Drowning a frog respiratory rhythm generator in a wash of excitation: State-dependent architecture of a ventilatory oscillator
Curation statements for this article:-
Curated by eLife
eLife Assessment
Using isolated frog brainstem preparations, pharmacological manipulation of excitability, systematic extracellular unit mapping, and focal microinjections, this study provides important findings on whether the buccal rhythm generator is a discrete anatomical nucleus or a distributed, state-dependent network. The question is conceptually significant and of interest to researchers working within respiratory neurobiology and rhythmogenicity in general, and the preparation and experimental strategy are generally appropriate. However, the evidence for the strongest architectural claims is incomplete, with pseudoreplication in pooled unit-mapping analyses, inconsistent statistical reporting, and limited controls in necessity/sufficiency experiments. Overall, although data are largely convincing, substantial revision and more nuanced interpretation of the results are required before claims of state-dependent architectural reorganization can be considered well-supported.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
Breathing begins at birth and continues to death, providing a unique system to understand the architecture of rhythm-generating circuits in vertebrates. In mammals and frogs, three discrete rhythmogenic brainstem nuclei appear essential for generating different phases of breathing, with individual neurons intrinsically tuned to each phase. This discrete multi-oscillator architecture differs fundamentally from the distributed spinal locomotor networks, where rhythmogenesis spans multiple segments and the active phase of individual neurons depends on network state. However, whether these discrete respiratory oscillators are strictly essential and whether the phase of their neurons remains constant across states is coming under increasing scrutiny.
Here, we focus on the frog buccal oscillator, which shares anatomical and functional similarities with the mammalian preBötzinger Complex. We developed a technique to silence motor neurons in frogs and mapped premotor unit activity within and surrounding the putative buccal oscillator under standard conditions and during heightened network excitability. We found that increasing network excitation caused the rhythmically active area to expand, in part by converting premotor lung units into buccal units.
Next, we located the buccal area using excitatory and inhibitory microinjections and established the minimum number of inhibitory injections required to suppress buccal bursts. We then increased network excitability and again tested whether inhibitory microinjections could suppress the rhythm. While inhibitory microinjections could reverse local excitatory effects, they were unable to abolish the buccal rhythm under elevated excitation.
Our data shows that not only does the region containing rhythmic premotor buccal units enlarge as network excitation increases, but the essential requirement for a discrete, rudimentary buccal oscillator is also lost. Thus, although rostral-caudal segments of the frog brainstem generally contribute to discrete phases of ventilatory motor patterns, our findings suggest that oscillator identity should be refined: oscillators should be viewed as promiscuous flexible functional entities that expand or contract to occupy different extents of the brainstem, rather than as fixed anatomical nuclei.
Key points summary
-
The respiratory rhythm is hypothesized to be generated by specialized brainstem circuits comprised of individual non-redundant, discrete, and intrinsically rhythmic oscillators.
-
These oscillators are essential in the impoverished network, often generating rhythmic output in isolation. The inspiratory preBötzinger Complex (preBötC) is the most important of these oscillators in mammals.
-
Isolated brainstem preparations in frogs have revealed a ventilatory rhythm-generating circuit closely resembling that of mammals. Particularly, the Buccal Area shares anatomical and functional similarities to the preBötC.
-
We demonstrate that the discrete Buccal Area is rearranged with mild increases in network excitation to a diffuse network that appears to generate the buccal rhythm instead.
-
These findings support the broader hypothesis that respiratory rhythm-generating circuits can switch to being diffuse and redundant, with discrete oscillators quickly drowning in a sea of excitation.
Article activity feed
-

eLife Assessment
Using isolated frog brainstem preparations, pharmacological manipulation of excitability, systematic extracellular unit mapping, and focal microinjections, this study provides important findings on whether the buccal rhythm generator is a discrete anatomical nucleus or a distributed, state-dependent network. The question is conceptually significant and of interest to researchers working within respiratory neurobiology and rhythmogenicity in general, and the preparation and experimental strategy are generally appropriate. However, the evidence for the strongest architectural claims is incomplete, with pseudoreplication in pooled unit-mapping analyses, inconsistent statistical reporting, and limited controls in necessity/sufficiency experiments. Overall, although data are largely convincing, substantial revision and more …
eLife Assessment
Using isolated frog brainstem preparations, pharmacological manipulation of excitability, systematic extracellular unit mapping, and focal microinjections, this study provides important findings on whether the buccal rhythm generator is a discrete anatomical nucleus or a distributed, state-dependent network. The question is conceptually significant and of interest to researchers working within respiratory neurobiology and rhythmogenicity in general, and the preparation and experimental strategy are generally appropriate. However, the evidence for the strongest architectural claims is incomplete, with pseudoreplication in pooled unit-mapping analyses, inconsistent statistical reporting, and limited controls in necessity/sufficiency experiments. Overall, although data are largely convincing, substantial revision and more nuanced interpretation of the results are required before claims of state-dependent architectural reorganization can be considered well-supported.
-

Reviewer #1 (Public review):
Summary:
The authors test whether the frog buccal ventilatory rhythm generator behaves as a discrete, anatomically localized oscillator or as a distributed, state-dependent network. They combine reduced preparations (segment/subsegment work), systematic extracellular unit surveys over a defined grid, and local AMPA/GABA microinjections in a hemisected brainstem preparation. Based on these approaches, the authors conclude that mild global excitation (bath AMPA) broadens the distribution of rhythmically active units and renders a previously defined "buccal area" functionally non-identifiable as a unique necessary/sufficient locus.
The central idea is plausible, and the overall experimental strategy is appropriate for the question being asked. However, in its current form, the manuscript overstates the strength …
Reviewer #1 (Public review):
Summary:
The authors test whether the frog buccal ventilatory rhythm generator behaves as a discrete, anatomically localized oscillator or as a distributed, state-dependent network. They combine reduced preparations (segment/subsegment work), systematic extracellular unit surveys over a defined grid, and local AMPA/GABA microinjections in a hemisected brainstem preparation. Based on these approaches, the authors conclude that mild global excitation (bath AMPA) broadens the distribution of rhythmically active units and renders a previously defined "buccal area" functionally non-identifiable as a unique necessary/sufficient locus.
The central idea is plausible, and the overall experimental strategy is appropriate for the question being asked. However, in its current form, the manuscript overstates the strength of inference supporting the "expansion" and "loss of necessity/sufficiency" conclusions. This is primarily due to (a) statistical treatment of unit-mapping data that does not respect clustering by preparation/animal, (b) inconsistent statistical reporting across sections, and (c) limited interpretability of focal inhibitory perturbations under a globally excited state.
Strengths:
(1) The manuscript addresses a clear mechanistic question with broader relevance: whether rhythm generation is best conceptualized as a localized kernel or as an emergent distributed property that changes with excitatory state.
(2) The authors use convergent approaches (reduced preparations, mapping, and necessity/sufficiency-style pharmacological perturbations), which is appropriate for circuit-level inference.
(3) A strong element is the within-unit analysis supporting state-dependent changes in phase coupling for a subset of units ("lung" units adopting a buccal-like pattern). The authors' offline PCA-based spike sorting (with cluster-quality selection via silhouette score) provides some reassurance that the reported pre/post injection changes are not simply driven by unit misidentification.
Weaknesses:
(1) Pseudoreplication in unit-survey statistics undermines the main mapping inference. The Methods state that "Units were pooled from multiple preparations" and that chi-squared tests were used to compare proportions across conditions (baseline vs 60 nM AMPA). The Results similarly report proportion changes (e.g., 110 units pooled from three preparations vs 137 units pooled from three additional animals) analyzed with chi-squared tests. Because many units come from the same preparation/animal, independence is unlikely to hold; therefore, inference about state-dependent reorganization at the systems level should be made at the preparation/animal level or via hierarchical models that explicitly account for clustering.
(2) Statistical methods are inconsistently described and need harmonization. In the segment dose-response "Analysis," values are described as compared to zero using a "One-sample t-test." Yet Table 1 is titled as using a "Wilcoxon One-sample Test." These discrepancies must be resolved throughout (Methods, Results, figure legends, and tables), including clear reporting of the unit of n and exact test statistics.
(3) Unit classification and operational definitions raise interpretational concerns. The unit classification scheme defines "buccal units" as those firing during buccal bursts as well as lung bursts, and explicitly notes that "no units were found which fired only during buccal bursts." This is a consequential result, and it currently reads more like a limitation of detection/classification (or state-space sampled) than a robust biological conclusion. Without additional evidence, it weakens claims about a distinct buccal rhythmogenic module and complicates the interpretation of "buccal identity" changes under excitation.
(4) Microinjection mapping: high exclusion rate and alternative explanations for 'loss of necessity' under excitation. The manuscript reports that 15 experiments were conducted, but 9 were excluded because the buccal area was not found or the preparation was "overdriven." This exclusion rate is too high to leave implicit; it raises concerns about selection bias and demands transparent accounting. Moreover, under baseline conditions, GABA (or AMPA-GABA) microinjections reliably reduce/abolish buccal bursts, but under bath 60 nM AMPA, the same injections produce no significant change in instantaneous frequency. This pattern can be interpreted as network redistribution, but it can also reflect state-dependent changes in gain, dynamic range, or local pharmacological impact (e.g., inhibition being comparatively underpowered in the globally excited state). Additional controls/analyses are required to distinguish these explanations.
-

Reviewer #2 (Public review):
Summary:
In this manuscript, the authors investigate the response of the amphibian respiratory rhythm generator under varying excitability conditions. They use pharmacological agents to increase and/ or decrease synaptic excitability and demonstrate the resilience of buccal rhythms under different conditions. They employ these results to formulate their primary thesis, that there is no obligatory locus of the buccal respiratory rhythm in the frog, and that their respiratory rhythmogenic mechanisms should be considered diffuse and anatomically distributed across a larger brainstem region.
Strengths:
This manuscript is well written, with a sufficiently large number of experiments, for which the authors should be congratulated.
Weaknesses:
The presented results don't support the authors' main conclusions, and …
Reviewer #2 (Public review):
Summary:
In this manuscript, the authors investigate the response of the amphibian respiratory rhythm generator under varying excitability conditions. They use pharmacological agents to increase and/ or decrease synaptic excitability and demonstrate the resilience of buccal rhythms under different conditions. They employ these results to formulate their primary thesis, that there is no obligatory locus of the buccal respiratory rhythm in the frog, and that their respiratory rhythmogenic mechanisms should be considered diffuse and anatomically distributed across a larger brainstem region.
Strengths:
This manuscript is well written, with a sufficiently large number of experiments, for which the authors should be congratulated.
Weaknesses:
The presented results don't support the authors' main conclusions, and the interpretation of the data is heavily biased toward their hypothesis. This impregnates an unsubstantiated narrative in the Abstract, Introduction, and Discussion of this manuscript, which must be reexamined with the following points in consideration:
(1) The authors seem to confuse degeneracy with redundancy. For instance, at line 54, they state, "These findings support the broader hypothesis that respiratory rhythm-generating circuits can switch to being diffuse and redundant, with discrete oscillators quickly drowning in a sea of excitations."
Redundancy means having the same component repeated multiple times to buffer the failure of any single component, whereas degeneracy means different functional components that compensate for one another under perturbations (Goaillard and Marder, ARN 2021)
Since the premotor-lung units get converted to buccal units under high excitability, this suggests a degenerate mechanism for respiratory rhythm generation- rather than a redundant mechanism, where there should be multiple buccal units that get recruited under different excitability conditions.
(2) Line 83, "but the essential requirement for a discrete, rudimentary buccal oscillator is also lost".
This statement is not supported by the data presented in this study. How does the expansion of the buccal unit imply that the essential requirement for discreteness is lost? Under increased excitability, does the burst/rhythm initiation zone also expand? Or does it still remain centered around the location of buccal units under physiological conditions? Increased excitability can lead to recruitment of a larger area, without a change in the location of the rhythmogenic kernel.
(3) Line 86, "... oscillators should be viewed as promiscuous flexible functional entities that expand or contract...".
Oscillators can be regarded as promiscuous only if, under physiological conditions, they switch positions. Under high excitability, only the flexibility argument holds, which has been established in mammals before (e.g., CA Del Negro, K Kam, JA Hayes, JL Feldman, The Journal of physiology 587 (6), 1217-1231; CA Del Negro, C Morgado-Valle, JL Feldman,Neuron 34 (5), 821-830; NA Baertsch, LJ Severs, TM Anderson, JM Ramirez, Proceedings of the National Academy of Sciences 116 (15), 7493-7502; NA Baertsch, HC Baertsch, JM Ramirez Nature communications 9 (1), 843).
Results:
(4) Interpretation of data in Figure 6.
How does the Buccal activity and L2 Power stroke change with 60nm AMPA (in CN5)? Does the increase in the Buccal neurons and decrease in power stroke neurons also reflect in the CN5 activity? Also see comments on Figure 9 data below.
(5) Interpretation of data in Figure 7.
Here, classifying buccal neurons solely by spiking may obscure the fact that the 'silent' neurons under baseline conditions were part of the rhythmic network but could not spike due to subthreshold inputs. 60 nM AMPA increased their firing in response to previously subthreshold synchronous inputs during the buccal burst. Intracellular recordings are required to negate this possibility and establish that the neuronal classification is robust.
(6) Interpretation of data in Figure 8.
"Lung units can transform into buccal units under excitation".
CN5 buccal and lung bursts need to be compared before and after AMPA injection. From Figure 8 A-D, it is apparent that the example Unit2's activity increases during the buccal bursts, after AMPA injection. However, they are also present in buccal burst pre-AMPA, albeit with less frequency.It is striking that the pre-AMPA epoch (panel A) is less than half of the post-AMPA epoch. This would, in itself, lead to a biased estimate of lung units that are active under the baseline condition during the buccal bursts.
Figure 8G, meta-analysis of lung units spiking during the baseline buccal bursts is warranted to interpret the main claim of this figure. Similarly, analysis of spiking per lung burst for the post-AMPA condition is essential for comparing the lung unit's contribution under high excitability.
(7) Interpretation of data in Figure 9
"Buccal area loses importance under increased excitation."
This interpretation is not fully supported by the data presented in this manuscript. Under 60 nm AMPA, does the ratio of lung burst to buccal burst change in CN5? This analysis is crucial for determining whether the lung units are indeed converted into buccal bursts at the expense of lung activity or whether their appearance during buccal bursts is incidental due to increased excitability. In the baseline, there are 4-5 buccal bursts per lung burst, whereas under high excitability, there are 2-3 buccal bursts per lung burst (Figure 9 A-B). This seems inconsistent with the conclusion that increased excitability converts lung units into buccal units (Figures 6 &7).
Could the authors comment on the connectivity between the lung and the buccal units? Results in Figure 9A-B indicate that lung units may receive an efference copy of buccal units, and under high excitability, their spikes may generate negative feedback onto the buccal units, terminating their bursts. This could explain the decrease in the buccal-to-lung burst in high-AMPA conditions. This type of circuit interaction resembles the mammalian breathing CPG, in which the parafacial/RTN (which controls the abdominal muscles) and preBötC (which controls the diaphragm) interact and cross-inhibit each other.
(8) Line 382.
"Buccal-like bursting produced from two independent slices".
The two "independent" slices have portions of the same anatomical kernel, the buccal rhythm generator. This experiment is like the sandwich slice preparation of preBötC (Del Negro Lab), in which two thinner slices exhibit rhythmic activity. Thus, the two slices are not independent; they are anatomically adjacent and functionally overlapping.
-

Reviewer #3 (Public review):
Summary:
This study uses isolated frog brainstem preparations to test whether inspiratory rhythm generation is confined to a narrowly defined neural center or instead reflects the activity of a distributed and adaptable network. Building on prior rodent work, the authors examine structural and functional parallels between the frog Buccal Area and the mammalian preBötzinger complex. By increasing excitatory drive, they assess whether a localized rhythmogenic region can expand into a broader network that participates in buccal rhythm generation, providing insight into how respiratory circuits are dynamically reconfigured across physiological states.
Strengths:
The work presents compelling evidence that ventilatory rhythm generation is supported by a flexible, state-dependent network rather than a fixed …
Reviewer #3 (Public review):
Summary:
This study uses isolated frog brainstem preparations to test whether inspiratory rhythm generation is confined to a narrowly defined neural center or instead reflects the activity of a distributed and adaptable network. Building on prior rodent work, the authors examine structural and functional parallels between the frog Buccal Area and the mammalian preBötzinger complex. By increasing excitatory drive, they assess whether a localized rhythmogenic region can expand into a broader network that participates in buccal rhythm generation, providing insight into how respiratory circuits are dynamically reconfigured across physiological states.
Strengths:
The work presents compelling evidence that ventilatory rhythm generation is supported by a flexible, state-dependent network rather than a fixed anatomical locus. The experimental preparation is well-suited to address these questions, and the data are generally of high quality. The demonstration that increased excitation recruits a more distributed network parallels observations in mammalian systems and strengthens the translational relevance of the findings. Overall, the analyses are thoughtful, and the interpretations are largely well supported by the results.
Weaknesses:
Some issues limit the strength of the conclusions. First, the study does not address the transition from eupnea to gasping in mammals, which could provide important physiological context for the observed AMPA-induced network reorganization. Second, the reported transformation of lung-active neurons into buccal-active neurons would benefit from additional analyses to clarify whether neurons switch identities or acquire dual activity. Finally, the necessity and sufficiency experiments in Figure 9 require further support, particularly through AMPA dose-response analyses and more comprehensive GABA manipulations, to confirm that network expansion does not obscure the continued functional importance of the core buccal region.
-

Author response:
Reviewer #1 (Public review):
Hierarchical Inference (Unit Survey)
We agree that pooling units across preparations can overstate the strength of inference if preparation-level clustering is ignored. We will therefore reanalyze the unit-survey dataset using a hierarchical approach in which the preparation/animal is treated as the unit of inference. Our pooled dataset was derived from three chunk preparations exposed to AMPA and three baseline preparations, allowing us to report per-preparation proportions and variability as requested.
A preliminary reanalysis of the buccal segment preparations is summarized below. In this analysis, the unit of inference is shifted from individual recorded units to the preparation level (n = 3 baseline; n = 3 at 60 nM AMPA), thereby accounting for potential within-preparation dependence.
Author response:
Reviewer #1 (Public review):
Hierarchical Inference (Unit Survey)
We agree that pooling units across preparations can overstate the strength of inference if preparation-level clustering is ignored. We will therefore reanalyze the unit-survey dataset using a hierarchical approach in which the preparation/animal is treated as the unit of inference. Our pooled dataset was derived from three chunk preparations exposed to AMPA and three baseline preparations, allowing us to report per-preparation proportions and variability as requested.
A preliminary reanalysis of the buccal segment preparations is summarized below. In this analysis, the unit of inference is shifted from individual recorded units to the preparation level (n = 3 baseline; n = 3 at 60 nM AMPA), thereby accounting for potential within-preparation dependence.
Author response table 1.
The distribution of units for each of the three preparations per condition is as follows:
Using the proportion of buccal units per preparation as the dependent variable:
Baseline (n = 3): mean proportion of buccal units = 6.5% (SD 5.7%).
60 nM AMPA (n = 3): mean proportion of buccal units = 53.2% (SD 6.0%).
Absolute difference in proportions = 46.7% (95% CI 33.4% to 59.8%).
Independent-samples t-test on per-preparation proportions: t(4) = 9.77, p = 0.0006.
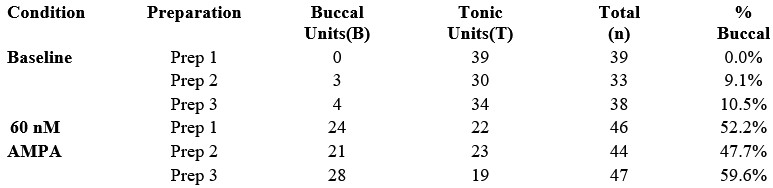
Thus, this preliminary hierarchical reanalysis indicates that the observed recruitment is consistent across preparations and is not driven by outlier data from a single animal. These results support substantial expansion of the buccal oscillator with excitation.
Statistical Standardization: In the revision, we will better justify our use of parametric and non-parametric versions of the one-sample tests and review usage in the Methods, Table 1, and figure legends for consistency.
Exclusion criteria for microinjection experiments: We will extend the description of these experiments by including a flow diagram summarizing the 15 attempted microinjection experiments and documenting the technical reasons for the 9 exclusions. These exclusions reflected the technical requirements of the preparation: (a) the buccal area had to be localized before AMPA excitation so that the effects of buccal-area manipulation during excitation could be interpreted reliably, which was not always possible; and (b) preparations had to exhibit sufficiently sustained periods of consecutive buccal bursting to permit quantification of buccal burst frequency, whereas some preparations expressed motor patterns dominated by lung bursts.
Pharmacological Potency and Necessity: We will revise the wording of this section to make the causal interpretation more precise. Our data already show that local GABA microinjections can reverse the excitatory effects of local AMPA microinjections, providing an internal control for local pharmacological efficacy of GABA when the local network is excited. Notably, the local AMPA concentration used in these experiments (5 µM) is nearly two orders of magnitude greater than the 60 nM concentration used in bath application. We therefore interpret the failure of focal GABA inhibition to abolish rhythm during global excitation as being consistent with expansion of rhythmogenic capacity beyond the spatial reach of the local injection, rather than with failure of the GABA manipulation itself.
Finding an inhibitory site that remains sensitive in bath applied AMPA is an interesting experiment but this would require identifying the anatomical substrate of a brainstem circuit for a non-ventilatory circuit in Rana that is guaranteed not to undergo reconfiguration with AMPA. This is beyond the scope of the current manuscript; based on our work to identify the neuronal substrate for ventilation in Rana, this would take at least five years to complete. In addition, having identified such a circuit there would be no guarantee that AMPA would not cause reconfiguration in this case too. With regards to transection boundaries and location of injections, we agree these would be useful refinements. We used the location of nerves as reliable landmarks to guide transections and located the buccal area using stereotactic coordinates to guide micropipette insertion and functional criteria (AMPA and GABA sufficiency and necessity tests) to locate the exact position based on our previous work.
Unit Classification: We will review the nomenclature we use to define units to ensure it does not cause confusion and provide more explicit criteria for unit classes. This will include clarification of the absence of “buccal-only” units as currently defined. Specifically, when both buccal and lung rhythms are present, units active during buccal bursts are also active during lung bursts in our preparation. This does not conflict with the multiple interacting oscillator model we have proposed previously. Rather, recruitment of buccal-area neurons during lung bursts is consistent with a model in which the lung oscillator excites the buccal oscillator. It is also consistent with prior evidence that lung bursts persist after buccal-area ablation. In addition, burst frequency during lung episodes exceeds buccal burst frequency during intervening buccal periods. We will revise the text to make this logic clearer.
Reviewer #2 (Public review):
(1) Degeneracy vs. Redundancy
We agree that degeneracy is the more precise term for the phenomenon our data demonstrate, in which structurally and functionally distinct neurons (lung units) acquire the capacity to participate in buccal rhythm generation under excitation. The Discussion already uses this language (e.g., "necessity and sufficiency may not work in a large degenerate network where rhythm generation is distributed across many elements"), but we used the word "redundant" in the Key Points Summary and Abstract in the broader sense of distributed robustness that a wider readership could grasp. Nonetheless, we recognize the distinction drawn by Goaillard and Marder (2021) and, considering the reviewers concerns, we will revise the Abstract and Key Points to adopt the degeneracy framework consistently.
(2) Loss of Essential Requirement for a Discrete Oscillator
The reviewer asks whether expansion of the rhythmically active region necessarily implies loss of the rhythmogenic kernel. We believe our necessity and sufficiency experiments (Figure 9) directly address this. Under baseline conditions, GABA microinjection into the buccal area reliably abolishes buccal bursting; under 60 nM bath AMPA, the same injection at the same location and volume has no significant effect on buccal frequency. If the kernel remained essential and the surrounding recruitment were merely supplementary, local inhibition of the kernel should still slow or abolish the rhythm. It does not. We interpret this as evidence that the essential requirement for the discrete buccal area is lost under excitation, not merely that a larger area has been recruited around a still-critical core. We acknowledge, however, that the word "lost" could be read as implying permanent elimination rather than state-dependent suspension, and we will temper this language in the revision.
(3) Novelty Relative to Mammalian Studies
We appreciate the reviewer drawing attention to the cited mammalian literature (Del Negro et al., 2002, 2009; Baertsch et al., 2018, 2019), which we discuss in detail in the manuscript. However, we respectfully note that our findings extend this literature in several ways that the public review does not acknowledge. First, Baertsch et al. demonstrated recruitment of tonic or silent neurons to become phasically active during inspiration; we show that neurons already assigned to one oscillator phase (lung) can be dynamically reassigned to another (buccal), which represents a qualitatively different form of reconfiguration. Second, we developed a novel approach to functionally ablate motor neuron pools using high-frequency nerve stimulation, enabling the unit survey to be interpreted at the premotor level which was not achieved in the mammalian studies cited. Third, our data provide the first demonstration of state-dependent oscillator expansion in a non-mammalian tetrapod, offering evolutionary context that strengthens the generality of the principle. We will revise the term "promiscuous" if it overstates the claim, but we maintain that our data support the conclusion that oscillator boundaries are flexible, which goes beyond what has been shown in mammals.
(4) Figure 6, CN5 Output Under AMPA
The reviewer asks whether the shift in premotor unit composition is reflected in CN5 motor output. This is a reasonable question. As noted in the manuscript, 60 nM AMPA produces only minor changes in the overt motor pattern as recorded from CN5, which is precisely why we interpret the premotor changes as a reorganization of the network's internal architecture that is not readily apparent from motor output alone. This is in sharp contrast to observations of substantive network reconfiguration in mammals in which eupnea is replaced by the pathological condition of gasping. We will add quantification of CN5 burst parameters (amplitude, duration, frequency) under baseline and 60 nM AMPA to make this point explicit.
(5) Subthreshold Recruitment vs. Network Expansion
The reviewer suggests that neurons classified as newly rhythmic under AMPA may have been part of the rhythmic network all along, receiving subthreshold inputs at baseline. We are grateful to the reviewer for highlighting this and hope they would agree that the literature clearly demonstrates that all respiratory neurons receive subthreshold phasic inputs of one kind or another, perhaps providing a clue that reconfiguration is a common feature of respiratory networks generally. Regardless of the implications for other animals, we agree this is likely the mechanism at work in the frog, and indeed our manuscript states that "this increase in the number and proportion of premotor buccal units is due in part to recruitment of sub-threshold buccal neurons that, under low excitability, only fire during lung bursts," citing intracellular evidence from Kogo and Remmers (1994) that lung neurons in this region receive subthreshold buccal-timed input. We note that this observation does not diminish our conclusion and likely explains the mechanism by which network expansion occurs. Whether one calls these neurons "newly recruited" or "pushed above threshold," the functional consequence is the same: a larger population of neurons is now rhythmically active during buccal bursts, and the necessity of the original buccal area is lost. We will clarify this reasoning in the revision and acknowledge the limitation that additional intracellular recordings from our preparation would be needed to fully characterize the subthreshold dynamics.
(6) Figure 8, Epoch Length and Meta-analysis
The reviewer notes that the pre-AMPA epoch appears shorter than the post-AMPA epoch in Figure 8A, which could bias unit classification. We will address this in the revision by reporting epoch durations explicitly and addressing its implication on spike counts where appropriate. Regarding the request for meta-analysis of lung unit spiking during baseline buccal bursts: this analysis is part of the rationale for the phase-recruitment panels, and we will expand Figure 8 to include the requested cross-condition comparisons (lung unit activity during baseline buccal bursts, and during post-AMPA lung bursts) as also suggested by Reviewer 3.
(7) Figure 9, Buccal-to-Lung Burst Ratio
The reviewer observes that the ratio of buccal to lung bursts decreases from approximately 4-5:1 under baseline to 2-3:1 under 60 nM AMPA, and suggests this is inconsistent with conversion of lung units into buccal units. We do not believe this is inconsistent. The buccal-to-lung burst ratio reflects the overt motor pattern, which is determined by the interaction of multiple oscillators and is influenced by AMPA at both buccal and lung levels. A change in this ratio does not speak to whether individual premotor units have acquired buccal-timed activity; the unit survey and the single-unit transformation data (Figure 8) address that question directly. Regarding the alternative model involving efference copy and cross-inhibition: this is an interesting hypothesis, but it is speculative and not tested by the current dataset. We are happy to discuss lung-buccal interactions more fully in the revision, including the parallels to parafacial/preBötC interactions in mammals, but we note that our data on unit transformation are better explained by network reconfiguration than by a feedback model that remains to be tested.
(8) "Independent" Slices
The reviewer compares our Level 2 transection to the preBötC sandwich slice preparation and argues the two resulting slices are not independent. We take the reviewer's point that "independent" may be read as implying no shared developmental or functional origin, which is not our intent. By "independent" we mean that the two physically separated slices can each generate rhythmic output without being synaptically connected to each other. This is, in fact, our central point: rhythmogenic capacity is distributed across a region broad enough to endow two separated slices with independent rhythm-generating capability when excited. We note that the analogy to the sandwich slice is imperfect because in our Level 1 cuts, only the rostral slice containing the buccal area generates rhythm -- the caudal slice does not -- whereas Level 2 cuts that bisect the buccal area produce rhythmicity in both halves, consistent with distributed capacity specifically within the buccal region. We will revise the wording to clarify what we mean by "independent" in this context.
Reviewer #3 (Public review):
Physiological Parallels: We will expand the Discussion to place these findings in a broader comparative context, including the eupnea-to-gasping transition in mammals as an example of state-dependent reconfiguration of respiratory networks. This will also allow us to clarify two advances that may otherwise be missed when comparing our work to that in mammals: (a) we developed a novel approach to functionally eliminate motor neurons, allowing mapped units to be interpreted as premotor; and (b) the state-dependent reconfiguration of the buccal oscillator occurred without qualitative changes in the overt lung-buccal motor pattern.
Unit Transformation Analysis: We will revise Figure 8 to improve clarity around the observed lung-to-buccal transformation by expanding the phase-recruitment panels as suggested and will revisit the operational definitions of lung and buccal unit identity to reduce ambiguity. The central observation is that some units active only during lung bursts under one condition become active during buccal bursts when network excitation is increased.
Saturation vs. Network Expansion: We will directly address the possibility that 60 nM bath-applied AMPA simply pushes the network toward a frequency ceiling. Two observations strongly argue against this interpretation: (a) 60 nM global AMPA produced only mild changes in buccal frequency, whereas local AMPA injection at much higher concentrations produced larger effects; and (b) local GABA was sufficient to reverse the effects of high-concentration local AMPA microinjections but insufficient to abolish rhythm during low-concentration global AMPA application. Together, these findings are more consistent with global AMPA endowing the network with distributed rhythm-generating capacity than with simple saturation of a discrete local oscillator. Notwithstanding these arguments, we will attempt to extend AMPA/GABA dose response experiment as suggested or add the lack of such experiments as a caveat to our interpretation.
Figure 9C Correction: We will correct the statistical markings in Figure 9C to align with the text in the Results regarding the significance of frequency changes under 60 nM AMPA.
In total, we believe these revisions will improve the rigor and clarity of the manuscript while preserving the central conclusion supported by the data: that the organization of the frog respiratory rhythmogenic network is state dependent and becomes more distributed under excitation.
-