
Reviewer #2 (Public review):
Summary:
The authors inspect the stability and compensatory plasticity in the retinotopic mapping in patients with congenital achromatopsia. They report an increased cortical thickness in central (eccentricities 0-2 deg) in V1 and the expansion of this effect to V2 (trend) and V3 in a cohort with an average age of adolescents.
In analyzing the receptive fields, they show that V1 had increased receptive field sizes in achromats, but there were no clear signs of reorganization filling in the rod-free area. In contrast, V3 showed an altered readout of V1 receptive fields. V3 of achromats oversampled the receptive fields bordering the rod-free zone, presumably to compensate and arrive at similar receptive fields as in the controls.
These findings support a retention of peripheral-V1 connectivity, but a reorganization of later hierarchical stages of the visual system to compensate for the loss, highlighting a balance between stability and compensation in different stages of the visual hierarchy.
Strengths:
The experiment is carefully analyzed, and the data convey a clear and interesting message about the capacities of plasticity.
Weaknesses:
The existence of unstable fixation and nystagmus in the patient group is alluded to, but not quantified or modeled out in the analyses. The authors may want to address this possible confound with a quantitative approach.
We have responded to this in the “Recommendations for the authors” section of this reviewer, as they included a more detailed description of these points there.
Recommendations for the authors:
Reviewer #1 (Recommendations for the authors):
(1) I think the term rod monochromats should be included early in the paper since it's a more intuitive term to describe this population.
We agree with the reviewer that the term “rod monochromats” is more intuitive as it clarifies the retinal source of the disease but have chosen the term achromats for consistency with a wide literature of published work in this group, including our own and our close collaborators’. To clarify, in the first mention of the group as achromats in the introduction we have now added this term:
“Achromatopsia (also known as rod monochromacy) causes cone photoreceptors in the retina to be inactive from birth (Aboshiha et al., 2014).”
(2) The paper essentially contains two definitions of 'eccentricity'. One (atlas/segments) comes from the Benson atlas and the other (functional) comes from pRF mapping. It would be good to make this distinction terminology clearer earlier in the paper. It would also be good to use more consistent terminology. I assume 'sampled atlas V1 eccentricity' in 3A is the same as 'V1 segment' in 1A?
For consistency we have now referred to these as V1 segment and sampled V1 segment in the figures when describing the atlas-based definition, and eccentricity for the measured pRF-based eccentricity.
(3) The 'stability vs. plasticity' framing in the introduction could be tightened slightly.
We have made the following changes following the reviewer’s comment:
“In the visual domain, the focal point of the debate on plasticity and stability has hinged on the extent to which retinal input deprivation can drive local reorganisation in early visual cortex, for example, for deprived tissue to take on inputs from spared retinal locations (Adams et al., 2007; Baker et al., 2005, 2008; Baseler et al., 2002, 2011; Calford et al., 2005; Dilks et al., 2009; Dumoulin & Knapen, 2018; Ferreira et al., 2016; Goesaert et al., 2014; Haak et al., 2015; Molz et al., 2023; Ritter et al., 2019; Schumacher et al., 2008). In reality visual impairment is a more global phenomenon, affecting all levels of visual processing, with complex dynamics beyond constricted local retinocortical projection zones(Carvalho et al., 2019).”
(4) Figure 1A, define the x axis as degrees.
We have now added the ° sign to all the tick labels indicating Benson map eccentricity.
(5) Figure 2B, is there room for pictures of the silent substitution/standard stimulus
We have now added images in a Supplement 5 to avoid cluttering the main Figure 2B
(6) Figure 2
Panel A has a slightly weird organization. The reader is supposed to compare the square symbols to each other, and the circles to each other, why not organize the figure so they are adjacent in the graph (i.e. non selective control, non-selective achromat, selective control, selective achromat)? That also helps the reader orient that in the non-selective conditions you have almost complete pRF coverage.
We have taken on the reviewer’s suggestion and changed the order.
In the inset, maybe use empty symbols? That's the traditional way to say that the square/circle applies to both red and black.
We prefer the current format.
Figure 2C - the symbols change to circles? Why not keep the symbols of A?
We have now changed the symbols of 2C&D.
I'd put the non-selective maps above the selective maps?
We appreciate the feedback but prefer to keep it as it is, as we feel the critical point is conveyed by the rod maps.
(7) 'We propose a new hierarchical model of neural adaptation'. These ideas are hardly new. There are also other models, that would explain your data (cumulative plasticity) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5953572/
We thank the reviewer for the reference. We have now cited it in our discussion and removed the word “new” form the mentioned sentence.
“Therefore, there is theoretically broader scope for experience-dependent reweighting of inputs (Beyeler et al., 2017; Makin & Krakauer, 2023) and to optimise use of inputs that are still available, more reliable, or more relevant in the impaired system. Conversely, higher-order visual areas may appear more plastic simply because they integrate the cumulative effects of learning from multiple lower stages (Beyeler et al., 2017).”
We propose a hierarchical model of neural adaptation…” [deleted the word new]
(8) Line 508. No image of the stimulus is contained in the paper
Corrected
(9) Line 620. I believe the Figure is 1B, not 1C.
Corrected
(10) Figure 4A. CF Size - add mm2 to the axes.
Corrected
Reviewer #2 (Recommendations for the authors):
I am not an expert on pRF mapping, and as such, I am unsure how to relate to pRF mapping performed in patients with unstable fixation (not quantified, but referred to) and nystagmus, such as the achromatic population here. Since the majority of the results hinge on this analysis, I would appreciate more data about the differences between the groups. Supplement 2, which is meant to speak to this, shows only the data from 3 typical participants, and in itself is not evidence for "no correlation between stable fixation and enhanced foveal". Additionally, I'd appreciate a clear methods explanation of how the authors address these confounds; this is too important a concern to be left for the discussion section.
We agree with the reviewer that eye movements could affect pRF measures. We have now also included data for all participants where we were able to obtain eye tracking measures and directly tested this relationship. Relevant results are copied below.
Recap of results: 1) as expected gaze was less stable in achromats than controls, 2) achromats with more stable gaze did not show more activation in the scotoma projections zone, which we might have observed if fixation instability masks signals in this region 3) Gaze instability was not correlated with pRF size and eccentricity across V1 in achromats. We note that the relationship between nystagmus and visual sampling is complex - patients experience a stable image and may sample only during a specific phase of the eye movement. It is therefore not inherently clear if and how nystagmus affects pRF size.
Relevant Manuscript text incorporating these analyses is copied below.
To quantify eye movement, we used the following methods added to the manuscript:
“Fixation stability
Participants’ gaze was tracked throughout all pRF mapping runs. Collecting reliable gaze data from individuals with nystagmus is a challenge because out of the box calibration procedures mostly fail without stable fixation. To account for this, we implemented a post-hoc custom calibration procedure (Tailor et al., 2021). The eye-tracker was first precalibrated on a typically sighted individual. Then, before every other run, we collected gaze data from a 5-point fixation task (at fixation and above, below, left, and right of fixation at 5 eccentricity). This data allowed us to subsequently map the patient's recorded gaze coordinates to their precise locations on the screen. In 10 out of the 14 achromats we acquired reliable enough data to assess fixation stability.
Calibration data processing: We first removed the first 0.5 seconds for each fixation location to allow for fixation to arrive on the target. We then performed (a) blink removal, (b) filtered out time points with eye movement velocity outliers (±2SD), and (c) filtered out any positions >3SDs to the left or right of the mean fixation location, and >1SD above or below. We took the median of the remaining gaze measurements as an approximate fixation estimate. The resulting 5 median fixation locations were used to fit an affine transformation that remapped the recorded gaze positions into screen space.
Quantifying fixation stability: after applying the transformation of the post-hoc calibration, data was filtered for blinks and extreme velocities (<2SD). For each functional run, fixation instability was measured as the standard deviation of gaze x-positions across 1second windows. Measures when then averaged across the two run repeats.”
Results (coverage section):
“Another potential confound in our findings is fixation instability. In pRF mapping, which is usually conducted under photopic (cone-dominant) conditions, unstable fixation can cause a signal drop in the foveal projection zone. As expected due to nystagmus, the achromatopsia group showed higher fixation instability compared to controls (rodselective: t(9.08)=-3.19, p=0.01; non-selective: t<sub<(9.41)=-4.88, p<0.001 degrees-offreedom corrected for unequal-variance; see Supplement Figure S2a). However, several lines of evidence suggest this instability cannot fully account for the lack of "filling in" in achromats. First, within the achromat group, we found no correlation between fixation stability and coverage (rod-selective: spearman-r(8) = -0.36, p=0.31; non-selective spearman-r(8)=0.07,p=0.85); Individuals with more stable, control-like fixation did not show more signal inside the scotoma (see Supplement 2). Second, in adults with achromatopsia, typically with less severe nystagmus (Kohl et al., 1993), two recent studies also found absence of filling in (Anderson et al., 2024; Molz et al., 2023).
So, while we cannot fully exclude nystagmus masking foveal signals in the cortex of some patients, this converging evidence from structural and functional MRI measures across different studies and groups, strongly suggests that the deprived cortex does not substantially ‘fill in’ with peripheral rod inputs in achromatopsia.”
Results (pRF size + eccentricity):
“Larger pRFs indicate that neuronal populations in achromats’ V1 cortex, combine information across larger areas in visual space than in typically sighted controls. This could reflect true neural tuning differences as well as be driven by larger eye movement. However, fixation instability in achromats do not significantly correlate with pRF size in our sample (rod-selective: spearman-r(8) = -0.41, p=0.24; non-selective spearman-r(8)=0.37,p=0.29)
It has been shown that fitting artefacts around scotoma edges, can give rise to similar outward eccentricity shifts (Binda et al., 2013). However, when accounting for fitting artefacts around the foveal scotoma edge by modelling the rod-free zone during pRF fitting, pRF size and eccentricity differences remain unchanged (see Supplement 3). Finally, we found no significant correlations between gaze stability and the eccentricity shift (rod-selective: spearman-r(8) = 0.58, p=0.08; non-selective spearman-r(8)=0.09,p=0.8, Supplement 4D)
Together, these analyses reveal subtle differences in how V1 of achromats responds to rod signals outside the foveal zone, which are consistent with results from other studies (Molz et al. 2023, Anderson et al. 2024). While we found no direct evidence that these are being driven by confounding factors such as eye-movements or fitting artefacts, more work is needed to understand the underlying processes that give rise to these shifts.”
The following text has been added to Supplement 2
“As expected, achromats showed significant higher fixation instability compared to controls (as reported in the main text). We found no significant correlation between fixation instability and either coverage, pRF size, eccentricity in achromats. Results of Spearman R correlations in both rod- and non-selective conditions are reported in the figure. We note that the relationship between nystagmus and visual sampling is complex- patients experience a stable image and may sample only during specific eyemovement phases. It is therefore not fully clear if and how nystagmus should give rise to altered pRFs.”
The field connectivity analysis similarly seems to be used only on task data from the same design; if it was replicated from resting-state data, that would be a good way to show consistency which is independent of measures requiring fixation.
We agree that resting-state data would be valuable; however, we did not collect such data in these individuals due to time limitations. Instead, we demonstrate the consistency and reliability of our results by replicating our findings across two different stimulation conditions (rod-selective and non-selective), which differ in luminance, contrast and signal amplitude in both groups and for controls also in the photoreceptors involved. The convergence of results across these distinct visual conditions strengthens our confidence in the reliability of the observed effects. Also, notably, CF estimates have been shown to be robust to large eye movements, and therefore also to differences in fixation stability across groups (Tangtartharakul et al., 2023).
The authors may want to contextualize their findings in relation to what reorganization exists in cases of late-onset loss of part of the visual field on one hand (stroke recovery), and in the case of complete blindness from early life on the other, as both speak to different levels of plasticity the visual system is capable of.
We thank the reviewer for their comment and have added a new paragraph discussing this topic.
Discussion:
“Our findings on hierarchical adaptation have broader implications for other visual disorders, depending on their timing and nature. For instance, a central scotoma acquired in adulthood, as in macular degeneration, may not trigger the same V3 sampling shifts (Haak et al., 2016), suggesting a sensitive window for this form of plasticity, after which connective fields remain more stable. This also raises questions about congenital blindness, where the absence of any driving input could lead to weakening or repurposing of hierarchical connections (Saccone et al., 2024). Moreover, principles may differ between a deprived but structurally intact cortex, as in retinal dystrophies, and a physically damaged cortex, as in stroke. In the latter, more extensive reorganisation may be required to sample effectively from surviving, and potentially disparate, regions of V1. Perceptual training effects in stroke rehabilitation may reflect such dynamics (Cavanaugh et al., 2025; Elshout et al., 2021).”
A more minor point: Can the authors clarify what the dark adaptation is used for, and provide the supplementary analysis showing that the duration difference for some of the participants didn't impact the results (stated but not shown).
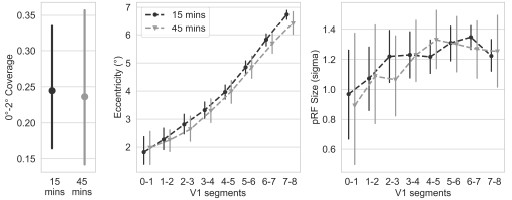
The dark adaptation period before the rod-selective condition allowed rod photoreceptors to recover from bleaching caused by prior mesopic light exposure, ensuring optimal rod sensitivity under scotopic conditions. To verify that our 15-minute adaptation period was sufficient, we tested 10 control participants with an extended 45-minute adaptation period. As we found no differences in the resulting rod maps between standard and extended adaptation protocols, these participants were combined with the main control group for all analyses. Author response image 5 are the plots for the two dark adaptation periods.
Author response image 5.
-

Author response:
We would like to thank the reviewers for their positive evaluation of our work, and their comments inspiring useful discussion. We will provide an in-depth response once one of the key authors has returned from parental leave (in some months), but below we share initial thoughts:
Both reviewers asked to see more gaze data to understand how eye movements in patients with achromatopsia might drive our results. We will expand our analyses of eye tracking data and discuss the implications in more depth, but would like to note that our key findings (no change in signal coverage in the foveal rod-scotoma projection zone in achromats, and changes in connective fields) are both robust to eye movement, and unlikely to be driven by gaze differences. Where this is less clear (i.e., population Receptive Field eccentricities are …
Author response:
We would like to thank the reviewers for their positive evaluation of our work, and their comments inspiring useful discussion. We will provide an in-depth response once one of the key authors has returned from parental leave (in some months), but below we share initial thoughts:
Both reviewers asked to see more gaze data to understand how eye movements in patients with achromatopsia might drive our results. We will expand our analyses of eye tracking data and discuss the implications in more depth, but would like to note that our key findings (no change in signal coverage in the foveal rod-scotoma projection zone in achromats, and changes in connective fields) are both robust to eye movement, and unlikely to be driven by gaze differences. Where this is less clear (i.e., population Receptive Field eccentricities are shifted outwards and increased in size), we have highlighted this and avoided drawing strong conclusions.
Reviewer 1 questioned why smaller connective fields (CFs) were observed in achromats, suggesting that their flatter V1 eccentricity tuning should predict larger CFs. It’s not straightforward to predict how V1's population receptive field (pRF) tuning profile shapes V3's sampling extent, as CFs are driven, but not dictated by V1 - they combine and integrate V1 signals. As we’re dealing with an atypically developed visual system, assumptions about expected relationships are complicated further. We believe that the most relevant aspect of pRF data to the interpretability of V3 CF extent, is the ratio between V1 and V3 pRF sizes. Our outcomes show that pRF sizes in achromats, while larger in V1, are more normalized in V3, predicting more local V3 sampling from V1. This is what our quantifications of CF size show across two independent measures with different stimuli. We will provide further data to address reviewer 1's various queries about the potential causes of the pRF eccentricity shifts in achromats, the relationship between pRFs and CFs, and methodological details of CF fits.
We thank the reviewers again for their insightful comments and look forward to providing more comprehensive responses to their queries substantiated with data as soon as possible.
-

-

eLife Assessment
This important study examines the stability and compensatory plasticity in the retinotopic mapping in patients with congenital achromatopsia. It provides convincing evidence for a stable mapping of the visual field in V1, alongside changes of the readout from V1 into V3, which shows revised receptive field location and size. With the controlling for potential confounding variables, this paper would be of interest to scientists studying the visual system, brain plasticity, and development.
-

Reviewer #1 (Public review):
Summary:
This paper examines plasticity in early cortical (V1-V3) areas in an impressively large number of rod monochromats (individuals with achromatopia). The paper examines three things:
(1) Cortical thickness. It is now well established that early complete blindness leads to increases in cortical thickness. This paper shows increased thickness confined to the foveal projection zone within achromats. This paper replicates the work by Molz (2022) and Lowndes (2021), but the detailed mapping of cortical thickness as a function of eccentricity and the inclusion of higher visual areas is particularly elegant.
(2) Failure to show largescale reorganization of early visual areas using retinotopic mapping. This is a replication of a very recent study by Molz et al. but I believe, given anatomical variability (and …
Reviewer #1 (Public review):
Summary:
This paper examines plasticity in early cortical (V1-V3) areas in an impressively large number of rod monochromats (individuals with achromatopia). The paper examines three things:
(1) Cortical thickness. It is now well established that early complete blindness leads to increases in cortical thickness. This paper shows increased thickness confined to the foveal projection zone within achromats. This paper replicates the work by Molz (2022) and Lowndes (2021), but the detailed mapping of cortical thickness as a function of eccentricity and the inclusion of higher visual areas is particularly elegant.
(2) Failure to show largescale reorganization of early visual areas using retinotopic mapping. This is a replication of a very recent study by Molz et al. but I believe, given anatomical variability (and the very large n in this study) and how susceptible pRF findings are to small changes in procedure, this replication is also of interest.
(3) Connective field modelling, examining the connections between V3-V1. The paper finds changes in the pattern of connections, and smaller connective fields in individuals with achromatopsia than normally sighted controls, and suggests that these reflect compensatory plasticity, with V3 compensating for the lower resolution V1 signal in individuals with achromatopsia.
Strengths:
This is a carefully done study (both in terms of data collection and analysis) that is an impressive amount of work. I have a number of methodological comments but I hope they will be considered as constructive engagement - this work is highly technical with a large number of factors to consider.
Weaknesses:
(1) Effects of eye-movements
I have some concerns with how the effects of eye-movements are being examined. There are two main reasons the authors give for excluding eye-movements as a factor in their results. Both explanations have limitations.
a) The first is that R2 values are similar across groups in the foveal confluence. This is fine as far as it goes, but R2 values are going to be low in that region. So this shows that eye-movements don't affect coverage (the number of voxels that generate a reliable pRF), but doesn't show that eye-movements aren't impacting their other measures.
b) The authors don't see a clear relationship between coverage and fixation stability. This seems to rest on a few ad hoc examples. (What happens if one plots mean fixation deviation vs. coverage (and sets the individuals who could not be calibrated as the highest value of calibrated fixation deviation. Does a relationship then emerge?).
In any case, I wouldn't expect coverage to be particularly susceptible to eye-movements. If a voxel in the cortex entirely projects to the scotoma then it should be robustly silent. The effects of eye-movements will be to distort the size and eccentricity estimates of voxels that are not entirely silent.
There are many places in the paper where eye-movements might be playing an important role.
Examples include the larger pRF sizes observed in achromats. Are those related to fixation instability? Given that fixation instability is expected to increase pRF size by a fixed amount, that would explain why ratios are close to 1 in V3 (Figure 4).
(2) Topography
The claim of no change in topography is a little confusing given that you do see a change in eccentricity mapping in achromats.
Either this result is real, in which case there *is* a change in topography, albeit subtle, or it's an artifact.
Perhaps these results need a little bit of additional scrutiny.
One reason for concern is that you see different functions relating eccentricity to V1 segments depending on the stimulus. That almost certainly reflects biases in the modelling, not reorganization - the curves of Figure 2D are exactly what Binda et al. predict.
Another reason for concern is that I'm very surprised that you see so little effect of including/not including the scotoma - the differences seem more like what I'd expect from simply repeating the same code twice. (The quickest sanity check is just to increase the size of the estimated scotoma to be even bigger?).
I'd also look at voxels that pass an R2>0.2 threshold for both the non-selective and selective stimulus. Are the pRF sizes the same for both stimuli? Are the eccentricity estimates? If not, that's another clear warning sign.
(3) Connective field modelling
Let's imagine a voxel on the edge of the scotoma. It will tend to have a connective field that borders the scotoma, and will be reduced in size (since it will likely exclude the cortical region of V1 that is solely driven by resting state activity). This predicts your rod monochromat data. The interesting question is why this doesn't happen for controls. One possibility is that there is top-down 'predictive' activity that smooths out the border of the scotoma (there's some hint of that in the data), e.g., Masuda and Wandell.
One thing that concerns me is that the smaller connective fields don't make sense intuitively. When there is a visual stimulus, connective fields are predominantly driven by the visual signal. In achromats, there is a large swath of cortex (between 1-2.5 degrees) which shows relatively flat tuning as regards eccentricity. The curves for controls are much steeper, See Figure 2b. This predicts that visually driven connective fields should be larger for achromats. So, what's going on? The beta parameter is not described (and I believe it can alter connective field sizes). Similarly, it's possible to get very small connective fields, but there wasn't a minimum size described in the thresholding. I might be missing something obvious, but I'm just deeply confused as to how the visual maps and the connectome maps can provide contradictory results given that the connectome maps are predominantly determined by the visual signal. Some intuition would be helpful.
Some analyses might also help provide the reader with insight. For example, doing analyses separately on V3 voxels that project entirely to scotoma regions, project entirely to stimulus-driven regions, and V3 voxels that project to 'mixed' regions.
The finding that pRF sizes are larger in achromats by a constant factor as a function of eccentricity is what differences in eye-movements would predict. It would be worth examining the relationship between pRF sizes and fixation stability.
-

Reviewer #2 (Public review):
Summary:
The authors inspect the stability and compensatory plasticity in the retinotopic mapping in patients with congenital achromatopsia. They report an increased cortical thickness in central (eccentricities 0-2 deg) in V1 and the expansion of this effect to V2 (trend) and V3 in a cohort with an average age of adolescents.
In analyzing the receptive fields, they show that V1 had increased receptive field sizes in achromats, but there were no clear signs of reorganization filling in the rod-free area.
In contrast, V3 showed an altered readout of V1 receptive fields. V3 of achromats oversampled the receptive fields bordering the rod-free zone, presumably to compensate and arrive at similar receptive fields as in the controls.These findings support a retention of peripheral-V1 connectivity, but a …
Reviewer #2 (Public review):
Summary:
The authors inspect the stability and compensatory plasticity in the retinotopic mapping in patients with congenital achromatopsia. They report an increased cortical thickness in central (eccentricities 0-2 deg) in V1 and the expansion of this effect to V2 (trend) and V3 in a cohort with an average age of adolescents.
In analyzing the receptive fields, they show that V1 had increased receptive field sizes in achromats, but there were no clear signs of reorganization filling in the rod-free area.
In contrast, V3 showed an altered readout of V1 receptive fields. V3 of achromats oversampled the receptive fields bordering the rod-free zone, presumably to compensate and arrive at similar receptive fields as in the controls.These findings support a retention of peripheral-V1 connectivity, but a reorganization of later hierarchical stages of the visual system to compensate for the loss, highlighting a balance between stability and compensation in different stages of the visual hierarchy.
Strengths:
The experiment is carefully analyzed, and the data convey a clear and interesting message about the capacities of plasticity.
Weaknesses:
The existence of unstable fixation and nystagmus in the patient group is alluded to, but not quantified or modeled out in the analyses. The authors may want to address this possible confound with a quantitative approach.
-