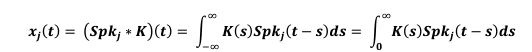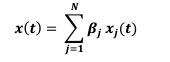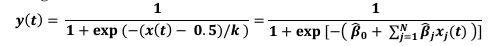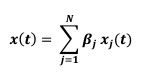Complementary cognitive roles for D2-MSNs and D1-MSNs during interval timing
Curation statements for this article:-
Curated by eLife
eLife Assessment
This valuable study examines the activity and function of dorsomedial striatal neurons in the estimation of time. The authors examine striatal activity as a function of time as well as the impact of optogenetic striatal manipulation on the animal's ability to estimate a time interval, providing solid evidence for their claims. The study could be further strengthened with a more rigorous characterization of activity and a stronger connection between their proposed model and the experimental data. The work will be of interest to neuroscientists examining how striatum contributes to behavior.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
The role of striatal pathways in cognitive processing is unclear. We studied dorsomedial striatal cognitive processing during interval timing, an elementary cognitive task that requires mice to estimate intervals of several seconds and involves working memory for temporal rules as well as attention to the passage of time. We harnessed optogenetic tagging to record from striatal D2-dopamine receptor-expressing medium spiny neurons (D2-MSNs) in the indirect pathway and from D1-dopamine receptor-expressing MSNs (D1-MSNs) in the direct pathway. We found that D2-MSNs and D1-MSNs exhibited distinct dynamics over temporal intervals as quantified by principal component analyses and trial-by-trial generalized linear models. MSN recordings helped construct and constrain a four-parameter drift-diffusion computational model in which MSN ensemble activity represented the accumulation of temporal evidence. This model predicted that disrupting either D2-MSNs or D1-MSNs would increase interval timing response times and alter MSN firing. In line with this prediction, we found that optogenetic inhibition or pharmacological disruption of either D2-MSNs or D1-MSNs increased interval timing response times. Pharmacologically disrupting D2-MSNs or D1-MSNs also changed MSN dynamics and degraded trial-by-trial temporal decoding. Together, our findings demonstrate that D2-MSNs and D1-MSNs had opposing dynamics yet played complementary cognitive roles, implying that striatal direct and indirect pathways work together to shape temporal control of action. These data provide novel insight into basal ganglia cognitive operations beyond movement and have implications for human striatal diseases and therapies targeting striatal pathways.
Article activity feed
-

-

-

-

eLife Assessment
This valuable study examines the activity and function of dorsomedial striatal neurons in the estimation of time. The authors examine striatal activity as a function of time as well as the impact of optogenetic striatal manipulation on the animal's ability to estimate a time interval, providing solid evidence for their claims. The study could be further strengthened with a more rigorous characterization of activity and a stronger connection between their proposed model and the experimental data. The work will be of interest to neuroscientists examining how striatum contributes to behavior.
-

Reviewer #1 (Public review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nosepoke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Thus, this task requires animals to estimate if at least 6 seconds have passed after the first nosepoke. After verifying that animals estimate the passage of 6 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2-MSNs increase activity, throughout this interval. They suggest that this activity follows a drift-diffusion model, in which activity increases (or decreases) to a threshold after which a decision is made. The authors next report …
Reviewer #1 (Public review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nosepoke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Thus, this task requires animals to estimate if at least 6 seconds have passed after the first nosepoke. After verifying that animals estimate the passage of 6 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2-MSNs increase activity, throughout this interval. They suggest that this activity follows a drift-diffusion model, in which activity increases (or decreases) to a threshold after which a decision is made. The authors next report that optogenetically inhibiting D1 or D2 MSNs, or pharmacologically blocking D1 and D2 receptors, increased the average wait time. This suggests that both D1 and D2 neurons contribute to the estimate of time, with a decrease in their activity corresponding to a decrease in the rate of 'drift' in their drift-diffusion model. Lastly, the authors examine MSN activity while pharmacologically inhibiting D1 or D2 receptors. The authors observe most recorded MSNs neurons decrease their activity over the interval, with the rate decreasing with D1/D2 receptor inhibition.
Major strengths:
The study employs a wide range of techniques - including animal behavioral training, electrophysiology, optogenetic manipulation, pharmacological manipulations, and computational modeling. The question posed by the authors - how striatal activity contributes to interval timing - is of importance to the field and has been the focus of many studies and labs. This paper contributes to that line of work by investigating whether D1 and D2 neurons have similar activity patterns during the timed interval, as might be expected based on prior work based on striatal manipulations. However, the authors find that D1 and D2 neurons have distinct activity patterns. They then provide a decision-making model that is consistent with all results. The data within the paper is presented very clearly, and the authors have done a nice job presenting the data in a transparent manner (e.g., showing individual cells and animals). Overall, the manuscript is relatively easy to read and clear, with sufficient detail given in most places regarding the experimental paradigm or analyses used.
Major weaknesses:
The results are based on a relatively small dataset (tens of cells).
Impact:
The task and data presented by the authors are very intriguing, and there are many groups interested in how striatal activity contributes to the neural perception of time. The authors perform a wide variety of experiments and analysis to examine how DMS activity influences time perception during an interval-timing task, allowing for insight into this process.
-

Reviewer #2 (Public review):
This study found that D1-MSNs and D2-MSNs have opposing dynamics during interval timing in a mouse-optimized interval timing task. Further optogenetic and pharmacologic inhibition of either D1 or D2 MSNs increased response time. This study provides useful experimental evidence in the coding of time in striatum. However, there are some major weaknesses in this study.
(1) Regarding the data in Figure S3, The variance within each mouse was too big, the authors need to figure out and explain what caused the large variance within the same mouse, or the authors need to increase the sample size.
(2) Regarding the results in Figure 3 C and D, Figure 6 H and Figure 7 D, what is the sample size? From the single data points in the figures, it seems that the authors were using the number of cells to do statistical tests …Reviewer #2 (Public review):
This study found that D1-MSNs and D2-MSNs have opposing dynamics during interval timing in a mouse-optimized interval timing task. Further optogenetic and pharmacologic inhibition of either D1 or D2 MSNs increased response time. This study provides useful experimental evidence in the coding of time in striatum. However, there are some major weaknesses in this study.
(1) Regarding the data in Figure S3, The variance within each mouse was too big, the authors need to figure out and explain what caused the large variance within the same mouse, or the authors need to increase the sample size.
(2) Regarding the results in Figure 3 C and D, Figure 6 H and Figure 7 D, what is the sample size? From the single data points in the figures, it seems that the authors were using the number of cells to do statistical tests and plot the figures. For example, Figure 3 C, if the authors use n= 32 D2 MSNs and n= 41D1 MSNs to do the statistical test, it could make small difference to be statistically significant. The authors should use the number of mice to do the statistical tests.
(3) Regarding the results in Figure 5, what is the reason for the increase in the response times? The authors should plot the position track during intervals (0-6 s) with or without optogenetic or pharmacologic inhibition. The authors can check Figure 3, 5, and 6 in paper https://doi.org/10.1016/j.cell.2016.06.032 for reference to analyze the data. -

Reviewer #3 (Public review):
Summary:
The cognitive striatum, also known as the dorsomedial striatum, receives input from brain regions involved in high-level cognition and plays a crucial role in processing cognitive information. However, despite its importance, the extent to which different projection pathways of the striatum contribute to this information processing remains unclear. In this paper, Bruce et al. conducted a study using various causal and correlational techniques to investigate how these pathways collectively contribute to interval timing in mice. Their results were consistent with previous research, showing that the direct and indirect striatal pathways perform opposing roles in processing elapsed time. Based on their findings, the authors proposed a revised computational model in which two separate accumulators track …
Reviewer #3 (Public review):
Summary:
The cognitive striatum, also known as the dorsomedial striatum, receives input from brain regions involved in high-level cognition and plays a crucial role in processing cognitive information. However, despite its importance, the extent to which different projection pathways of the striatum contribute to this information processing remains unclear. In this paper, Bruce et al. conducted a study using various causal and correlational techniques to investigate how these pathways collectively contribute to interval timing in mice. Their results were consistent with previous research, showing that the direct and indirect striatal pathways perform opposing roles in processing elapsed time. Based on their findings, the authors proposed a revised computational model in which two separate accumulators track evidence for elapsed time in opposing directions. These results have significant implications for understanding the neural mechanisms underlying cognitive impairment in neurological and psychiatric disorders, as disruptions in the balance between direct and indirect pathway activity are commonly observed in such conditions.
Strengths:
The authors employed a well-established approach to study interval timing and employed optogenetic tagging to observe the behavior of specific cell types in the striatum. Additionally, the authors utilized two complementary techniques to assess the impact of manipulating the activity of these pathways on behavior. Finally, the authors utilized their experimental findings to enhance the theoretical comprehension of interval timing using a computational model.
Weaknesses:
The behavioral task used in this study is best suited for investigating elapsed time perception rather than interval timing. Timing bisection tasks are often employed to study interval timing in humans and animals. Given the systemic delivery of pharmacological interventions, it is difficult to conclude that the effects are specific to the dorsomedial striatum. Future studies should use the local infusion of drugs into the dorsomedial striatum.
-

Author response:
The following is the authors’ response to the previous reviews.
Public Reviews:
Reviewer #1 (Public Review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nose poke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Thus, this task requires animals to estimate if at least 6 seconds have passed after the first nose poke. After verifying that animals estimate the passage of 6 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2MSNs increase activity, throughout this interval. They suggest that this activity follows a driftdiffusion model, in which activity …
Author response:
The following is the authors’ response to the previous reviews.
Public Reviews:
Reviewer #1 (Public Review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nose poke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Thus, this task requires animals to estimate if at least 6 seconds have passed after the first nose poke. After verifying that animals estimate the passage of 6 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2MSNs increase activity, throughout this interval. They suggest that this activity follows a driftdiffusion model, in which activity increases (or decreases) to a threshold after which a decision is made. The authors next report that optogenetically inhibiting D1 or D2 MSNs, or pharmacologically blocking D1 and D2 receptors, increased the average wait time. This suggests that both D1 and D2 neurons contribute to the estimate of time, with a decrease in their activity corresponding to a decrease in the rate of 'drift' in their drift-diffusion model. Lastly, the authors examine MSN activity while pharmacologically inhibiting D1 or D2 receptors. The authors observe most recorded MSNs neurons decrease their activity over the interval, with the rate decreasing with D1/D2 receptor inhibition.
We appreciate the careful read by this reviewer.
Major strengths:
The study employs a wide range of techniques - including animal behavioral training, electrophysiology, optogenetic manipulation, pharmacological manipulations, and computational modeling. The question posed by the authors - how striatal activity contributes to interval timing - is of importance to the field and has been the focus of many studies and labs. This paper contributes to that line of work by investigating whether D1 and D2 neurons have similar activity patterns during the timed interval, as might be expected based on prior work based on striatal manipulations. However, the authors find that D1 and D2 neurons have distinct activity patterns. They then provide a decision-making model that is consistent with all results. The data within the paper is presented very clearly, and the authors have done a nice job presenting the data in a transparent manner (e.g., showing individual cells and animals). Overall, the manuscript is relatively easy to read and clear, with sufficient detail given in most places regarding the experimental paradigm or analyses used.
We are glad that our main points come clearly through.
Major weaknesses:
One weakness to me is the impact of identifying whether D1 and D2 had similar or different activity patterns. Does observing increasing/decreasing activity in D2 versus D1, or different activity patterns in D1 and D2, support one model of interval timing over another, or does it further support a more specific idea of how DMS contributes to interval timing?
This is a great point - we were not clear. We observe distinct patterns of D2 and D1-MSN activity, but that disrupting either D2-MSNs or D1-MSNs led to increased response time. The model that this supports is that D2-MSNs and D1-MSN ensemble activity represents temporal evidence. This is a very specific model that can be rigorously tested in future work. We have now made this very clear in the abstract (Page 2).
“We found that D2-MSNs and D1-MSNs exhibited distinct dynamics over temporal intervals as quantified by principal component analyses and trial-by-trial generalized linear models. MSN recordings helped construct and constrain a fourparameter drift-diffusion computational model in which MSN ensemble activity represented the accumulation of temporal evidence. This model predicted that disrupting either D2-MSNs or D1-MSNs would increase interval timing response times and alter MSN firing. In line with this prediction, we found that optogenetic inhibition or pharmacological disruption of either D2-MSNs or D1-MSNs increased interval timing response times.”
And in the results on Page 18:
“Because both D2-MSNs and D1-MSNs accumulate temporal evidence, disrupting either MSN type in the model changed the slope. The results were obtained by simultaneously decreasing the drift rate D (equivalent to lengthening the neurons’ integration time constant) and lowering the level of network noise 𝝈: D = 𝟎. 𝟏𝟐𝟗, 𝝈 = 𝟎. 𝟎𝟒𝟑 for D2-MSNs in Fig 4A (in red; changes in noise had to accompany changes in drift rate to preserve switch response time variance. See Methods); and 𝑫 = 𝟎. 𝟏𝟐𝟐, 𝝈 = 𝟎. 𝟎𝟒𝟑 for D1-MSNs in Fig 4B (in blue). The model predicted that disrupting either D2-MSNs or D1-MSNs would increase switch response times (Fig 4C and Fig 4D) and would shift MSN dynamics.”
And in the discussion (Page 30):
“Striatal MSNs are critical for temporal control of action (Emmons et al., 2017; Gouvea et al., 2015; Mello et al., 2015). Three broad models have been proposed for how striatal MSN ensembles represent time: 1) the striatal beat frequency model, in which MSNs encode temporal information based on neuronal synchrony (Matell and Meck, 2004); 2) the distributed coding model, in which time is represented by the state of the network (Paton and Buonomano, 2018); and 3) the DDM, in which neuronal activity monotonically drifts toward a threshold after which responses are initiated (Emmons et al., 2017; Simen et al., 2011; Wang et al., 2018). While our data do not formally resolve these possibilities, our results show that D2-MSNs and D1MSNs exhibit opposing changes in firing rate dynamics in PC1 over the interval. Past work by our group and others has demonstrated that PC1 dynamics can scale over multiple intervals to represent time (Emmons et al., 2020, 2017; Gouvea et al., 2015; Mello et al., 2015; Wang et al., 2018). We find that low-parameter DDMs account for interval timing behavior with both intact and disrupted striatal D2- and D1-MSNs. While other models can capture interval timing behavior and account for MSN neuronal activity, our model does so parsimoniously with relatively few parameters (Matell and Meck, 2004; Paton and Buonomano, 2018; Simen et al., 2011). We and others have shown previously that ramping activity scales to multiple intervals, and DDMs can be readily adapted by changing the drift rate (Emmons et al., 2017; Gouvea et al., 2015; Mello et al., 2015; Simen et al., 2011). Interestingly, decoding performance was high early in the interval; indeed, animals may have been focused on this initial interval (Balci and Gallistel, 2006) in making temporal comparisons and deciding whether to switch response nosepokes.”
Regarding the reviewer’s specific question – it is not clear why D1-MSNs and D2-MSNs have opposing patterns of activity, as integration of temporal evidence can certainly be achieved increasing or decreasing firing rates alone. These patterns have been seen in motor control. Prefrontal neurons, which control striatal ramping, also ramp up and down. We have now included a paragraph on Page 30 explicitly discussing these ideas; however, future experiments will be required to investigate the source of the divergent patterns of activity among D2-MSNs and D1-MSNs.
“D2-MSNs and D1-MSNs play complementary roles in movement. For instance, stimulating D1-MSNs facilitates movement, whereas stimulating D2-MSNs impairs movement (Kravitz et al., 2010). Both populations have been shown to have complementary patterns of activity during movements with MSNs firing at different phases of action initiation and selection (Tecuapetla et al., 2016). Further dissection of action selection programs reveals that opposing patterns of activation among D2MSNs and D1-MSNs suppress and guide actions, respectively, in the dorsolateral striatum (Cruz et al., 2022). A particular advantage of interval timing is that it captures a cognitive behavior within a single dimension — time. When projected along the temporal dimension, it was surprising that D2-MSNs and D1-MSNs had opposing patterns of activity. Ramping activity in the prefrontal cortex can increase or decrease; and prefrontal neurons project to and control striatal ramping activity (Emmons et al., 2020, 2017; Wang et al., 2018). It is possible that differences in D2MSNs and D1-MSNs reflect differences in cortical ramping, which may themselves reflect more complex integrative or accumulatory processes. Further experiments are required to investigate these differences. Past pharmacological work from our group and others has shown that disrupting D2- or D1-MSNs slows timing (De Corte et al., 2019b; Drew et al., 2007, 2003; Stutt et al., 2024) and are in agreement with pharmacological and optogenetic results in this manuscript. Computational modeling predicted that disrupting either D2-MSNs or D1-MSNs increased selfreported estimates of time, which was supported by both optogenetic and pharmacological experiments.”
I found the results presented in Figures 2 and 3 to be a little confusing or misleading. In Figure 2, the authors appear to claim that D1 neurons decrease their activity over the time interval while D2 neurons increase activity. The authors use this result to suggest that D1/D2 activity patterns are different. In Figure 3, a different analysis is done, and this time D2 neurons do not significantly increase their activity with time, conflicting with Figure 2. While in both figures, there is a significant difference between the mean slopes across the population, the secondary effect of positive/negative slope for D2/D1 neurons changes. I find this especially confusing as the authors refer back to the positive/negative slope for D2/D1 neurons result throughout the rest of the text.
We were not clear. First, we attempted to quantify these differences based on PCA and slope. We have rephrased our characterization of these differences by changing text on (Page 9) to:
“These PETHs revealed that for the 6-second interval immediately after trial start, many putative D2-MSN neurons appeared to ramp up while many putative D1-MSNs appeared to ramp down. For 32 putative D2-MSNs average PETH activity increased over the 6-second interval immediately after trial start, whereas for 41 putative D1-MSNs, average PETH activity decreased. Accordingly, D2-MSNs and D1-MSNs had differences in activity early in the interval (0-5 seconds; F = 4.5, p = 0.04 accounting for variance between mice) but not late in the interval (5-6 seconds; F = 1.9, p = 0.17 accounting for variance between mice). Examination of a longer interval of 10 seconds before to 18 seconds after trial start revealed the greatest separation in D2-MSN and D1-MSN dynamics during the 6-second interval after trial start (Fig S2). Strikingly, these data suggest that D2-MSNs and D1-MSNs might display distinct dynamics during interval timing.”
We have rephrased our discussion on PCA to quantify differences in Fig 2G-H using data-driven methods (Page 12):
“To quantify differences between D2-MSNs vs D1-MSNs in Fig 2G-H, we turned to principal component analysis (PCA), a data-driven tool to capture the diversity of neuronal activity (Kim et al., 2017a). Work by our group and others has uniformly identified PC1 as a linear component among corticostriatal neuronal ensembles during interval timing (Bruce et al., 2021; Emmons et al., 2020, 2019, 2017; Kim et al., 2017a; Narayanan et al., 2013; Narayanan and Laubach, 2009; Parker et al., 2014; Wang et al., 2018). We analyzed PCA calculated from all D2-MSN and D1MSN PETHs over the 6-second interval immediately after trial start. PCA identified time-dependent ramping activity as PC1 (Fig 3A), a key temporal signal that explained 54% of variance among tagged MSNs (Fig 3B; variance for PC1 p = 0.009 vs 46 (44-49)% for any pattern of PC1 variance derived from random data; Narayanan, 2016). Consistent with population averages from Fig 2G&H, D2-MSNs and D1-MSNs had opposite patterns of activity with negative PC1 scores for D2MSNs and positive PC1 scores for D1-MSNs (Fig 3C; PC1 for D2-MSNs: -3.4 (-4.6 – 2.5); PC1 for D1-MSNs: 2.8 (-2.8 – 4.9); F = 8.8, p = 0.004 accounting for variance between mice (Fig S3A); Cohen’s d = 0.7; power = 0.80; no reliable effect of sex (F = 0.44, p = 0.51) or switching direction (F = 1.73, p = 0.19)).”
And finally, we directly investigate the heart of the reviewer’s question by explicitly comparing PC1 scores – a data-driven analysis of neuronal patterns that explain the least variance – and show that they are less than 0 for D2-MSNs (i.e., negatively correlated with a down-ramping pattern, or ramping up), and greater than 0 for D1MSNs (i.e., positively correlated with an up-ramping pattern):
“Importantly, PC1 scores for D2-MSNs were significantly less than 0 (signrank D2MSN PC1 scores vs 0: p = 0.02), implying that because PC1 ramps down, D2-MSNs tended to ramp up. Conversely, PC1 scores for D1-MSNs were significantly greater than 0 (signrank D1-MSN PC1 scores vs 0: p = 0.05), implying that D1-MSNs tended to ramp down. Thus, analysis of PC1 in Fig 3A-C suggested that D2-MSNs (Fig 2G) and D1-MSNs (Fig 2H) had opposing ramping dynamics.”
We interpret these data on Page 16:
“Our analysis of average activity (Fig 2G-H) and PC1 (Fig 3A-C) suggested that D2MSNs and D1-MSNs might have opposing dynamics. However, past computational models of interval timing have relied on drift-diffusion dynamics that increases over the interval and accumulates evidence over time (Nguyen et al., 2020; Simen et al., 2011).”
The reviewer mentions our analysis of ‘mean slopes across the population’ -which we clarify as trial-by-trial slope analysis, which is distinct from the population averages in 2G-H and 3A-C. We have now made this clear (Page 12).
“To interrogate these dynamics at a trial-by-trial level, we calculated the linear slope of D2-MSN and D1-MSN activity over the first 6 seconds of each trial using generalized linear modeling (GLM) of effects of time in the interval vs trial-by-trial firing rate (Latimer et al., 2015). Note that this analysis focuses on each trial rather than population averages in Fig 2G-H and Fig 3A-C.”
Finally, as the reviewer suggests, we have removed the term ‘slope’ from the rest of the paper, as the increasing/decreasing comes from averages and analyses of PC1. We have removed all discussion of ‘opposing’ slope or ‘increasing/decreasing’ slope.
It is a bit unclear to me how the authors chose the parameters for the model, and how well the model explains behavior is quantified. It seems that the authors didn't perform cross-validation across trials (i.e., they chose parameters that explained behavior across all trials combined, rather than choosing parameters from a subset of trials and determining whether those parameters are robust enough to explain behavior on held-out trials). I think this would increase the robustness of the result.
In addition, it remains a bit unclear to me how the authors changed the specific parameters they did to model the optogenetic manipulation. It seems these parameters were chosen because they fit the manipulation data. This makes me wonder if this model is flexible enough that there is almost always a set of parameters that would explain any experimental result; in other words, I'm not sure this model has high explanatory power.
We are glad the reviewer raised these points. First, we have now included a complete exploration of the parameter space, exactly as the reviewer recommends. These are described in the methods (Page 41):
“Selection of DDMs parameters. Our goal was to build DDMs with dynamics that produce “response times” according to the observed distribution of mice switch times. The selection of parameter values in Fig 4 was done in three steps. First, we fit the distribution of the mice behavioral data with a Gamma distribution and found its fitting values for shape 𝜶𝑴 and rate 𝜷𝑴 (Table S2 and Fig S8; R2 Data vs Gamma ≥ 𝟎. 𝟗𝟒). We recognized that the mean 𝝁𝑴 and the coefficient of variation 𝑪𝑽𝑴 are directly related to the shape and rate of the Gamma distribution by formulas 𝝁𝑴 = 𝜶𝑴/𝜷𝑴 and 𝑪𝑽𝑴 = 𝟏/√𝜶𝑴. Next, we fixed parameters 𝑭 and 𝒃 in DDM (e.g., for D2-MSNs: 𝑭 = 𝟏, 𝒃 = 𝟎. 𝟓𝟐) and simulated the DDM for a range of values for 𝑫 and 𝝈. For each pair (𝑫, 𝝈), one computational “experiment” generated 500 response times with mean 𝝁 and coefficient of variation 𝑪𝑽. We repeated the “experiment” 10 times and took the group median of 𝝁 and 𝑪𝑽 to obtain the simulation-based statistical measures 𝝁𝑺 and 𝑪𝑽𝑺. Last, we plotted 𝑬𝝁 = |(𝝁𝑺 − 𝝁𝑴)/𝝁𝑴| and 𝑬𝒄𝒗 = |𝑪𝑽𝑺 − 𝑪𝑽𝑴|, the respective relative error and the absolute error to data (Fig S7). We considered that parameter values (𝑫, 𝝈) provided a good DDM fit of mice behavioral data whenever 𝑬𝝁 ≤ 𝟎. 𝟎𝟓 and 𝑬𝒄𝒗
And included a new Fig S7 which shows the parameter space:
These new data clearly comment on the parameter space of our model.
Finally, the reviewer mentions cross-validation. We did this at length on our model and data fits. We used 10-fold cross-validation as fitlm needs enough data for the individual fits. We found that the fit was extremely stable – i.e, we ended up with standard deviations in R2<0.004 for all comparisons. Thus, we added the following point to the methods on Page 41:
“10-fold cross-validation revealed highly stable fits between gamma, models and data.”
Lastly, the results are based on a relatively small dataset (tens of cells).
This is an important point. Although it is a small optogenetically-tagged dataset, we have adequate statistical power and large effect sizes, which we now detail in the text on Page 12:
“Consistent with population averages from Fig 2G&H, D2-MSNs and D1-MSNs had opposite patterns of activity with negative PC1 scores for D2-MSNs and positive PC1 scores for D1-MSNs (Fig 3C; PC1 for D2-MSNs: -3.4 (-4.6 – 2.5); PC1 for D1MSNs: 2.8 (-2.8 – 4.9); F = 8.8, p = 0.004 accounting for variance between mice (Fig S3A); Cohen’s d = 0.7; power = 0.80; no reliable effect of sex (F = 0.44, p = 0.51) or switching direction (F = 1.73, p = 0.19)).”
And:
“GLM analysis also demonstrated that D2-MSNs had significantly different slopes (0.01 spikes/second (-0.10 – 0.10)), which were distinct from D1-MSNs (-0.20 (-0.47– 0.06; Fig 3D; F = 8.9, p = 0.004 accounting for variance between mice (Fig S3B); Cohen’s d = 0.8; power = 0.98; no reliable effect of sex (F = 0.02, p = 0.88) or switching direction (F = 1.72, p = 0.19)).”
And we have included the reviewers point as a limitation on Page 33:
“Second, although we had adequate statistical power and medium-to-large effect sizes, optogenetic tagging is low-yield, and it is possible that recording more of these neurons would afford greater opportunity to identify more robust results and alternative coding schemes, such as neuronal synchrony.”
Impact:
The task and data presented by the authors are very intriguing, and there are many groups interested in how striatal activity contributes to the neural perception of time. The authors perform a wide variety of experiments and analysis to examine how DMS activity influences time perception during an interval-timing task, allowing for insight into this process. However, the significance of the key finding -- that D1 and D2 activity is distinct across time -- remains somewhat ambiguous to me.
Again, we are glad that the reviewer appreciated our main point, and we very much appreciate the additional points about interpretation, model parameters, and statistical power. If there is any way we can clarify the text further we are happy to do so.
Reviewer #2 (Public Review):
(1) Regarding the results in Figure 2 and Figure 5: for the heatmaps in Fig.2F and Fig.2E, the overall activity pattern of D1 and D2 MSNs looks very similar, both D1 and D2 MSNs contains neurons showing decreasing or increasing activity during interval timing. And the optogenetic and pharmacologic inhibition of either D1 or D2 MSNs resulted in similar behavior outcomes. To me, the D1 and D2 MSN activities were more complementary than opposing.
This is a great point. In our last revision, R3 suggested that complementary means opposing – and suggested we change the title to reflect this. Our original title was ‘Complementary cognitive roles for D2-MSNs and D1-MSNs during interval timing’ – and we have changed the title back to this. We have clarified what we meant by complementary in the abstract (Page 2):
“Together, our findings demonstrate that D2-MSNs and D1-MSNs had opposing dynamics yet played complementary cognitive roles, implying that striatal direct and indirect pathways work together to shape temporal control of action.”
And on Page 30:
“These data, when combined with our model predictions, demonstrate that despite opposing dynamics, D2-MSNs and D1-MSN contribute complementary temporal evidence to controlling actions in time.”
If the authors want to emphasize the opposing side of D1 and D2 MSNs, then the manipulation experiments need to be re-designed, since the average activity of D2 MSNs increased, while D1 MSNs decreased during interval timing, instead of using inhibitory manipulations in both pathways, the authors should use inhibitory manipulation in D2-MSNs, while using optogenetic or pharmacology to activate D1-MSNs. In this way, the authors can demonstrate the opposing role of D1 and D2 MSNs and the functions of increased activity in D2-MSNs and decreased activity in D1-MSNs.
These are great ideas, which we agree with. We would like to emphasize the complementary nature as noted in our original title, and not the opposing side of D1/D2 MSNs. The experiments proposed by reviewer are certainly worth doing, but would likely be quite complex to find the right stimulation parameters to affect timing without affecting movement – and we have now included them as an important limitation / future direction (Page 33):
“Fifth, we did not deliver stimulation to the striatum because our pilot experiments triggered movement artifacts or task-specific dyskinesias (Kravitz et al., 2010). Future stimulation approaches carefully titrated to striatal physiology may affect interval timing without affecting movement.”
(2) Regarding the results in Figure 3 C and D, Figure 6 H and Figure 7 D, what is the sample size? From the single data points in the figures, it seems that the authors were using the number of cells to do statistical tests and plot the figures. For example, Figure 3 C, if the authors use n= 32 D2 MSNs and n= 41D1 MSNs to do the statistical test, it could make a small difference to be statistically significant. The authors should use the number of mice to do the statistical tests.
These are important points that were discussed at length in the prior review. First, for the sample size, we now have detailed in our Table 1:
Second, we have detailed our statistical approach which explicitly deals with repeated observations of neurons across mice (Page 43):
“Statistics. All data and statistical approaches were reviewed by the Biostatistics, Epidemiology, and Research Design Core (BERD) at the Institute for Clinical and Translational Sciences (ICTS) at the University of Iowa. All code and data are made available at http://narayanan.lab.uiowa.edu/article/datasets. We used the median to measure central tendency and the interquartile range to measure spread. We used Wilcoxon nonparametric tests to compare behavior between experimental conditions and Cohen’s d to calculate effect size. Analyses of putative single-unit activity and basic physiological properties were carried out using custom routines for MATLAB. For all neuronal analyses, variability between animals was accounted for using generalized linear-mixed effects models and incorporating a random effect for each mouse into the model, which allows us to account for inherent betweenmouse variability. We used fitglme in MATLAB and verified main effects using lmer in R. We accounted for variability between MSNs in pharmacological datasets in which we could match MSNs between saline, D2 blockade, and D1 blockade. P values < 0.05 were interpreted as significant.”
We have formally reviewed this approach with professional biostatisticians at the University of Iowa.
Finally, we note that we do have adequate statistical power for analysis of Fig 3C and D: we have adequate statistical power and large effect sizes, which we now detail in the text on Page 12:
“Consistent with population averages from Fig 2G&H, D2-MSNs and D1-MSNs had opposite patterns of activity with negative PC1 scores for D2-MSNs and positive PC1 scores for D1-MSNs (Fig 3C; PC1 for D2-MSNs: -3.4 (-4.6 – 2.5); PC1 for D1MSNs: 2.8 (-2.8 – 4.9); F = 8.8, p = 0.004 accounting for variance between mice (Fig S3A); Cohen’s d = 0.7; power = 0.80; no reliable effect of sex (F = 0.44, p = 0.51) or switching direction (F = 1.73, p = 0.19)).”
And, on Page 12:
“GLM analysis also demonstrated that D2-MSNs had significantly different slopes (0.01 spikes/second (-0.10 – 0.10)), which were distinct from D1-MSNs (-0.20 (-0.47– 0.06; Fig 3D; F = 8.9, p = 0.004 accounting for variance between mice (Fig S3B); Cohen’s d = 0.8; power = 0.98; no reliable effect of sex (F = 0.02, p = 0.88) or switching direction (F = 1.72, p = 0.19)).”
And we have included the reviewers point as a limitation on Page 33:
“Second, although we had adequate statistical power and medium-to-large effect sizes, optogenetic tagging is low-yield, and it is possible that recording more of these neurons would afford greater opportunity to identify more robust results and alternative coding schemes, such as neuronal synchrony.”
(3) Regarding the results in Figure 5, wly at is the reason for the increase in the response times? The authors should plot the position track during intervals (0-6 s) with or without optogenetic or pharmacologic inhibition. The authors can check Figures 3, 5, and 6 in the paper https://doi.org/10.1016/j.cell.2016.06.032 for reference to analyze the data.
These are key points, and we are glad the reviewer raised them. Our interpretation is that response time increases – without reliable changes in other task-specific movements such as nosepoke reaction time or traversal time (Fig S9). This was lacking in our prior manuscript, and we are glad the reviewer raised it. We have now added this to Page 30
“Our interpretation is that because the activity of D2-MSN and D1-MSN ensembles represents the accumulation evidence, pharmacological/optogenetic disruption of D2-MSN/D1-MSN activity slows this accumulation process, leading to slower interval timing-response times (Fig 5) without changing other task-specific movements (Fig S9). These results provide new insight into how opposing patterns of striatal MSN activity control behavior in similar ways and show that they play a complementary role in elementary cognitive operations.”
Regarding the tracking of velocity, we unfortunately do not have this information reliably across all conditions. This citation is a beautiful landmark paper, and we are working on collecting this information in our new datasets going forward. We have included this as a major limitation (Page 34):
“Still, future work combining motion tracking/accelerometry with neuronal ensemble recording and optogenetics and including bisection tasks may further unravel timing vs. movement in MSN dynamics (Robbe, 2023; Tecuapetla et al., 2016).”
Once again, we are appreciative of the thoughtful points raised by this reviewer.
Reviewer #3 (Public Review):
Summary:
The cognitive striatum, also known as the dorsomedial striatum, receives input from brain regions involved in high-level cognition and plays a crucial role in processing cognitive information. However, despite its importance, the extent to which different projection pathways of the striatum contribute to this information processing remains unclear. In this paper, Bruce et al. conducted a study using various causal and correlational techniques to investigate how these pathways collectively contribute to interval timing in mice. Their results were consistent with previous research, showing that the direct and indirect striatal pathways perform opposing roles in processing elapsed time. Based on their findings, the authors proposed a revised computational model in which two separate accumulators track evidence for elapsed time in opposing directions. These results have significant implications for understanding the neural mechanisms underlying cognitive impairment in neurological and psychiatric disorders, as disruptions in the balance between direct and indirect pathway activity are commonly observed in such conditions.
Strengths:
The authors employed a well-established approach to study interval timing and employed optogenetic tagging to observe the behavior of specific cell types in the striatum. Additionally, the authors utilized two complementary techniques to assess the impact of manipulating the activity of these pathways on behavior. Finally, the authors utilized their experimental findings to enhance the theoretical comprehension of interval timing using a computational model.
We very much appreciate the considered read and comments by the reviewer, and recognition of the breadth of techniques in this manuscript.
Weaknesses:
The behavioral task used in this study is best suited for investigating elapsed time perception, rather than interval timing. Timing bisection tasks are often employed to study interval timing in humans and animals. In the optogenetic experiment, the laser was kept on for too long (18 seconds) at high power (12 mW). This has been shown to cause adverse effects on population activity (for example, through heating the tissue) that are not necessarily related to their function during the task epochs. Given the systemic delivery of pharmacological interventions, it is difficult to conclude that the effects are specific to the dorsomedial striatum. Future studies should use the local infusion of drugs into the dorsomedial striatum.
These are important points. We agree with them completely and have now included responses to them. First, bisection tasks certainly have advantages – we have justified our approach in the discussion (Page 32):
“Our task version has been used extensively to study interval timing in mice and humans (Balci et al., 2008; Bruce et al., 2021; Stutt et al., 2024; Tosun et al., 2016; Weber et al., 2023). However, temporal bisection tasks, in which animals hold during a temporal cue and respond at different locations depending on cue length, have advantages in studying how animals time an interval because animals are not moving while estimating cue duration (Paton and Buonomano, 2018; Robbe, 2023; Soares et al., 2016). Our interval timing task version – in which mice switch between two response nosepokes to indicate their interval estimate has elapsed – has been used extensively in rodent models of neurodegenerative disease (Larson et al., 2022; Weber et al., 2024, 2023; Zhang et al., 2021), as well as in humans (Stutt et al., 2024). This version of interval timing involves motor timing, which engages executive function and has more translational relevance for human diseases than perceptual timing or bisection tasks (Brown, 2006; Farajzadeh and Sanayei, 2024; Nombela et al., 2016; Singh et al., 2021). Furthermore, because many therapeutics targeting dopamine receptors are used clinically, these findings help describe how dopaminergic drugs might affect cognitive function and dysfunction. Future studies of D2-MSNs and D1-MSNs in temporal bisection and other timing tasks may further clarify the relative roles of D2- and D1-MSNs in interval timing and time estimation.”
Second – we have included an explicit control that has the same laser that is on for the same epoch as in the experimental animal – and find no effects. This is now detailed in the methods: (Page 37):
“To control for heating and nonspecific effects of optogenetics, we performed control experiments in mice without opsins using identical laser parameters in D2-cre or D1-cre mice (Fig S6).”
And in the results (Page 21):
“To control for heating and nonspecific effects of optogenetics, we performed control experiments in D2-cre mice without opsins using identical laser parameters; we found no reliable effects for opsin-negative controls (Fig S6).”
And on Page 21:
“As with D2-MSNs, we found no reliable effects with opsin-negative controls in D1MSNs (Fig S6).”
We have now detailed these results in Figure S6:
Regarding focal pharmacology, we performed this experiment with focal infusion of D1/D2 antagonists in our prior work, which we have now cited (Page 4):
“Similar behavioral effects were found with systemic (Stutt et al., 2024) or focal infusion of D2 or D1 antagonists locally within the dorsomedial striatum (De Corte et al., 2019a).”
Comments on revised version:
Thank you for the comprehensive revisions. Most of my (addressable) concerns were addressed. The current version of your manuscript appears significantly improved.
Once again, we appreciate the reviewer’s constructive and insightful comments and careful review of our manuscript. Their comments have been extremely helpful.
-

-

eLife assessment
This valuable study examines the activity and function of dorsomedial striatal neurons in estimating time. The authors used various causal and correlational techniques to investigate how these pathways collectively contribute to interval timing in mice and found that the direct and indirect striatal pathways perform opposing roles in processing elapsed time. The evidence is solid. The manuscript would interest neuroscientists examining how striatum contributes to behavior.
-

Reviewer #1 (Public Review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nose poke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Thus, this task requires animals to estimate if at least 6 seconds have passed after the first nose poke. After verifying that animals estimate the passage of 6 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2-MSNs increase activity, throughout this interval. They suggest that this activity follows a drift-diffusion model, in which activity increases (or decreases) to a threshold after which a decision is made. The authors next report …Reviewer #1 (Public Review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nose poke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Thus, this task requires animals to estimate if at least 6 seconds have passed after the first nose poke. After verifying that animals estimate the passage of 6 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2-MSNs increase activity, throughout this interval. They suggest that this activity follows a drift-diffusion model, in which activity increases (or decreases) to a threshold after which a decision is made. The authors next report that optogenetically inhibiting D1 or D2 MSNs, or pharmacologically blocking D1 and D2 receptors, increased the average wait time. This suggests that both D1 and D2 neurons contribute to the estimate of time, with a decrease in their activity corresponding to a decrease in the rate of 'drift' in their drift-diffusion model. Lastly, the authors examine MSN activity while pharmacologically inhibiting D1 or D2 receptors. The authors observe most recorded MSNs neurons decrease their activity over the interval, with the rate decreasing with D1/D2 receptor inhibition.Major strengths:
The study employs a wide range of techniques - including animal behavioral training, electrophysiology, optogenetic manipulation, pharmacological manipulations, and computational modeling. The question posed by the authors - how striatal activity contributes to interval timing - is of importance to the field and has been the focus of many studies and labs. This paper contributes to that line of work by investigating whether D1 and D2 neurons have similar activity patterns during the timed interval, as might be expected based on prior work based on striatal manipulations. However, the authors find that D1 and D2 neurons have distinct activity patterns. They then provide a decision-making model that is consistent with all results. The data within the paper is presented very clearly, and the authors have done a nice job presenting the data in a transparent manner (e.g., showing individual cells and animals). Overall, the manuscript is relatively easy to read and clear, with sufficient detail given in most places regarding the experimental paradigm or analyses used.Major weaknesses:
One weakness to me is the impact of identifying whether D1 and D2 had similar or different activity patterns. Does observing increasing/decreasing activity in D2 versus D1, or different activity patterns in D1 and D2, support one model of interval timing over another, or does it further support a more specific idea of how DMS contributes to interval timing?I found the results presented in Figures 2 and 3 to be a little confusing or misleading. In Figure 2, the authors appear to claim that D1 neurons decrease their activity over the time interval while D2 neurons increase activity. The authors use this result to suggest that D1/D2 activity patterns are different. In Figure 3, a different analysis is done, and this time D2 neurons do not significantly increase their activity with time, conflicting with Figure 2. While in both figures, there is a significant difference between the mean slopes across the population, the secondary effect of positive/negative slope for D2/D1 neurons changes. I find this especially confusing as the authors refer back to the positive/negative slope for D2/D1 neurons result throughout the rest of the text.
It is a bit unclear to me how the authors chose the parameters for the model, and how well the model explains behavior is quantified. It seems that the authors didn't perform cross-validation across trials (i.e., they chose parameters that explained behavior across all trials combined, rather than choosing parameters from a subset of trials and determining whether those parameters are robust enough to explain behavior on held-out trials). I think this would increase the robustness of the result.
In addition, it remains a bit unclear to me how the authors changed the specific parameters they did to model the optogenetic manipulation. It seems these parameters were chosen because they fit the manipulation data. This makes me wonder if this model is flexible enough that there is almost always a set of parameters that would explain any experimental result; in other words, I'm not sure this model has high explanatory power.
Lastly, the results are based on a relatively small dataset (tens of cells).
Impact:
The task and data presented by the authors are very intriguing, and there are many groups interested in how striatal activity contributes to the neural perception of time. The authors perform a wide variety of experiments and analysis to examine how DMS activity influences time perception during an interval-timing task, allowing for insight into this process. However, the significance of the key finding -- that D1 and D2 activity is distinct across time -- remains somewhat ambiguous to me. -

Reviewer #2 (Public Review):
(1) Regarding the results in Figure 2 and Figure 5: for the heatmaps in Fig.2F and Fig.2E, the overall activity pattern of D1 and D2 MSNs looks very similar, both D1 and D2 MSNs contains neurons showing decreasing or increasing activity during interval timing. And the optogenetic and pharmacologic inhibition of either D1 or D2 MSNs resulted in similar behavior outcomes. To me, the D1 and D2 MSN activities were more complementary than opposing. If the authors want to emphasize the opposing side of D1 and D2 MSNs, then the manipulation experiments need to be re-designed, since the average activity of D2 MSNs increased, while D1 MSNs decreased during interval timing, instead of using inhibitory manipulations in both pathways, the authors should use inhibitory manipulation in D2-MSNs, while using optogenetic or …
Reviewer #2 (Public Review):
(1) Regarding the results in Figure 2 and Figure 5: for the heatmaps in Fig.2F and Fig.2E, the overall activity pattern of D1 and D2 MSNs looks very similar, both D1 and D2 MSNs contains neurons showing decreasing or increasing activity during interval timing. And the optogenetic and pharmacologic inhibition of either D1 or D2 MSNs resulted in similar behavior outcomes. To me, the D1 and D2 MSN activities were more complementary than opposing. If the authors want to emphasize the opposing side of D1 and D2 MSNs, then the manipulation experiments need to be re-designed, since the average activity of D2 MSNs increased, while D1 MSNs decreased during interval timing, instead of using inhibitory manipulations in both pathways, the authors should use inhibitory manipulation in D2-MSNs, while using optogenetic or pharmacology to activate D1-MSNs. In this way, the authors can demonstrate the opposing role of D1 and D2 MSNs and the functions of increased activity in D2-MSNs and decreased activity in D1-MSNs.
(2) Regarding the results in Figure 3 C and D, Figure 6 H and Figure 7 D, what is the sample size? From the single data points in the figures, it seems that the authors were using the number of cells to do statistical tests and plot the figures. For example, Figure 3 C, if the authors use n= 32 D2 MSNs and n= 41D1 MSNs to do the statistical test, it could make a small difference to be statistically significant. The authors should use the number of mice to do the statistical tests.
(3) Regarding the results in Figure 5, what is the reason for the increase in the response times? The authors should plot the position track during intervals (0-6 s) with or without optogenetic or pharmacologic inhibition. The authors can check Figures 3, 5, and 6 in the paper https://doi.org/10.1016/j.cell.2016.06.032 for reference to analyze the data.
-

Reviewer #3 (Public Review):
Summary:
The cognitive striatum, also known as the dorsomedial striatum, receives input from brain regions involved in high-level cognition and plays a crucial role in processing cognitive information. However, despite its importance, the extent to which different projection pathways of the striatum contribute to this information processing remains unclear. In this paper, Bruce et al. conducted a study using various causal and correlational techniques to investigate how these pathways collectively contribute to interval timing in mice. Their results were consistent with previous research, showing that the direct and indirect striatal pathways perform opposing roles in processing elapsed time. Based on their findings, the authors proposed a revised computational model in which two separate accumulators track …Reviewer #3 (Public Review):
Summary:
The cognitive striatum, also known as the dorsomedial striatum, receives input from brain regions involved in high-level cognition and plays a crucial role in processing cognitive information. However, despite its importance, the extent to which different projection pathways of the striatum contribute to this information processing remains unclear. In this paper, Bruce et al. conducted a study using various causal and correlational techniques to investigate how these pathways collectively contribute to interval timing in mice. Their results were consistent with previous research, showing that the direct and indirect striatal pathways perform opposing roles in processing elapsed time. Based on their findings, the authors proposed a revised computational model in which two separate accumulators track evidence for elapsed time in opposing directions. These results have significant implications for understanding the neural mechanisms underlying cognitive impairment in neurological and psychiatric disorders, as disruptions in the balance between direct and indirect pathway activity are commonly observed in such conditions.Strengths:
The authors employed a well-established approach to study interval timing and employed optogenetic tagging to observe the behavior of specific cell types in the striatum. Additionally, the authors utilized two complementary techniques to assess the impact of manipulating the activity of these pathways on behavior. Finally, the authors utilized their experimental findings to enhance the theoretical comprehension of interval timing using a computational model.Weaknesses:
The behavioral task used in this study is best suited for investigating elapsed time perception, rather than interval timing. Timing bisection tasks are often employed to study interval timing in humans and animals. In the optogenetic experiment, the laser was kept on for too long (18 seconds) at high power (12 mW). This has been shown to cause adverse effects on population activity (for example, through heating the tissue) that are not necessarily related to their function during the task epochs. Given the systemic delivery of pharmacological interventions, it is difficult to conclude that the effects are specific to the dorsomedial striatum. Future studies should use the local infusion of drugs into the dorsomedial striatum.Comments on revised version:
Thank you for the comprehensive revisions. Most of my (addressable) concerns were addressed. The current version of your manuscript appears significantly improved.
-

Author response:
The following is the authors’ response to the original reviews.
Reviewer #1 (Public Review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nose poke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Critically, this task thus requires animals to estimate if at least 6 seconds have passed after the first nose poke - this is the key aspect of the task focused on here. After verifying that animals reliably estimate the passage of 6 seconds by leaving on average after 9 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2-MSNs increase activity, throughout …
Author response:
The following is the authors’ response to the original reviews.
Reviewer #1 (Public Review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nose poke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Critically, this task thus requires animals to estimate if at least 6 seconds have passed after the first nose poke - this is the key aspect of the task focused on here. After verifying that animals reliably estimate the passage of 6 seconds by leaving on average after 9 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2-MSNs increase activity, throughout this interval. They suggest that this activity follows a drift-diffusion model, in which activity increases (or decreases) to a threshold after which a decision (to leave) is made. The authors next report that optogenetically inhibiting D1 or D2 MSNs, or pharmacologically blocking D1 and D2 receptors, increased the average wait time of the animals to 10 seconds on average. This suggests that both D1 and D2 neurons contribute to the estimate of time, with a decrease in their activity corresponding to a decrease in the rate of
'drift' in their drift-diffusion model. Lastly, the authors examine MSN activity while pharmacologically inhibiting D1 or D2 receptors. The authors observe most recorded MSNs neurons decrease their activity over the interval, with the rate decreasing with D1/D2 receptor inhibition.
Major strengths:
The study employs a wide range of techniques - including animal behavioral training, electrophysiology, optogenetic manipulation, pharmacological manipulations, and computational modeling. The behavioral task used by the authors is quite interesting and a nice way to probe interval timing in rodents. The question posed by the authors - how striatal activity contributes to interval timing - is of importance to the field and has been the focus of many studies and labs; thus, this paper can meaningfully contribute to that conversation. The data within the paper is presented very clearly, and the authors have done a nice job presenting the data in a transparent manner (e.g., showing individual cells and animals). Overall, the manuscript is relatively easy to read and clear, with sufficient detail given in most places regarding the experimental paradigm or analyses used.
We are glad our main points came through to the reviewer.
Major weaknesses:
I perceive two major weaknesses. The first is the impact or contextualization of their results in terms of the results of the field more broadly. More specifically, it was not clear to me how the authors are interpreting the striatal activity in the context of what others have observed during interval timing tasks. In other words - what was the hypothesis going into this experiment? Does observing increasing/decreasing activity in D2 versus D1 support one model of interval timing over another, or does it further support a more specific idea of how DMS contributes to interval timing? Or was the main question that we didn't know if D2 or D1 neurons had differential activity during interval timing?
This is a helpful comment. Our hypothesis is that D1 and D2 MSNs had similar patterns of activity. Our rationale is prior behavioral work from our group describing that blocking striatal D1 and D2 dopamine receptors had similar behavioral effects on interval timing (De Corte et al., 2019; Stutt et al., 2023), We rewrote our introduction with this idea in mind (Line 89)
“We and others have found that striatal MSNs encode time across multiple intervals by time-dependent ramping activity or monotonic changes in firing rate across a temporal interval (Emmons et al., 2017; Gouvea et al., 2015; Mello et al., 2015; Wang et al., 2018). However, the respective roles of D2-MSNs and D1-MSNs are unknown. Past work has shown that disrupting either D2-dopamine receptors (D2) or D1-dopamine receptors (D1) powerfully impairs interval timing by increasing estimates of elapsed time (Drew et al., 2007; Meck, 2006). Similar behavioral effects were found with systemic (Stutt et al., 2024) or local dorsomedial striatal D2 or D1 disruption (De Corte et al., 2019a). These data lead to the hypothesis that D2 MSNs and D1 MSNs have similar patterns of ramping activity across a temporal interval.
We tested this hypothesis with a combination of optogenetics, neuronal ensemble recording, computational modeling, and behavioral pharmacology. We use a well-described mouse-optimized interval timing task (Balci et al., 2008; Bruce et al., 2021; Larson et al., 2022; Stutt et al., 2024; Tosun et al., 2016; Weber et al., 2023). Strikingly, optogenetic tagging of D2-MSNs and D1-MSNs revealed distinct neuronal dynamics, with D2-MSNs tending to increase firing over an interval and D1-MSNs tending to decrease firing over the same interval, similar to opposing movement dynamics (Cruz et al., 2022; Kravitz et al., 2010; Tecuapetla et al., 2016). MSN dynamics helped construct and constrain a four-parameter drift-diffusion computational model of interval timing, which predicted that disrupting either D2MSNs or D1-MSNs would increase interval timing response times. Accordingly, we found that optogenetic inhibition of either D2-MSNs or D1-MSNs increased interval timing response times. Furthermore, pharmacological blockade of either D2- or D1receptors also increased response times and degraded trial-by-trial temporal decoding from MSN ensembles. Thus, D2-MSNs and D1-MSNs have opposing temporal dynamics yet disrupting either MSN type produced similar effects on behavior. These data demonstrate how striatal pathways play complementary roles in elementary cognitive operations and are highly relevant for understanding the pathophysiology of human diseases and therapies targeting the striatum.”
In the second, I felt that some of the conclusions suggested by the authors don't seem entirely supported by the data they present, or the data presented suggests a slightly more complicated story. Below I provide additional detail on some of these instances.
Regarding the results presented in Figures 2 and 3:
I am not sure the PC analysis adds much to the interpretation, and potentially unnecessarily complicates things. In particular, running PCA on a matrix of noisy data that is smoothed with a Gaussian will often return PCs similar to what is observed by the authors, with the first PC being a line up/down, the 2nd PC being a parabola that is up/down, etc. Thus, I'm not sure that there is much to be interpreted by the specific shape of the PCs here.
We are glad the reviewer raised this point. First, regarding the components in noisy data, what the reviewer says is correct, but usually, the variance explained by PC1 is small. This is the reason we include scree plots in our PC analysis (Fig 3B and Fig 6G). When we compare our PC1s to variance explained in random data, our PC1 variance is always stronger. We have now included this in our manuscript:
First, we generated random data and examined how much variance PC1 might generate.
We added this to the methods (Line 634)
“The variance of PC1 was empirically compared against data generated from 1000 iterations of data from random timestamps with identical bins and kernel density estimates. Average plots were shown with Gaussian smoothing for plotting purposes only.”
These data suggested that our PC1 was stronger than that observed in random data (Line 183):
“PCA identified time-dependent ramping activity as PC1 (Fig 3A), a key temporal signal that explained 54% of variance among tagged MSNs (Fig 3B; variance for PC1 p = 0.009 vs 46 (44-49)% variance for PC1 derived from random data; Narayanan, 2016).”
And in the pharmacology data (Line 367):
“The first component (PC1), which explained 54% of neuronal variance, exhibited “time-dependent ramping”, or monotonic changes over the 6 second interval immediately after trial start (Fig 6F-G; variance for PC1 p = 0.001 vs 46 (45-47)% variance in random data; Narayanan, 2016).”
Second, we note that we have used this analysis extensively in the past, and PC1 has always been identified as a linear ramping in our work and in work by others (Line 179):
“Work by our group and others has uniformly identified PC1 as a linear component among corticostriatal neuronal ensembles during interval timing (Bruce et al., 2021; Emmons et al., 2020, 2019, 2017; Kim et al., 2017a; Narayanan et al., 2013; Narayanan and Laubach, 2009; Parker et al., 2014; Wang et al., 2018).”
Third, we find that PC1 is highly correlated to the GLM slope (Line 205):
“Trial-by-trial GLM slope was correlated with PC1 scores in Fig 3A-C (PC1 scores vs. GLM slope r = -0.60, p = 10-8).”
Fourth, our goal was not to heavily interpret PC1 – but to compare D1 vs. D2 MSNs, or compare population responses to D2/D1 pharmacology. We have now made this clear in introducing PCA analyses in the results (Line 177):
“To quantify differences in D2-MSNs vs D1-MSNs, we turned to principal component analysis (PCA), a data-driven tool to capture the diversity of neuronal activity (Kim et al., 2017a).”
Finally, despite these arguments the reviewer’s point is well taken. Accordingly, we have removed all analyses of PC2 from the manuscript which may have been overly interpretative.
We have now removed language that interpreted the components, and we now find the discussion of PC1 much more data-driven. We have also removed much of the advanced PC analysis in Figure S9. Given our extensive past work using this exact analysis of PC1, we think PCA adds a considerable amount to our manuscript justified as the reviewer suggested.
I think an alternative analysis that might be both easier and more informative is to compute the slope of the activity of each neuron across the 6 seconds. This would allow the authors to quantify how many neurons increase or decrease their activity much like what is shown in Figure 2.
We agree – we now do exactly this analysis in Figure 3D. We now clarify this in detail, using the reviewer’s language to the methods (Line 648):
“To measure time-related ramping over the first 6 seconds of the interval, we used trial-by-trial generalized linear models (GLMs) at the individual neuron level in which the response variable was firing rate and the predictor variable was time in the interval or nosepoke rate (Shimazaki and Shinomoto, 2007). For each neuron, it’s time-related “ramping” slope was derived from the GLM fit of firing rate vs time in the interval, for all trials per neuron. All GLMs were run at a trial-by-trial level to avoid effects of trial averaging (Latimer et al., 2015) as in our past work (Bruce et al., 2021; Emmons et al., 2017; Kim et al., 2017b).”
And to the results (Line 194):
“To interrogate these dynamics at a trial-by-trial level, we calculated the linear slope of D2-MSN and D1-MSN activity over the first 6 seconds of each trial using generalized linear modeling (GLM) of effects of time in the interval vs trial-by-trial firing rate (Latimer et al., 2015).”
Relatedly, it seems that the data shown in Figure 2D *doesn't* support the authors' main claim regarding D2/D1 MSNs increasing/decreasing their activity, as the trial-by-trial slope is near 0 for both cell types.
This likely refers to Figure 3D. The reviewer is correct that the changes in slope are small and near 0. Our goal was to show that D2-MSN and D1-MSN slopes were distinct – rather than increasing and decreasing. We have added this to the abstract (Line 46)
“We found that D2-MSNs and D1-MSNs exhibited distinct dynamics over temporal intervals as quantified by principal component analyses and trial-by-trial generalized linear models.”
We have clarified this idea in our hypothesis (Line 96):
“These data led to the hypothesis that D2 MSNs and D1 MSNs have similar patterns of ramping activity across a temporal interval.”
We have added this idea to the results (Line 194)
“To interrogate these dynamics at a trial-by-trial level, we calculated the linear slope of D2-MSN and D1-MSN activity over the first 6 seconds of each trial using generalized linear modeling (GLM) of effects of time in the interval vs trial-by-trial firing rate (Latimer et al., 2015). Nosepokes were included as a regressor for movement. GLM analysis also demonstrated that D2-MSNs had significantly different slopes (-0.01 spikes/second (-0.10 – 0.10)), which were distinct from D1MSNs (-0.20 (-0.47– -0.06; Fig 3D; F = 8.9, p = 0.004 accounting for variance between mice (Fig S3B); Cohen’s d = 0.8; power = 0.98; no reliable effect of sex (F = 0.02, p = 0.88) or switching direction (F = 1.72, p = 0.19)). We found that D2-MSNs and D1-MSNs had significantly different slopes even when excluding outliers (4 outliers excluded outside of 95% confidence intervals; F = 7.51, p = 0.008 accounting for variance between mice) and when the interval was defined as the time between trial start and the switch response on a trial-by-trial basis for each neuron (F = 4.3, p = 0.04 accounting for variance between mice). Trial-by-trial GLM slope was correlated with PC1 scores in Fig 3A-C (PC1 scores vs. GLM slope r = -0.60, p = 108). These data demonstrate that D2-MSNs and D1-MSNs had distinct slopes of firing rate across the interval and were consistent with analyses of average activity and PC1, which exhibited time-related ramping.”
And Line 215:
“In summary, we used optogenetic tagging to record from D2-MSNs and D1-MSNs during interval timing. Analyses of average activity, PC1, and trial-by-trial firingrate slopes over the interval provide convergent evidence that D2-MSNs and D1MSNs had distinct and opposing dynamics during interval timing. These data provide insight into temporal processing by striatal MSNs.”
And in the discussion (Line 415):
“We describe how striatal MSNs work together in complementary ways to encode an elementary cognitive process, interval timing. Strikingly, optogenetic tagging showed that D2-MSNs and D1-MSNs had distinct dynamics during interval timing. “
We have now included a new plot with box plots to make the differences in Figure 3D clear
Other reviewers requested additional qualitative descriptions of our data, and we have referred to increases / decreases in this context.
Regarding the results in Figure 4:
The authors suggest that their data is consistent with a drift-diffusion model. However, it is unclear how well the output from the model fits the activity from neurons the authors recorded. Relatedly, it is unclear how the parameters were chosen for the D1/D2 versions of this model. I think that an alternate approach that would answer these questions is to fit the model to each cell, and then examine the best-fit parameters, as well as the ability of the model to predict activity on trials held out from the fitting process. This would provide a more rigorous method to identify the best parameters and would directly quantify how well the model captures the data.
We are glad the reviewer raised these points. Our goal was to use neuronal activity to fit behavioral activity, not the reverse. While we understand the reviewer’s point, we note that one behavioral output (switch time) can be encoded by many patterns of neuronal activity; thus, we are not sure we can use the model developed for behavior to fit diverse neuronal activity, or an ensemble of neurons. We have made this clear in the manuscript (Line 251):
“Our model aimed to fit statistical properties of mouse behavioral responses while incorporating MSN network dynamics. However, the model does not attempt to fit individual neurons’ activity, because our model predicts a single behavioral parameter – switch time – that can be caused by the aggregation of diverse neuronal activity.”
To attempt to do something close to what the reviewer suggested, we attempted to predict behavior directly from neuronal ensembles. We have now made this clear in the methods on Line 682):
“Analysis and modeling of mouse MSN-ensemble recordings. Our preliminary analysis found that, for sufficiently large number of neurons (𝑵 > 𝟏𝟏), each recorded ensemble of MSNs on a trial-by-trial basis could predict when mice would respond. We took the following approach: First, for each MSN, we convolved its trial-by-trial spike train 𝑺𝒑𝒌(𝒕) with a 1-second exponential kernel 𝑲(𝒕) = 𝒘 𝒆-𝒕/𝒘 if 𝒕 > 𝟎 and 𝑲(𝒕) = 𝟎 if 𝒕 ≤ 𝟎 (Zhou et al., 2018; here 𝒘 = 𝟏 𝒔). Therefore, the smoothed, convolved spiking activity of neuron 𝒋 (𝒋 = 𝟏, 𝟐, … 𝑵),
tracks and accumulates the most recent (one second, in average) firing-rate history of the 𝒋-th MSN, up to moment 𝒕. We hypothesized that the ensemble activity
(𝒙𝟏(𝒕), 𝒙𝟐(𝒕), … , 𝒙𝑵(𝒕)), weighted with some weights 𝜷𝒋 , could predict the trial switch time 𝒕∗ by considering the sum
and the sigmoid
that approximates the firing rate of an output unit. Here parameter 𝒌 indicates how fast 𝒙(𝒕) crosses the threshold 0.5 coming from below (if 𝒌 > 𝟎) or coming from above (if 𝒌 < 𝟎) and relates the weights 𝜷𝒋 to the unknowns 𝜷H𝒋 = 𝜷𝒋/𝒌 and 𝜷H𝟎 = −𝟎. 𝟓/𝒌. Next, we ran a logistic fit for every trial for a given mouse over the spike count predictor matrix 7𝒙𝟏(𝒕), 𝒙𝟐(𝒕), … , 𝒙𝑵(𝒕)9 from the mouse MSN recorded ensemble, and observed value 𝒕∗, estimating the coefficients 𝜷H𝟎 and 𝜷H𝒋, and so, implicitly, the weights 𝜷𝒋. From there, we compute the predicted switch time 𝒕∗𝒑𝒓𝒆𝒅 by condition 𝒙(𝒕) = 𝟎. 𝟓. Accuracy was quantified comparing the predicted accuracy within a 1 second window to switch time on a trial-by-trial basis (Fig S4).
And in the results (Line 254):
We first analyzed trial-based aggregated activity of MSN recordings from each mouse (𝒙𝒋(𝒕)) where 𝒋 = 𝟏, … , 𝑵 neurons. For D2-MSN or D1-MSN ensembles of 𝑵 > 𝟏𝟏, we found linear combinations of their neuronal activities, with some 𝜷𝒋 coefficients,
that could predict the trial-by-trial switch response times (accuracy > 90%, Fig S4; compared with < 20% accuracy for Poisson-generated spikes of same trial-average firing rate). The predicted switch time 𝒕∗𝒑𝒓𝒆𝒅 was defined by the time when the weighted ensemble activity 𝒙(𝒕) first reached the value 𝒙) = 0.5. Finally, we built DDMs to account for this opposing trend (increasing vs decreasing) of MSN dynamics and for ensemble threshold behavior defining 𝒕∗𝒑𝒓𝒆𝒅; see the resulting model (Equations 1-3) and its simulations (Figure 4A-B).”
And we have added a new figure, Figure S4, that demonstrates these trial-by-trial predictions of switch response times.
Note that we have included predictions from shuffled data similar to what the reviewer suggested based on shuffled data. Predictions are derived from neuronal ensembles on that trial; thus we could not apply a leave-one-out approach to trial-by-trial predictions.
These models are highly predictive for larger ensembles and poorly predictive for smaller ensembles. We think this model adds to the manuscript and we are glad the reviewer suggested it.
Relatedly, looking at the raw data in Figure 2, it seems that many neurons either fire at the beginning or end of the interval, with more neurons firing at the end, and more firing at the beginning, for D2/D1 neurons respectively. Thus, it's not clear to me whether the drift-diffusion model is a good model of activity. Or, perhaps the model is supposed to be related to the aggregate activity of all D1/D2 neurons? (If so, this should be made more explicit. The comment about fitting the model directly to the data also still stands).
Our model was inspired by the aggregate activity. We have now made this clear in the results (Line 227):
“Our data demonstrate that D2-MSNs and D1-MSNs have opposite activity patterns. However, past computational models of interval timing have relied on drift-diffusion dynamics with a positive slope that accumulates evidence over time (Nguyen et al., 2020; Simen et al., 2011). To reconcile how these MSNs might complement to effect temporal control of action, we constructed a four-parameter drift-diffusion model (DDM). Our goal was to construct a DDM inspired by average differences in D2MSNs and D1-MSNs that predicted switch-response time behavior.”
Further, it's unclear to me how, or why, the authors changed the specific parameters they used to model the optogenetic manipulation. Were these parameters chosen because they fit the manipulation data? This I don't think is in itself an issue, but perhaps should be clearly stated, because otherwise it sounds a bit odd given the parameter changes are so specific. It is also not clear to me why the noise in the diffusion process would be expected to change with increased inhibition.
We have clarified that our parameters were chosen to best fit behavior (Line 266):
“The model’s parameters were chosen to fit the distribution of switch-response times:
𝑭 = 𝟏, 𝒃 = 𝟎. 𝟓𝟐 (so 𝑻 = 𝟎. 𝟖𝟕), 𝑫 = 𝟎. 𝟏𝟑𝟓, 𝝈 = 𝟎. 𝟎𝟓𝟐 for intact D2-MSNs (Fig 4A, in black); and 𝑭 = 𝟎, 𝒃 = 𝟎. 𝟒𝟖 (so 𝑻 = 𝟎. 𝟏𝟑), 𝑫 = 𝟎. 𝟏𝟒𝟏, 𝝈 = 𝟎. 𝟎𝟓𝟐 for intact D1-MSNs (Fig 4B, in black).”
Furthermore, we have clarified the approach to noise in the results (Line 247):
“The drift, together with noise 𝝃(𝒕) (of zero mean and strength 𝝈), leads to fluctuating accumulation which eventually crosses a threshold 𝑻 (see Equation 3; Fig 4A-B).”
And Line 279:
“The results were obtained by simultaneously decreasing the drift rate D (equivalent to lengthening the neurons’ integration time constant) and lowering the level of network noise 𝝈: D = 𝟎. 𝟏𝟐𝟗, 𝝈 = 𝟎. 𝟎𝟒𝟑 for D2-MSNs in Fig 4A (in red; changes in noise had to accompany changes in drift rate to preserve switch response time variance); and 𝑫 = 𝟎. 𝟏𝟐𝟐, 𝝈 = 𝟎. 𝟎𝟒𝟑 for D1-MSNs in Fig 4B (in blue). The model predicted that disrupting either D2-MSNs or D1-MSNs would increase switch response times (Fig 4C and Fig 4D) and would shift MSN dynamics.”
Regarding the results in Figure 6:
My comments regarding the interpretation of PCs in Figure 2 apply here as well. In addition, I am not sure that examining PC2 adds much here, given that the authors didn't examine such nonlinear changes earlier in the paper.
We agree – we removed PC2 for these reasons. We have also noted that the primary reason for PC1 was to compare results of D2/D1 blockade (Line 362):
“We noticed differences in MSN activity across the interval with D2 blockade and D1 blockade at the individual MSN level (Fig 6B-D) as well as at the population level (Fig 6E). We used PCA to quantify effects of D2 blockade or D1 blockade (Bruce et al., 2021; Emmons et al., 2017; Kim et al., 2017a). We constructed principal components (PC) from z-scored peri-event time histograms of firing rate from saline, D2 blockade, and D1 blockade sessions for all mice together. The first component (PC1), which explained 54% of neuronal variance, exhibited “timedependent ramping”, or monotonic changes over the 6 second interval immediately after trial start (Fig 6F-G; variance for PC1 p = 0.001 vs 46 (45-47)% variance in random data; Narayanan, 2016).”
As noted above, PC1 does not explain this level of variance in noisy data.
We also reworked Figure 6 to make the effects of D2 and D1 blockade more apparent by moving the matched sorting to the main figure:
A larger concern though that seems potentially at odds with the authors' interpretation is that there seems to be very little change in the firing pattern after D1 or D2 blockade. I see that in Figure 6F the authors suggest that many cells slope down (and thus, presumably, they are recoding more D1 cells), and that this change in slope is decreased, but this effect is not apparent in Figure 6C, and Figure 6B shows an example of a cell that seems to fire in the opposite direction (increase activity). I think it would help to show some (more) individual examples that demonstrate the summary effect shown by the authors, and perhaps the authors can comment on the robustness (or the variability) of this result.
These are important suggestions, we changed our analysis to better capture the variability and main effects in the data, exactly as the reviewer suggested. First, we now included 3 individual raster examples, exactly as the reviewer suggested
As the reviewer suggested, we wanted to compare variability for *all* MSNs. We sorted the same MSNs across saline, D2 blockade, and D1 blockade sessions. We detailed these sorting details in the methods (Line 618):
“Single-unit recordings were made using a multi-electrode recording system (Open
Ephys, Atlanta, GA). After the experiments, Plexon Offline Sorter (Plexon, Dallas, TX), was used to remove artifacts. Principal component analysis (PCA) and waveform shape were used for spike sorting. Single units were defined as those 1) having a consistent waveform shape, 2) being a separable cluster in PCA space, and 3) having a consistent refractory period of at least 2 milliseconds in interspike interval histograms. The same MSNs were sorted across saline, D2 blockade, and D1 blockade sessions by loading all sessions simultaneously in Offline Sorter and sorted using the preceding criteria. MSNs had to have consistent firing in all sessions to be included. Sorting integrity across sessions was quantified by comparing waveform similarity via correlation coefficients between sessions.”
To confirm that we were able to track neurons across sessions, we quantified waveform similarity (Line 353):
“We analyzed 99 MSNs in sessions with saline, D2 blockade, and D1 blockade. We matched MSNs across sessions based on waveform and interspike intervals; waveforms were highly similar across sessions (correlation coefficient between matched MSN waveforms: saline vs D2 blockade r = 1.00 (0.99 – 1.00 rank sum vs correlations in unmatched waveforms p = 3x10-44; waveforms; saline vs D1 blockade r = 1.00 (1.00 – 1.00), rank sum vs correlations in unmatched waveforms p = 4x10-50). There were no consistent changes in MSN average firing rate with D2 blockade or D1 blockade (F = 1.1, p = 0.30 accounting for variance between MSNs; saline: 5.2 (3.3 – 8.6) Hz; D2 blockade 5.1 (2.7 – 8.0) Hz; F = 2.2, p = 0.14; D1 blockade 4.9 (2.4 – 7.8) Hz).”
As noted above, this enabled us to compare activity for the same MSNs across sessions in a new Figure 6 (previously, this analysis had been in Figure S9), and used PCA to quantify this variability.
By tracking neurons across saline, D2 blockade, and D1 blockade, readers can see all the variability in MSNs. We added these data to the results (Line 362):
“We noticed differences in MSN activity across the interval with D2 blockade and D1 blockade at the individual MSN level (Fig 6B-D) as well as at the population level (Fig 6E). We used PCA to quantify effects of D2 blockade or D1 blockade (Bruce et al., 2021; Emmons et al., 2017; Kim et al., 2017a). We constructed principal components (PC) from z-scored peri-event time histograms of firing rate from saline, D2 blockade, and D1 blockade sessions for all mice together. The first component (PC1), which explained 54% of neuronal variance, exhibited “timedependent ramping”, or monotonic changes over the 6 second interval immediately after trial start (Fig 6F-G; variance for PC1 p = 0.001 vs 46 (45-47)% variance in random data; Narayanan, 2016). Interestingly, PC1 scores shifted with D2 blockade (Fig 6F; PC1 scores for D2 blockade: -0.6 (-3.8 – 4.7) vs saline: -2.3 (-4.2 – 3.2), F = 5.1, p = 0.03 accounting for variance between MSNs; no reliable effect of sex (F = 0.2, p = 0.63) or switching direction (F = 2.8, p = 0.10)). PC1 scores also shifted with D1 blockade (Fig 6F; PC1 scores for D1 blockade: -0.0 (-3.9 – 4.5), F = 5.8, p = 0.02 accounting for variance between MSNs; no reliable effect of sex (F = 0.0, p = 0.93) or switching direction (F = 0.9, p = 0.34)). There were no reliable differences in PC1 scores between D2 and D1 blockade. Furthermore, PC1 was distinct even when sessions were sorted independently and assumed to be fully statistically independent (Figure S10; D2 blockade vs saline: F = 5.8, p = 0.02; D1 blockade vs saline: F = 4.9, p = 0.03; all analyses accounting for variance between mice). Higher components explained less variance and were not reliably different between saline and D2 blockade or D1 blockade. Taken together, this data-driven analysis shows that D2 and D1 blockade produced similar shifts in MSN population dynamics represented by PC1. When combined with the major contributions of D1/D2 MSNs to PC1 (Fig 3C) these findings indicate that pharmacological D2 blockade and D1 blockade disrupt ramping-related activity in the striatum.”
Finally, we included the data in which sessions were sorted independently and assumed to be fully statistically independent in a new Figure S10.
And in the results (Line 376):
“Furthermore, PC1 was distinct even when sessions were sorted independently and assumed to be fully statistically independent (Figure S10; D2 blockade vs saline: F = 5.8, p = 0.02; D1 blockade vs saline: F = 4.9, p = 0.03; all analyses accounting for variance between mice). Higher components explained less variance and were not reliably different between saline and D2 blockade or D1 blockade.”
These changes strengthen the manuscript and better show the main effects and variability of the data.
Regarding the results in Figure 7:
I am overall a bit confused about what the authors are trying to claim here. In Figure 7, they present data suggesting that D1 or D2 blockade disrupts their ability to decode time in the interval of interest (0-6 seconds). However, in the final paragraph of the results, the authors seem to say that by using another technique, they didn't see any significant change in decoding accuracy after D1 or D2 blockade. What do the authors make of this?
This was very unclear. The second classifier was predicting response time, but it was confusing, and we removed it.
Impact:
The task and data presented by the authors are very intriguing, and there are many groups interested in how striatal activity contributes to the neural perception of time. The authors perform a wide variety of experiments and analysis to examine how DMS activity influences time perception during an interval-timing task, allowing for insight into this process. However, the significance of the key finding - that D2/D1 activity increases/ decreases with time - remains somewhat ambiguous to me. This arises from a lack of clarity regarding the initial hypothesis and the implications of this finding for advancing our understanding of striatal functions.
As noted above, we clarified our hypothesis and implications, and strengthened several aspects of the data as suggested by this reviewer.
Reviewer #2 (Public Review):
Summary:
In the present study, the authors investigated the neural coding mechanisms for D1- and D2expressing striatal direct and indirect pathway MSNs in interval timing by using multiple strategies. They concluded that D2-MSNs and D1-MSNs have opposing temporal dynamics yet disrupting either type produced similar effects on behavior, indicating the complementary roles of D1- and D2- MSNs in cognitive processing. However, the data was incomplete to fully support this major finding. One major reason is the heterogenetic responses within the D1-or D2MSN populations. In addition, there are additional concerns about the statistical methods used. For example, the majority of the statistical tests are based on the number of neurons, but not the number of mice. It appears that the statistical difference was due to the large sample size they used (n=32 D2-MSNs and n=41 D1-MSNs), but different neurons recorded in the same mouse cannot be treated as independent samples; they should use independent mouse-based statistical analysis.
Strengths:
The authors used multiple approaches including awake mice behavior training, optogeneticassistant cell-type specific recording, optogenetic or pharmacological manipulation, neural computation, and modeling to study neuronal coding for interval timing.
We appreciate the reviewer’s careful read recognizing the breadth of our approach.
Weaknesses:
(1) More detailed behavior results should be shown, including the rate of the success switches, and how long it takes to wait in the second nose poke to get a reward. For line 512 and the Figure 1 legend, the reviewer is not clear about the reward delivery. The methods appear to state that the mouse had to wait for 18s, then make nose pokes at the second port to get the reward. What happens if the mouse made the second nose poke before 18 seconds, but then exited? Would the mouse still get the reward at 18 seconds? Similarly, what happens if the mice made the third or more nosepokes within 18 seconds? It is important to clarify because, according to the method described, if the mice made a second nose poke before 18 seconds, this already counted as the mouse making the "switch." Lastly, what if the mice exited before 6s in the first nosepoke?
We completely agree. We have now completely revised Figure 1 to include many of these task details.
We have clarified remaining details in the methods (Line 548):
“Interval timing switch task. We used a mouse-optimized operant interval timing task described in detail previously (Balci et al., 2008; Bruce et al., 2021; Tosun et al., 2016; Weber et al., 2023). Briefly, mice were trained in sound-attenuating operant chambers, with two front nosepokes flanking either side of a food hopper on the front wall, and a third nosepoke located at the center of the back wall. The chamber was positioned below an 8-kHz, 72-dB speaker (Fig 1A; MedAssociates, St. Albans, VT). Mice were 85% food restricted and motivated with 20 mg sucrose pellets (BioServ, Flemington, NJ). Mice were initially trained to receive rewards during fixed ratio nosepoke response trials. Nosepoke entry and exit were captured by infrared beams. After shaping, mice were trained in the “switch” interval timing task. Mice self-initiated trials at the back nosepoke, after which tone and nosepoke lights were illuminated simultaneously. Cues were identical on all trial types and lasted the entire duration of the trial (6 or 18 seconds). On 50% of trials, mice were rewarded for a nosepoke after 6 seconds at the designated first ‘front’ nosepoke; these trials were not analyzed. On the remaining 50% of trials, mice were rewarded for nosepoking first at the ‘first’ nosepoke location and then switching to the ‘second’ nosepoke location; the reward was delivered for initial nosepokes at the second nosepoke location after 18 seconds when preceded by a nosepoke at the first nosepoke location. Multiple nosepokes at each nosepokes were allowed. Early responses at the first or second nosepoke were not reinforced. Initial responses at the second nosepoke rather than the first nosepoke, alternating between nosepokes, going back to the first nosepoke after the second nosepoke were rare after initial training. Error trials included trials where animals responded only at the first or second nosepoke and were also not reinforced. We did not analyze error trials as they were often too few to analyze; these were analyzed at length in our prior work (Bruce et al., 2021).
Switch response time was defined as the moment animals departed the first nosepoke before arriving at the second nosepoke. Critically, switch responses are a time-based decision guided by temporal control of action because mice switch nosepokes only if nosepokes at the first location did not receive a reward after 6 seconds. That is, mice estimate if more than 6 seconds have elapsed without receiving a reward to decide to switch responses. Mice learn this task quickly (3-4 weeks), and error trials in which an animal nosepokes in the wrong order or does not nosepoke are relatively rare and discarded. Consequently, we focused on these switch response times as the key metric for temporal control of action. Traversal time was defined as the duration between first nosepoke exit and second nosepoke entry and is distinct from switch response time when animals departed the first nosepoke. Nosepoke duration was defined as the time between first nosepoke entry and exit for the switch response times only. Trials were self-initiated, but there was an intertrial interval with a geometric mean of 30 seconds between trials.”
And in the results on Line 131:
“We investigated cognitive processing in the striatum using a well-described mouseoptimized interval timing task which requires mice to respond by switching between two nosepokes after a 6-second interval (Fig 1A; see Methods; (Balci et al., 2008; Bruce et al., 2021; Larson et al., 2022; Tosun et al., 2016; Weber et al., 2023)). In this task, mice initiate trials by responding at a back nosepoke, which triggers auditory and visual cues for the duration of the trial. On 50% of trials, mice were rewarded for nosepoking after 6 seconds at the designated ‘first’ front nosepoke; these trials were not analyzed. On the remaining 50% of trials, mice were rewarded for nosepoking at the ‘first’ nosepoke and then switching to the ‘second’ nosepoke; initial nosepokes at the second nosepoke after 18 seconds triggered reward when preceded by a first nosepoke. The first nosepokes occurred before switching responses and the second nosepokes occurred much later in the interval in anticipation of reward delivery at 18 seconds (Fig 1B-D). During the task, movement velocity peaked before 6 seconds as mice traveled to the front nosepoke (Fig 1E).
We focused on the switch response time, defined as the moment mice exited the first nosepoke before entering the second nosepoke. Switch responses are a timebased decision guided by temporal control of action because mice switch nosepokes only if nosepoking at the first nosepokes does not lead to a reward after 6 seconds (Fig 1B-E). Switch responses are guided by internal estimates of time because no external cue indicates when to switch from the first to the second nosepoke (Balci et al., 2008; Bruce et al., 2021; Tosun et al., 2016; Weber et al., 2023). We defined the first 6 seconds after trial start as the ‘interval’, because during this epoch mice are estimating whether 6 seconds have elapsed and if they need to switch responses. In 30 mice, switch response times were 9.3 seconds (8.4 – 9.7; median (IQR)); see Table 1 for a summary of mice, experiments, trials, and sessions). We studied dorsomedial striatal D2-MSNs and D1-MSNs using a combination of optogenetics and neuronal ensemble recordings in 9 transgenic mice (4 D2-Cre mice switch response time 9.7 (7.0 – 10.3) seconds; 5 D1-Cre mice switch response time 8.2 (7.7 – 8.7) seconds; rank sum p = 0.73; Table 1).”
(2) There are a lot of time parameters in this behavior task, the description of those time parameters is mentioned in several parts, in the figure legend, supplementary figure legend, and methods, but was not defined clearly in the main text. It is inconvenient, sometimes, confusing for the readers. The authors should make a schematic diagram to illustrate the major parameters and describe them clearly in the main text.
We agree. We have clarified this in a new schematic, shading the interval in gray:
And in the results on line 131:
“We focused on the switch response time, defined as the moment mice exited the first nosepoke before entering the second nosepoke. Switch responses are a time-based decision guided by temporal control of action because mice switch nosepokes only if nosepoking at the first nosepokes does not lead to a reward after 6 seconds (Fig 1BE). Switch responses are guided by internal estimates of time because no external cue indicates when to switch from the first to the second nosepoke (Balci et al., 2008; Bruce et al., 2021; Tosun et al., 2016; Weber et al., 2023). We defined the first 6 seconds after trial start as the ‘interval’, because during this epoch mice are estimating whether 6 seconds have elapsed and if they need to switch responses. In 30 mice, switch response times were 9.3 seconds (8.4 – 9.7; median (IQR)); see Table 1 for a summary of mice, experiments, trials, and sessions). We studied dorsomedial striatal D2-MSNs and D1-MSNs using a combination of optogenetics and neuronal ensemble recordings in 9 transgenic mice (4 D2-Cre mice switch response time 9.7
(7.0 – 10.3) seconds; 5 D1-Cre mice switch response time 8.2 (7.7 – 8.7) seconds; rank sum p = 0.73; Table 1).”
(3) In Line 508, the reviewer suggests the authors pay attention to those trials without "switch". It would be valuable to compare the MSN activity between those trials with or without a "switch".
This is a great suggestion. We analyzed such error trials and MSN activity in Figure 6 of Bruce et al., 2021. However, this manuscript was not designed to analyze errors, as they are rare beyond initial training (Bruce et al., 2021 focused on early training), and too inconsistent to permit robust analysis. This was added to the methods on Line 567:
“Early responses at the first or second nosepoke were not reinforced. Initial responses at the second nosepoke rather than the first nosepoke, alternating between nosepokes, going back to the first nosepoke after the second nosepoke were rare after initial training. Error trials included trials where animals responded only at the first or second nosepoke and were also not reinforced. We did not analyze error trials as they were often too few to analyze; these were analyzed at length in our prior work (Bruce et al., 2021).”
(4) The definition of interval is not very clear. It appears that the authors used a 6-second interval in analyzing the data in Figure 2 and Figure 3. But from my understanding, the interval should be the time from time "0" to the "switch", when the mice start to exit from the first nose poke.
We have now defined it explicitly in the schematic:
Incidentally, this reviewer asked us to analyze a longer epoch – this analysis beautifully justifies our focus on the first 6 seconds (now in Figure S2).
We focus on the first six seconds as there are few nosepokes and switch responses during this epoch; however, we consider the reviewer’s definition and analyze the epoch the reviewer suggests from 0 to the switch in analyses below.
(5) For Figure 2 C-F, the authors only recorded 32 D2-MSNs in 4 mice, and 41 D1-MSNs in 5 mice. The sample size is too small compared to the sample size usually used in the field. In addition to the small sample size, the single-cell activity exhibited heterogeneity, which created potential issues.
We are glad the reviewer raised these points. First, our tagging dataset is relatively standard for optogenetic tagging. Second, we now include Cohen’s d for both PC and slope results for all optogenetic tagging analysis, which demonstrate that we have adequate statistical power and medium-to-large effect sizes (Line 186):
“In line with population averages from Fig 2G&H, D2-MSNs and D1-MSNs had opposite patterns of activity with negative PC1 scores for D2-MSNs and positive PC1 scores for D1-MSNs (Fig 3C; PC1 for D2-MSNs: -3.4 (-4.6 – 2.5); PC1 for D1MSNs: 2.8 (-2.8 – 4.9); F = 8.8, p = 0.004 accounting for variance between mice (Fig S3A); Cohen’s d = 0.7; power = 0.80; no reliable effect of sex (F = 0.44, p = 0.51) or switching direction (F = 1.73, p = 0.19)).”
And Line 197:
“GLM analysis also demonstrated that D2-MSNs had significantly different slopes (0.01 spikes/second (-0.10 – 0.10)), which were distinct from D1-MSNs (-0.20 (-0.47– 0.06; Fig 3D; F = 8.9, p = 0.004 accounting for variance between mice (Fig S3B); Cohen’s d = 0.8; power = 0.98; no reliable effect of sex (F = 0.02, p = 0.88) or switching direction (F = 1.72, p = 0.19)).”
We added boxplots to Figure 3, which better highlight differences in these distributions.
However, the reviewer’s point is well-taken, and we have added a caveat to the discussion exactly as the reviewer suggested (Line 496):
“Second, although we had adequate statistical power and medium-to-large effect sizes, optogenetic tagging is low-yield, and it is possible that recording more of these neurons would afford greater opportunity to identify more robust results and alternative coding schemes, such as neuronal synchrony.”
For both D1 and D2 MSNs, the authors tried to make conclusions on the "trend" of increasing in D2-MSNs and decreasing in D1-MSNs populations, respectively, during the interval. However, such a conclusion is not sufficiently supported by the data presented. It looks like the single-cell activity patterns can be separated into groups: one is a decreasing activity group, one is an increasing activity group and a small group for on and off response. Because of the small sample size, the author should pay attention to the variance across different mice (which needs to be clearly presented in the manuscript), instead of pooling data together and analyzing the mean activity.
We were not clear – we now do exactly as the reviewer suggested. We are not pooling any data – instead – as we state on line 620 - we are using linear-mixed effects models to account for mouse-specific and neuron-specific variance. This approach was developed with our statistics core for exactly the reasons the reviewer suggested (see letter). We state this explicitly in the methods (Line 704):
“Statistics. All data and statistical approaches were reviewed by the Biostatistics,
Epidemiology, and Research Design Core (BERD) at the Institute for Clinical and Translational Sciences (ICTS) at the University of Iowa. All code and data are made available at http://narayanan.lab.uiowa.edu/article/datasets. We used the median to measure central tendency and the interquartile range to measure spread. We used Wilcoxon nonparametric tests to compare behavior between experimental conditions and Cohen’s d to calculate effect size. Analyses of putative single-unit activity and basic physiological properties were carried out using custom routines for MATLAB.
For all neuronal analyses, variability between animals was accounted for using generalized linear-mixed effects models and incorporating a random effect for each mouse into the model, which allows us to account for inherent between-mouse variability. We used fitglme in MATLAB and verified main effects using lmer in R. We accounted for variability between MSNs in pharmacological datasets in which we could match MSNs between saline, D2 blockade, and D1 blockade. P values < 0.05 were interpreted as significant.”
We have now stated in the results that we are explicitly accounting for variance between mice (Line 186):
“In line with population averages from Fig 2G&H, D2-MSNs and D1-MSNs had opposite patterns of activity with negative PC1 scores for D2-MSNs and positive PC1 scores for D1-MSNs (Fig 3C; PC1 for D2-MSNs: -3.4 (-4.6 – 2.5); PC1 for D1MSNs: 2.8 (-2.8 – 4.9); F = 8.8, p = 0.004 accounting for variance between mice (Fig S3A); Cohen’s d = 0.7; power = 0.80; no reliable effect of sex (F = 0.44, p = 0.51) or switching direction (F = 1.73, p = 0.19)).”
And on Line 197:
“GLM analysis also demonstrated that D2-MSNs had significantly different slopes (0.01 spikes/second (-0.10 – 0.10)), which were distinct from D1-MSNs (-0.20 (-0.47– 0.06; Fig 3D; F = 8.9, p = 0.004 accounting for variance between mice (Fig S3B); Cohen’s d = 0.8; power = 0.98; no reliable effect of sex (F = 0.02, p = 0.88) or switching direction (F = 1.72, p = 0.19)).”
All statistics in the manuscript now explicitly account for variance between mice.
This is the approach that was recommended by our the Biostatistics, Epidemiology, and
Research Design Core (BERD) at the Institute for Clinical and Translational Sciences (ICTS) at the University of Iowa, who reviews all of our work.
We note that these Cohen d values usually interpret as medium or large.
We performed statistical power calculations and include these to aid readers’ interpretation. These are all >0.8.
Finally, the reviewer uses the word ‘trend’. We define p values <0.05 as significant in the methods, and do not interpret trends (on line 717):
“P values < 0.05 were interpreted as significant.”
And, we have now plotted values for each mouse in a new Figure S3.
As noted in the figure legend, mouse-specific effects were analyzed using linear models that account for between-mouse variability, as discussed with our statisticians. However, the reviewer’s point is well taken, and we have added this idea to the discussion as suggested (Line 496):
“Second, although we had adequate statistical power and medium-to-large effect sizes, optogenetic tagging is low-yield, and it is possible that recording more of these neurons would afford greater opportunity to identify more robust results and alternative coding schemes, such as neuronal synchrony.”
(6) For Figure 2, from the activity in E and F, it seems that the activity already rose before the trial started, the authors should add some longer baseline data before time zero for clarification and comparison and show the timing of the actual start of the activity with the corresponding behavior. What behavior states are the mice in when initiating the activity?
This is a key point. First, we are not certain what state the animal is in until they initiate trials at the back nosepoke (“Start”). Therefore, we cannot analyze this epoch.
However, we can show neuronal activity during a longer epoch exactly as the reviewer suggested. Although there are modulations, the biggest difference between D2 and D1 MSNs is during the 0-6 second interval. This analysis supports our focus on the 0-6 second interval. We have included this as a new Figure S2.
(7) The authors were focused on the "switch " behavior in the task, but they used an arbitrary 6s time window to analyze the activity, and tried to correlate the decreasing or increasing activities of MSNs to the neural coding for time. A better way to analyze is to sort the activity according to the "switch" time, from short to long intervals. This way, the authors could see and analyze whether the activity of D1 or D2 MSNs really codes for the different length of interval, instead of finding a correlation between average activity trends and the arbitrary 6s time window.
This is a great suggestion. We did exactly this and adjusted our linear models on a trialby-trial basis to account for time between the start of the interval and the switch. This is now added to the methods (line 656):
“We performed additional sensitivity analysis excluding outliers and measuring firing rate from the start of the interval to the time of the switch response on a trialby-trial level for each neuron.”
And to the results (Line 201):
“We found that D2-MSNs and D1-MSNs had a significantly different slope even when excluding outliers (4 outliers excluded outside of 95% confidence intervals; F=7.51, p=0.008 accounting for variance between mice) and when the interval was defined as the time between trial start and the switch response on a trial-by-trial basis for each neuron (F=4.3, p=0.04 accounting for variance between mice).”
We now state our justification for focusing on the first 6 seconds of the interval (Line 134)
“Switch responses are guided by internal estimates of time and temporal control of action because no external cue indicates when to switch from the first to the second nosepoke (Balci et al., 2008; Bruce et al., 2021; Tosun et al., 2016; Weber et al., 2023). We defined the first 6 seconds after trial start as the ‘interval’, because during this epoch mice are estimating whether 6 seconds have elapsed and if they need to switch responses.”
As noted previously, epoch is now justified by Figure S2E.
And we note that this focus minimizes motor confounds (Line 511):
“Four lines of evidence argue that our findings cannot be directly explained by motor confounds: 1) D2-MSNs and D1-MSNs diverge between 0-6 seconds after trial start well before the first nosepoke (Fig S2), 2) our GLM accounted for nosepokes and nosepoke-related βs were similar between D2-MSNs and D1-MSNs, 3) optogenetic disruption of dorsomedial D2-MSNs and D1-MSNs did not change task-specific movements despite reliable changes in switch response time, and 4) ramping dynamics were quite distinct from movement dynamics. Furthermore, disrupting D2-MSNs and D1-MSNs did not change the number of rewards animals received, implying that these disruptions did not grossly affect motivation. Still, future work combining motion tracking with neuronal ensemble recording and optogenetics and including bisection tasks may further unravel timing vs. movement in MSN dynamics (Robbe, 2023).”
We are glad the reviewer suggested this analysis as it strengthens our manuscript.
Reviewer #3 (Public Review):
Summary:
The cognitive striatum, also known as the dorsomedial striatum, receives input from brain regions involved in high-level cognition and plays a crucial role in processing cognitive information. However, despite its importance, the extent to which different projection pathways of the striatum contribute to this information processing remains unclear. In this paper, Bruce et al. conducted a study using a range of causal and correlational techniques to investigate how these pathways collectively contribute to interval timing in mice. Their results were consistent with previous research, showing that the direct and indirect striatal pathways perform opposing roles in processing elapsed time. Based on their findings, the authors proposed a revised computational model in which two separate accumulators track evidence for elapsed time in opposing directions. These results have significant implications for understanding the neural mechanisms underlying cognitive impairment in neurological and psychiatric disorders, as disruptions in the balance between direct and indirect pathway activity are commonly observed in such conditions.
Strengths:
The authors employed a well-established approach to study interval timing and employed optogenetic tagging to observe the behavior of specific cell types in the striatum. Additionally, the authors utilized two complementary techniques to assess the impact of manipulating the activity of these pathways on behavior. Finally, the authors utilized their experimental findings to enhance the theoretical comprehension of interval timing using a computational model.
We are grateful for the reviewer’s consideration of our work and for recognizing the strengths of our approach.
Weaknesses:
The behavioral task used in this study is best suited for investigating elapsed time perception, rather than interval timing. Timing bisection tasks are often employed to study interval timing in humans and animals.
This is a key point, and the reviewer is correct. We use our task because of its’ translational validity; as far as we know, temporal bisection tasks have been used less often in human disease and in rodent models. We have included a new paragraph describing this in the discussion (Line 472):
“Because interval timing is reliably disrupted in human diseases of the striatum such as Huntington’s disease, Parkinson’s disease, and schizophrenia (Hinton et al., 2007; Singh et al., 2021; Ward et al., 2011), these results have relevance to human disease. Our task version has been used extensively to study interval timing in mice and humans (Balci et al., 2008; Bruce et al., 2021; Stutt et al., 2024; Tosun et al., 2016; Weber et al., 2023). However, temporal bisection tasks, in which animals hold during a temporal cue and respond at different locations depending on cue length, have advantages in studying how animals time an interval because animals are not moving while estimating cue duration (Paton and Buonomano, 2018; Robbe, 2023; Soares et al., 2016). Our interval timing task version – in which mice switch between two response nosepokes to indicate their interval estimate has elapsed – has been used extensively in rodent models of neurodegenerative disease (Larson et al., 2022; Weber et al., 2024, 2023; Zhang et al., 2021), as well as in humans (Stutt et al., 2024). Furthermore, because many therapeutics targeting dopamine receptors are used clinically, these findings help describe how dopaminergic drugs might affect cognitive function and dysfunction. Future studies of D2-MSNs and D1-MSNs in temporal bisection and other timing tasks may further clarify the relative roles of D2- and D1-MSNs in interval timing and time estimation.”
Furthermore, we have modified the use of the definition of interval timing in the abstract, introduction, and results to reflect the reviewers comment. For instance, in the abstract (Line 43):
“We studied dorsomedial striatal cognitive processing during interval timing, an elementary cognitive task that requires mice to estimate intervals of several seconds and involves working memory for temporal rules as well as attention to the passage of time.”
However, we think it is important to use the term ‘interval timing’ as it links to past work by our group and others.
The main results from unit recording (opposing slopes of D1/D2 cell firing rate, as shown in Figure 3D) appear to be very sensitive to a couple of outlier cells, and the predictive power of ensemble recording seems to be only slightly above chance levels.
This is a key point raised by other reviewers as well. We have now included measures of statistical power (as we interpret the reviewer’s comment of predictive power), effect size, and perform additional sensitivity analyses (Line 187):
“PC1 scores for D1-MSNs (Fig 3C; PC1 for D2-MSNs: -3.4 (-4.6 – 2.5); PC1 for D1MSNs: 2.8 (-4.9 – -2.8); F=8.8, p = 0.004 accounting for variance between mice (Fig S3A); Cohen’s d = 0.7; power = 0.80; no reliable effect of sex (F=1.9, p=0.17) or switching direction (F=0.1, p=0.75)).”
And on Line 197:
“GLM analysis also demonstrated that D2-MSNs had significantly different slopes (0.01 spikes/second (-0.10 – 0.10)), which were distinct from D1-MSNs (-0.20 (-0.45– 0.06; Fig 3D; F=8.9, p = 0.004 accounting for variance between mice (Fig S3B); Cohen’s d = 0.8; power = 0.98). We found that D2-MSNs and D1-MSNs had a significantly different slope even when excluding outliers (4 outliers excluded outside of 95% confidence intervals; F=7.51, p=0.008 accounting for variance between mice) and when the interval was defined as the time between trial start and the switch response on a trial-by-trial basis for each neuron (F=4.3, p=0.04 accounting for variance between mice).”
These are medium-to-large Cohen’s d results, and we have adequate statistical power. These results are not easily explained by chance.
We also added boxplots, which highlight the differences in distribution.
Finally, we note that our conclusions are drawn from many convergent analyses (on Line 216):
“Analyses of average activity, PC1, and trial-by-trial firing-rate slopes over the interval provide convergent evidence that D2-MSNs and D1-MSNs had distinct and opposing dynamics during interval timing.”
In the optogenetic experiment, the laser was kept on for too long (18 seconds) at high power (12 mW). This has been shown to cause adverse effects on population activity (for example, through heating the tissue) that are not necessarily related to their function during the task epochs.
This is an important point. We are well aware of heating effects with optogenetics and other potential confounds. For the exact reasons noted by the reviewer, we had opsinnegative controls – where the laser was on for the exact same amount of time (18 seconds) and at the same power (12 mW)– in Figure S5. We have now better highlighted these controls in the methods (Line 598):
“In animals injected with optogenetic viruses, optical inhibition was delivered via bilateral patch cables for the entire trial duration of 18 seconds via 589-nm laser light at 12 mW power on 50% of randomly assigned trials. We performed control experiments in mice without opsins using identical laser parameters in D2-cre or D1-cre mice (Fig S6).”
And in results (Line 298):
“Importantly, we found no reliable effects for D2-MSNs with opsin-negative controls (Fig S6).”
And Line 306):
“As with D2-MSNs, we found no reliable effects with opsin-negative controls in D1MSNs (Fig S6).”
We have highlighted these data in Figure S6:
Furthermore, the effect of optogenetic inhibition is similar to pharmacological effects in this manuscript and in our prior work (De Corte et al., 2019; Stutt et al., 2024) on line 459):
“Past pharmacological work from our group and others has shown that disrupting D2- or D1-MSNs slows timing (De Corte et al., 2019b; Drew et al., 2007, 2003; Stutt et al., 2024), in line with pharmacological and optogenetic results in this manuscript.”
And in the discussion section on Line 488:
“Our approach has several limitations. First, systemic drug injections block D2- and D1-receptors in many different brain regions, including the frontal cortex, which is involved in interval timing (Kim et al., 2017a). D2 blockade or D1 blockade may have complex effects, including corticostriatal or network effects that contribute to changes in D2-MSN or D1-MSN ensemble activity. We note that optogenetic inhibition of D2-MSNs and D1-MSNs produces similar effects to pharmacology in Figure 5.”
Given the systemic delivery of pharmacological interventions, it is difficult to conclude that the effects are specific to the dorsomedial striatum. Future studies should use the local infusion of drugs into the dorsomedial striatum.
This is a great point - we did this experiment in De Corte et al, 2019 with local drug infusions. This earlier study was the departure point for this experiment. We now point this out in the introduction (Line 92):
“Past work has shown that disrupting either D2-dopamine receptors (D2) or D1dopamine receptors (D1) powerfully impairs interval timing by increasing estimates of elapsed time (Drew et al., 2007; Meck, 2006). Similar behavioral effects were found with systemic (Stutt et al., 2024) or local dorsomedial striatal D2 or D1 disruption (De Corte et al., 2019a). These data lead to the hypothesis that D2 MSNs and D1 MSNs have similar patterns of ramping activity across a temporal interval.”
However, the reviewer makes a great point - and we will develop this in our future work (Line 485):
“Future studies might extend our work combining local pharmacology with neuronal ensemble recording.”
Recommendations for the authors:
Reviewer #1 (Recommendations For The Authors):
Just a few minor notes:
(1) Figures 2C and D should have error bars.
We agree. We added error bars to these figures and other rasters as recommended.
(2) Figures 2G and H seem to be smoothed - how was this done?
We added these details.
(3) It is unclear what the 'neural network machine learning classifier' mentioned in lines 193-199 adds if the data relevant to this analysis isn't presented. I would potentially include this.
We agree. This analysis was confusing and not relevant to our main points; consequently, we removed it.
Reviewer #2 (Recommendations For The Authors):
Major:
(1) For Figure 2, the description of the main results in (C-F) in the main text is too brief and is not clear.
We have added to and clarified this text (Line 147)
“Striatal neuronal populations are largely composed of MSNs expressing D2dopamine or D1-dopamine receptors. We optogenetically tagged D2-MSNs and D1MSNs by implanting optrodes in the dorsomedial striatum and conditionally expressing channelrhodopsin (ChR2; Fig S1) in 4 D2-Cre (2 female) and 5 D1-Cre transgenic mice (2 female). This approach expressed ChR2 in D2-MSNs or D1MSNs, respectively (Fig 2A-B; Kim et al., 2017a). We identified D2-MSNs or D1MSNs by their response to brief pulses of 473 nm light; neurons that fired within 5 milliseconds were considered optically tagged putative D2-MSNs (Fig S1B-C). We tagged 32 putative D2-MSNs and 41 putative D1-MSNs in a single recording session during interval timing. There were no consistent differences in overall firing rate between D2-MSNs and D1-MSNs (D2-MSNs: 3.4 (1.4 – 7.2) Hz; D1-MSNs 5.2 (3.1 – 8.6) Hz; F = 2.7, p = 0.11 accounting for variance between mice). Peri-event rasters and histograms from a tagged putative D2-MSN (Fig 2C) and from a tagged putative D1-MSN (Fig 2D) demonstrate prominent modulations for the first 6 seconds of the interval after trial start. Z-scores of average peri-event time histograms (PETHs) from 0 to 6 seconds after trial start for each putative D2-MSN are shown in Fig 2E and for each putative D1-MSN in Fig 2F. These PETHs revealed that for the 6-second interval immediately after trial start, many putative D2-MSN neurons appeared to ramp up while many putative D1-MSNs appeared to ramp down. For 32 putative D2-MSNs average PETH activity increased over the 6second interval immediately after trial start, whereas for 41 putative D1-MSNs, average PETH activity decreased. These differences resulted in distinct activity early in the interval (0-1 seconds; F = 6.0, p = 0.02 accounting for variance between mice), but not late in the interval (5-6 seconds; F = 1.9, p = 0.17 accounting for variance between mice) between D2-MSNs and D1-MSNs. Examination of a longer interval of 10 seconds before to 18 seconds after trial start revealed the greatest separation in D2-MSN and D1-MSN dynamics during the 6-second interval after trial start (Fig S2). Strikingly, these data suggest that D2-MSNs and D1-MSNs might display opposite dynamics during interval timing.”
(2) For Figure3
(A) Is the PC1 calculated from all MSNs of all mice (4 D2, 5 D1 mice)?
We clarified this (Line 182):
“We analyzed PCA calculated from all D2-MSNs and D1-MSNs PETHs over the 6second interval immediately after trial start.”
And for pharmacology (Line 362):
“We noticed differences in MSN activity across the interval with D2 blockade and D1 blockade at the individual MSN level (Fig 6B-D) as well as at the population level (Fig 6E). We used PCA to quantify effects of D2 blockade or D1 blockade (Bruce et al., 2021; Emmons et al., 2017; Kim et al., 2017a). We constructed principal components (PC) from z-scored peri-event time histograms of firing rate from saline, D2 blockade, and D1 blockade sessions for all mice together.”
(B) The authors should perform PCA on single mouse data, and add the plot and error bar.
This is a great idea. We have now included this as a new Figure S3:
(C) As mentioned before, both D2-or D1- MSNs can be divided into three groups, it is not appropriate to put them together as each MSN is not an independent variable, the authors should do the statistics based on the individual mouse, and do the parametric or non-parametric comparison, and plot N (number of mice) based error bars.
We have done exactly this using a linear mixed effects model, as recommend by our statistics core. They have explicitly suggested that this is the best approach to these data (see letter). We have also included measures of statistical power and effect size (Line 704):
“All data and statistical approaches were reviewed by the Biostatistics, Epidemiology, and Research Design Core (BERD) at the Institute for Clinical and Translational Sciences (ICTS) at the University of Iowa. All code and data are made available at http://narayanan.lab.uiowa.edu/article/datasets. We used the median to measure central tendency and the interquartile range to measure spread. We used Wilcoxon nonparametric tests to compare behavior between experimental conditions and Cohen’s d to calculate effect size. Analyses of putative single-unit activity and basic physiological properties were carried out using custom routines for MATLAB.
For all neuronal analyses, variability between animals was accounted for using generalized linear-mixed effects models and incorporating a random effect for each mouse into the model, which allows to account for inherent between-mouse variability. We used fitglme in MATLAB and verified main effects using lmer in R. We accounted for variability between MSNs in pharmacological datasets in which we could match MSNs between saline, D2 blockade, and D1 blockade. P values < 0.05 were interpreted as significant.”
We have now included measures of ‘power’ (which we interpret to be statistical), effect size, and perform additional sensitivity analyses (Line 187):
“PC1 scores for D1-MSNs (Fig 3C; PC1 for D2-MSNs: -3.4 (-4.6 – 2.5); PC1 for D1MSNs: 2.8 (-4.9 – -2.8); F=8.8, p = 0.004 accounting for variance between mice (Fig S3A); Cohen’s d = 0.7; power = 0.80; no reliable effect of sex (F=1.9, p=0.17) or switching direction (F=0.1, p=0.75)).”
And Line 197:
“GLM analysis also demonstrated that D2-MSNs had significantly different slopes (0.01 spikes/second (-0.10 – 0.10)), which were distinct from D1-MSNs (-0.20 (-0.45– 0.06; Fig 3D; F=8.9, p = 0.004 accounting for variance between mice (Fig S3B); Cohen’s d = 0.8; power = 0.98). We found that D2-MSNs and D1-MSNs had a significantly different slope even when excluding outliers (4 outliers excluded outside of 95% confidence intervals; F=7.51, p=0.008 accounting for variance between mice) and when the interval was defined as the time between trial start and the switch response on a trial-by-trial bases for each neuron (F=4.3, p=0.04 accounting for variance between mice).”
These are medium-to-large Cohen’s d results, and we have adequate statistical power. These results are not easily explained by chance.
We also added boxplots, which highlight the differences in distributions.
(3) For results in Figure 5 and Figure S7, according to Figure 1 legend, lines 4 to 5, the response times were defined as the moment mice exit the first nose poke (on the left) to respond at the second nose poke; and according to method session (line 522), "switch" traversal time was defined as the duration between first nose poke exit and second nose poke entry. It seems that response time is the switch traversal time, they should be the same, but in Figures B and D, the response time showed a clear difference between the laser off and on groups, while in Figures S7 C, and G, there were no differences between laser off and on group for switch traversal time. Please reconcile these inconsistencies.
We were not clear. We now clarify – switch responses are the moment when mice depart the first nosepoke, whereas traversal time is the time between departing the first nosepoke and arriving at the second nosepoke. We have reworked our figures to make this clear.
And in the methods (Line 570):
“Switch response time was defined as the moment animals departed the first nosepoke before arriving at the second nosepoke. Critically, switch responses are a time-based decision guided by temporal control of action because mice switch nosepokes only if nosepokes at the first location did not receive a reward after 6 seconds. That is, mice estimate if more than 6 seconds have elapsed without receiving a reward to decide to switch responses. Mice learn this task quickly (3-4 weeks), and error trials in which an animal nosepokes in the wrong order or does not nosepoke are relatively rare and discarded. Consequently, we focused on these switch response times as the key metric for temporal control of action. Traversal time was defined as the duration between first nosepoke exit and second nosepoke entry and is distinct from switch response time when animals departed the first nosepoke. Nosepoke duration was defined as the time between first nosepoke entry and exit for the switch response times only. Trials were self-initiated, but there was an intertrial interval with a geometric mean of 30 seconds between trials.”
And in Figure S8, we have added graphics and clarified the legend.
(4) The first nose poke and second nose poke are very close, why did it take so long to move from the first nose poke to the second nose poke, even though the mouse already made the decision to switch? Please see Figure S1A, it took less than 6s from the back nose poke to the first nose poke, but it took more than 6s (up to 12s) from the first nose poke to the second nose poke, what were the mice's behavior during this period?
This is a key detail. There is no temporal urgency as only the initial nosepoke after 18 seconds leads to reward. In other words, making a second nosepoke prior to 18 seconds is not rewarded and, in well-trained animals, is wasted effort. We have added these details to the methods (Line 124):
“On the remaining 50% of trials, mice were rewarded for nosepoking at the ‘first’ nosepoke and then switching to the ‘second’ nosepoke; initial nosepokes at the second nosepoke after 18 seconds triggered reward when preceded by a first nosepoke. The first nosepokes occurred before switching responses and the second nosepokes occurred much later in the interval in anticipation of reward delivery at 18 seconds (Fig 1B-D). During the task, movement velocity peaked before 6 seconds as mice traveled to the front nosepoke (Fig 1E).”
And in Figure 1, as described in detail above.
(5) How many trials did mice perform in one day? How many recordings/day for how many days were performed?
These are key details that we have now added to Table 1.
We have added the number of recording sessions to the methods (Line 603):
“For optogenetic tagging, putative D1- and D2-MSNs were optically identified via 473-nm photostimulation. Units with mean post-stimulation spike latencies of ≤5 milliseconds and a stimulated-to-unstimulated waveform correlation ratio of >0.9 were classified as putative D2-MSNs or D1-MSNs (Ryan et al., 2018; Shin et al., 2018). Only one recording session was performed for each animal per day, and one recording session was included from each animal.”
And Line 606:
“Only one recording session was performed for each animal per day, and one recording session was included from saline, D2 blockade, and D1 blockade sessions.”
(6) For results in Figure 5, the authors should analyze the speed for the laser on and off group, since the dorsomedial striatum was reported to be related to control of speed (Yttri, Eric A., and Joshua T. Dudman. "Opponent and bidirectional control of movement velocity in the basal ganglia." Nature 533.7603 (2016): 402-406.).
We have some initial DeepLabCut data and have included it in a new Figure 1E.
B) DeepLabCut tracking of position during the interval timing revealed that mice moved quickly after trial start and then velocity was relatively constant throughout the trial
We measure movement speed using nosepoke duration and traversal time, which can give some measure of movement velocity.
In Yttri and Dudman, the mice are head-fixed and moving a joystick, whereas our mice are freely moving. However, we have now included the lack of motor control as a major limitation (Line 510):
“Finally, movement and motivation contribute to MSN dynamics (Robbe, 2023). Four lines of evidence argue that our findings cannot be directly explained by motor confounds: 1) D2-MSNs and D1-MSNs diverge between 0-6 seconds after trial start well before the first nosepoke (Fig S2), 2) our GLM accounted for nosepokes and nosepoke-related βs were similar between D2-MSNs and D1-MSNs, 3) optogenetic disruption of dorsomedial D2-MSNs and D1-MSNs did not change task-specific movements despite reliable changes in switch response time, and 4) ramping dynamics were quite distinct from movement dynamics. Furthermore, disrupting D2-MSNs and D1-MSNs did not change the number of rewards animals received, implying that these disruptions did not grossly affect motivation. Still, future work combining motion tracking with neuronal ensemble recording and optogenetics and including bisection tasks may further unravel timing vs. movement in MSN dynamics (Robbe, 2023).”
(7) Figure S3 (C, E, and F), statistics should be done based on N (number of mice), not on the number of recorded neurons.
We have removed this section, and all other statistics in the paper properly account for mouse-specific variance, as noted above.
(8) Figure S1
(A) Are these the results from all mice superposed together, or from one mouse on one given day? How many of the trials' data were superposed?
We included these details in a new Figure 1.
(B, C) How many trials were included?
(D) How many days did these data cover?
We have included a new Table 1 with these important details.
We have noted that only 1 recording session / mouse was included in analysis (Line 606):
“Only one recording session was performed for each animal per day, and one recording session was included from each animal.”
And Line 614:
“Only one recording session was performed for each animal per day, and one recording session was included from saline, D2 blockade, and D1 blockade sessions.”
(9) Figure S2
(A) Can the authors add coordinates of the brain according to the mouse brain atlas or, alternatively, show it using a coronal section?
Great idea – added to Figure S2 legend:
“Figure S1: A) Recording locations in the dorsomedial striatum (targeting AP +0.4, ML -1.4, DV -2.7). Electrode reconstructions for D2-Cre (red), D1-Cre (blue), and wild-type mice (green). Only the left striatum was implanted with electrodes in all animals.”
We have also added it to Figure S5 legend:
“Figure S5: Fiber optic locations from A) an opsin-expressing mouse with mCherrytagged halorhodopsin and bilateral fiber optics, and B) across 10 D2-Cre mice (red) and 6 D1-cre mice (blue) with fiber optics (targeting AP +0.9, ML +/-1.3, DV –2.5).”
(C) Why did the waveform of laser and no laser seem the same?
The optogenetically tagged spike waveforms are highly similar, indicating that optogenetically-triggered spikes are like other spikes. That is the main point – optogenetically stimulating the neuron does not change the waveform. We have added this detail to the legend of S1:
“Inset on bottom right – waveforms from laser trials (red) and trials without laser (blue). Across 73 tagged neurons, waveform correlation coefficients for laser trials vs. trials without laser was r = 0.97 (0.92-0.99). These data demonstrate that optogenetically triggered spikes are similar to non-optogenetically triggered spikes.”
(10) Figure S7, what was the laser power used in this experiment? Have the authors tried different laser powers?
We have now clarified the laser power on line 598:
“In animals injected with optogenetic viruses, optical inhibition was delivered via bilateral patch cables for the entire trial duration of 18 seconds via 589-nm laser light at 12 mW power on 50% of randomly assigned trials.”
And for Figure S6 (was S7 previously):
We did not try other laser powers; our parameters were chosen a priori based on our past work.
(11) In Figure S9, what method was used to sort the neurons?
We now clarify in the methods (Line 617):
“Electrophysiology. Single-unit recordings were made using a multi-electrode recording system (Open Ephys, Atlanta, GA). After the experiments, Plexon Offline Sorter (Plexon, Dallas, TX), was used to remove artifacts. Principal component analysis (PCA) and waveform shape were used for spike sorting. Single units were defined as those 1) having a consistent waveform shape, 2) being a separable cluster in PCA space, and 3) having a consistent refractory period of at least 2 milliseconds in interspike interval histograms. The same MSNs were sorted across saline, D2 blockade, and D1 blockade sessions by loading all sessions simultaneously in Offline Sorter and sorted using the preceding criteria. MSNs had to have consistent firing in all sessions to be included. Sorting integrity across sessions was quantified by comparing waveform similarity via R2 between sessions.”
And in the results (Line 353):
“We analyzed 99 MSNs in sessions with saline, D2 blockade, and D1 blockade. We matched MSNs across sessions based on waveform and interspike intervals; waveforms were highly similar across sessions (correlation coefficient between matched MSN waveforms: saline vs D2 blockade r = 1.00 (0.99 – 1.00 rank sum vs correlations in unmatched waveforms p = 3x10-44; waveforms; saline vs D1 blockade r = 1.00 (1.00 – 1.00), rank sum vs correlations in unmatched waveforms p = 4x10-50). There were no consistent changes in MSN average firing rate with D2 blockade or D1 blockade (F = 1.1, p = 0.30 accounting for variance between MSNs; saline: 5.2 (3.3 – 8.6) Hz; D2 blockade 5.1 (2.7 – 8.0) Hz; F = 2.2, p = 0.14; D1 blockade 4.9 (2.4 – 7.8) Hz).”
(C-F) statistics should be done based on the number of mice, not on the number of recorded neurons.
We agree, all experiments are now quantified using linear mixed effects models which formally accounts for variance contributed across animals, as discussed at length earlier in the review and with statistical experts at the University of Iowa.
(12) For results in Figure 6, did the authors do cell-type specific recording on D1 or D2 MSNs using optogenetic tagging? As the D1- or D2- MSNs account for ~50% of all MSNs, the inhibition of a considerable amount of neurons was not observed. The authors should discuss the relation between the results from optogenetic inhibition of D1- or D2- MSNs and pharmacological disruption of D1 or D2 dopamine receptors.
This is a great point. First, we did not combine cell-type specific recordings with tagging as it was difficult to get enough trials for analysis in a single session in the tagging experiments, and pharmacological interventions can further decrease performance. However, we have made our results in Figure 6 much more focused.
We have discussed the relationship between these data in the results (Line 380):
“This data-driven analysis shows that D2 and D1 blockade produced similar shifts in MSN population dynamics represented by PC1. When combined with major contributions of D1/D2 MSNs to PC1 (Fig 3C) these findings show that pharmacologically disrupting D2 or D1 MSNs can disrupt ramping-related activity in the striatum.”
And in the discussion (Line 417):
“Strikingly, optogenetic tagging showed that D2-MSNs and D1-MSNs had distinct dynamics during interval timing. MSN dynamics helped construct and constrain a four-parameter drift-diffusion model in which D2- and D1-MSN spiking accumulated temporal evidence. This model predicted that disrupting either D2MSNs or D1-MSNs would increase response times. Accordingly, we found that optogenetically or pharmacologically disrupting striatal D2-MSNs or D1-MSNs increased response times without affecting task-specific movements. Disrupting D2MSNs or D1-MSNs shifted MSN temporal dynamics and degraded MSN temporal encoding. These data, when combined with our model predictions, demonstrate that D2-MSNs and D1-MSNs contribute temporal evidence to controlling actions in time.”
And:
“D2-MSNs and D1-MSNs play complementary roles in movement. For instance, stimulating D1-MSNs facilitates movement, whereas stimulating D2-MSNs impairs movement (Kravitz et al., 2010). Both populations have been shown to have complementary patterns of activity during movements (Tecuapetla et al., 2016), with MSNs firing at different phases of action initiation and selection. Further dissection of action selection programs reveals that opposing patterns of activation among D2MSNs and D1-MSNs suppress and guide actions, respectively, in the dorsolateral striatum (Cruz et al., 2022). A particular advantage of interval timing is that it captures a cognitive behavior within a single dimension — time. When projected along the temporal dimension, it was surprising that D2-MSNs and D1-MSNs had opposing patterns of activity. Past pharmacological work from our group and others have shown that disrupting D2 or D1 MSNs slows timing (De Corte et al., 2019; Drew et al., 2007, 2003; Stutt et al., 2023), in line with pharmacological and optogenetic results in this manuscript. Computational modeling predicted that disrupting either D2-MSNs or D1-MSNs increased self-reported estimates of time, which was supported by both optogenetic and pharmacological experiments. Notably, these disruptions are distinct from increased timing variability reported with administrations of amphetamine, ventral tegmental area dopamine neuron lesions, and rodent models of neurodegenerative disease (Balci et al., 2008; Gür et al., 2020, 2019; Larson et al., 2022; Weber et al., 2023). Furthermore, our current data demonstrate that disrupting either D2-MSN or D1-MSN activity shifted MSN dynamics and degraded temporal encoding, supporting prior work (De Corte et al., 2019; Drew et al., 2007, 2003; Stutt et al., 2023). Our recording experiments do not identify where a possible response threshold T is instantiated, but downstream basal ganglia structures may have a key role in setting response thresholds (Toda et al., 2017).”
(13) For Figure 2, what is the error region for G and H? Is there a statistically significant difference between the start (e.g., 0-1 s) and the end (e.g., 5-6 s) time?
G and H are standard error, which we have now clarified.
And on Line 166:
“These differences resulted in distinct activity early in the interval (0-1 seconds; F = 6.0, p = 0.02 accounting for variance between mice), but not late in the interval (5-6 seconds; F = 1.9, p = 0.17 accounting for variance between mice) between D2-MSNs and D1-MSNs.”
Minor:
(1) Figure 2 legend showed the wrong label "Peri-event raster C) from a D2-MSN (red) and E) from a D1-MSN (blue). It should be (D).
Fixed, thank you.
(2) Figure 2. Missing legend for (E) and (F).
Fixed, thank you.
(3) Line 423: mistyped "\"
Fixed, thank you.
Reviewer #3 (Recommendations For The Authors):
- To clarify that complementary means opposing in this context, I suggest changing the title.
This is a helpful suggestion. We have changed it exactly as the reviewer suggested:
“Complementary opposing D2-MSNs and D1-MSNs dynamics during interval timing”
- I recommend adding a supplementary figure to demonstrate all the nose pokes in all trials in a given session. The current figures make it hard to assess the specifics of the behavior. For example, what happens if, in a long-interval trial, the mouse pokes in the second nose poke before 6 seconds? Is that behavior punished? Do they keep alternating between the nose poke or do they stick to one nose poke?
We agree. We think this is a main point, and we have now redesigned Figure 1 to describe these details:
And added these details to the methods (Line 548):
“Interval timing switch task. We used a mouse-optimized operant interval timing task described in detail previously (Balci et al., 2008; Bruce et al., 2021; Tosun et al., 2016; Weber et al., 2023). Briefly, mice were trained in sound-attenuating operant chambers, with two front nosepokes flanking either side of a food hopper on the front wall, and a third nosepoke located at the center of the back wall. The chamber was positioned below an 8-kHz, 72-dB speaker (Fig 1A; MedAssociates, St. Albans, VT). Mice were 85% food restricted and motivated with 20 mg sucrose pellets (BioServ, Flemington, NJ). Mice were initially trained to receive rewards during fixed ratio nosepoke response trials. Nosepoke entry and exit were captured by infrared beams. After shaping, mice were trained in the “switch” interval timing task. Mice self-initiated trials at the back nosepoke, after which tone and nosepoke lights were illuminated simultaneously. Cues were identical on all trial types and lasted the entire duration of the trial (6 or 18 seconds). On 50% of trials, mice were rewarded for a nosepoke after 6 seconds at the designated first ‘front’ nosepoke; these trials were not analyzed. On the remaining 50% of trials, mice were rewarded for nosepoking first at the ‘first’ nosepoke location and then switching to the ‘second’ nosepoke location; the reward was delivered for initial nosepokes at the second nosepoke location after 18 seconds when preceded by a nosepoke at the first nosepoke location. Multiple nosepokes at each nosepokes were allowed. Early responses at the first or second nosepoke were not reinforced. Initial responses at the second nosepoke rather than the first nosepoke, alternating between nosepokes, going back to the first nosepoke after the second nosepoke were rare after initial training. Error trials included trials where animals responded only at the first or second nosepoke and were also not reinforced. We did not analyze error trials as they were often too few to analyze; these were analyzed at length in our prior work (Bruce et al., 2021).”
- Figures 2E and 2F suggest that some D1 cells ramp up during the first 6 seconds, while others ramp down. The same is more or less true for D2s. I wonder if the analysis will lose its significance if the two outlier D1s are excluded from Figure 3D.
This is a great idea suggested by multiple reviewers. We repeated this analysis with outliers removed. We used a data-driven approach to remove outliers (Line 656):
“We performed additional sensitivity analysis excluding outliers outside of 95% confidence intervals and measuring firing rate from the start of the interval to the time of the switch response on a trial-by-trial level for each neuron.”
And described these data in the results (Line 201):
“We found that D2-MSNs and D1-MSNs had a significantly different slope even when excluding outliers (4 outliers excluded outside of 95% confidence intervals; F=7.51, p=0.008 accounting for variance between mice) and when the interval was defined as the time between trial start and the switch response on a trial-by-trial basis for each neuron (F=4.3, p=0.04 accounting for variance between mice).”
Finally, we removed the outliers the reviewers alluded to – two D1 MSNs – and found similar results (F=6.59, p=0.01 for main effect of D2 vs. D1 MSNs controlling for between-mouse variability). We elected to include the more data driven approach based on 95% confidence intervals.
-

-

Author Response
eLife assessment
This valuable study examines the activity and function of dorsomedial striatal neurons in estimating time. The authors examine striatal activity as a function of time and the impact of optogenetic striatal manipulation on the animal's ability to estimate a time interval. However, the task's design and methodology present several confounding factors that mean the evidence in support of the authors' claims is incomplete. With these limitations addressed, the work would be of interest to neuroscientists examining how striatum contributes to behavior.
We appreciate the editorial process and are grateful for the thorough, detailed, and constructive reviews. We will respond in detail to every point raised by reviewers in a full revision.
Public Reviews:
Reviewer #1 (Public Review):
Summary:
In this …
Author Response
eLife assessment
This valuable study examines the activity and function of dorsomedial striatal neurons in estimating time. The authors examine striatal activity as a function of time and the impact of optogenetic striatal manipulation on the animal's ability to estimate a time interval. However, the task's design and methodology present several confounding factors that mean the evidence in support of the authors' claims is incomplete. With these limitations addressed, the work would be of interest to neuroscientists examining how striatum contributes to behavior.
We appreciate the editorial process and are grateful for the thorough, detailed, and constructive reviews. We will respond in detail to every point raised by reviewers in a full revision.
Public Reviews:
Reviewer #1 (Public Review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nose poke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Critically, this task thus requires animals to estimate if at least 6 seconds have passed after the first nose poke - this is the key aspect of the task focused on here. After verifying that animals reliably estimate the passage of 6 seconds by leaving on average after 9 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2-MSNs increase activity, throughout this interval. They suggest that this activity follows a drift-diffusion model, in which activity increases (or decreases) to a threshold after which a decision (to leave) is made. The authors next report that optogenetically inhibiting D1 or D2 MSNs, or pharmacologically blocking D1 and D2 receptors, increased the average wait time of the animals to 10 seconds on average. This suggests that both D1 and D2 neurons contribute to the estimate of time, with a decrease in their activity corresponding to a decrease in the rate of 'drift' in their drift-diffusion model. Lastly, the authors examine MSN activity while pharmacologically inhibiting D1 or D2 receptors. The authors observe most recorded MSNs neurons decrease their activity over the interval, with the rate decreasing with D1/D2 receptor inhibition.
Major strengths:
The study employs a wide range of techniques - including animal behavioral training, electrophysiology, optogenetic manipulation, pharmacological manipulations, and computational modeling. The behavioral task used by the authors is quite interesting and a nice way to probe interval timing in rodents. The question posed by the authors - how striatal activity contributes to interval timing - is of importance to the field and has been the focus of many studies and labs; thus, this paper can meaningfully contribute to that conversation. The data within the paper is presented very clearly, and the authors have done a nice job presenting the data in a transparent manner (e.g., showing individual cells and animals). Overall, the manuscript is relatively easy to read and clear, with sufficient detail given in most places regarding the experimental paradigm or analyses used.
We are glad our main points came through to the reviewer.
Major weaknesses:
I perceive two major weaknesses. The first is the impact or contextualization of their results in terms of the results of the field more broadly. More specifically, it was not clear to me how the authors are interpreting the striatal activity in the context of what others have observed during interval timing tasks. In other words - what was the hypothesis going into this experiment? Does observing increasing/decreasing activity in D2 versus D1 support one model of interval timing over another, or does it further support a more specific idea of how DMS contributes to interval timing? Or was the main question that we didn't know if D2 or D1 neurons had differential activity during interval timing?
Our hypothesis, based on prior behavioral work from our group describing that blocking striatal D1 and D2 dopamine receptors impaired interval timing (De Corte et al., 2019; Stutt et al., 2023) was D1 and D2 MSNs would have similar patterns of activity during interval timing. We will clarify this in the revision.
In the second, I felt that some of the conclusions suggested by the authors don't seem entirely supported by the data they present, or the data presented suggests a slightly more complicated story. Below I provide additional detail on some of these instances.
Regarding the results presented in Figures 2 and 3:
I am not sure the PC analysis adds much to the interpretation, and potentially unnecessarily complicates things. In particular, running PCA on a matrix of noisy data that is smoothed with a Gaussian will often return PCs similar to what is observed by the authors, with the first PC being a line up/down, the 2nd PC being a parabola that is up/down, etc. Thus, I'm not sure that there is much to be interpreted by the specific shape of the PCs here.
These are insightful points. We will clarify details of our PCA analysis in the revision. We include PCA for comparisons with our past work (Emmons et al., 2017, 2021; Bruce et al., 2021). Second, it is true that these components can be observed in smoothed data; however, when we generated random data using identical parameters, we found that the variance explained by PC1 was not commonly observed in random data. Third, our goal is to compare between D1 and D2 MSNs, not to interpret the PCs. We will make this explicit in our revision.
I think an alternative analysis that might be both easier and more informative is to compute the slope of the activity of each neuron across the 6 seconds. This would allow the authors to quantify how many neurons increase or decrease their activity much like what is shown in Figure 2.
This is exactly the analysis shown in Figure 3D. We will clarify this in the revision.
Relatedly, it seems that the data shown in Figure 2D doesn't support the authors' main claim regarding D2/D1 MSNs increasing/decreasing their activity, as the trial-by-trial slope is near 0 for both cell types.
This likely refers to Figure 3D. In the revision, we will clarify this analysis, add error bars, and note that our goal was to differentiate D2 and D1 MSNs in this analysis. We will also add to this analysis to better make the poin that D2 and D1 MSNs are distinct, contrary to our hypothesis.
Regarding the results in Figure 4:
The authors suggest that their data is consistent with a drift-diffusion model. However, it is unclear how well the output from the model fits the activity from neurons the authors recorded. Relatedly, it is unclear how the parameters were chosen for the D1/D2 versions of this model. I think that an alternate approach that would answer these questions is to fit the model to each cell, and then examine the best-fit parameters, as well as the ability of the model to predict activity on trials held out from the fitting process. This would provide a more rigorous method to identify the best parameters and would directly quantify how well the model captures the data.
This is a great point. Our goal was to fit behavioral activity, not neuronal activity; in our revision, we will do exactly what the reviewer suggests and present data of fits to neuronal activity.
Relatedly, looking at the raw data in Figure 2, it seems that many neurons either fire at the beginning or end of the interval, with more neurons firing at the end, and more firing at the beginning, for D2/D1 neurons respectively. Thus, it's not clear to me whether the drift-diffusion model is a good model of activity. Or, perhaps the model is supposed to be related to the aggregate activity of all D1/D2 neurons? (If so, this should be made more explicit. The comment about fitting the model directly to the data also still stands).
Our model was inspired by the averages in Figure 2G&H; however, we will fit drift-diffusion models to individual neurons exactly as the reviewer suggests.
Further, it's unclear to me how, or why, the authors changed the specific parameters they used to model the optogenetic manipulation. Were these parameters chosen because they fit the manipulation data? This I don't think is in itself an issue, but perhaps should be clearly stated, because otherwise it sounds a bit odd given the parameter changes are so specific. It is also not clear to me why the noise in the diffusion process would be expected to change with increased inhibition.
We will clarify this in our revision, as this is an important point.
Regarding the results in Figure 6:
My comments regarding the interpretation of PCs in Figure 2 apply here as well. In addition, I am not sure that examining PC2 adds much here, given that the authors didn't examine such nonlinear changes earlier in the paper.
We agree – we will remove PC2 in Figure 6 and Figure S9 and add context to the PC analysis noting that we are including for 1) comparisons with past work, 2) our observed variance is much higher than observed in random/smoothed data, and 3) we are primarily interested in comparisons between conditions rather than interpreting the components.
A larger concern though that seems potentially at odds with the authors' interpretation is that there seems to be very little change in the firing pattern after D1 or D2 blockade. I see that in Figure 6F the authors suggest that many cells slope down (and thus, presumably, they are recoding more D1 cells), and that this change in slope is decreased, but this effect is not apparent in Figure 6C, and Figure 6B shows an example of a cell that seems to fire in the opposite direction (increase activity). I think it would help to show some (more) individual examples that demonstrate the summary effect shown by the authors, and perhaps the authors can comment on the robustness (or the variability) of this result.
We agree, although we note D1/D2 blockade changes PC1, which explains the most variance in MSN activity. In the revision, we will show more examples and comment on the robustness of PC1, exactly as the reviewer recommends. The changes in PC1 are rather consistent.
Also, it seems that if the authors want to claim that this manipulation lowers the drift rate. I think to make this claim, they could fit the DDM model and examine whether D is significantly lower.
This is a great idea – we will try to do this.
Regarding the results in Figure 7:
I am overall a bit confused about what the authors are trying to claim here. In Figure 7, they present data suggesting that D1 or D2 blockade disrupts their ability to decode time in the interval of interest (0-6 seconds). However, in the final paragraph of the results, the authors seem to say that by using another technique, they didn't see any significant change in decoding accuracy after D1 or D2 blockade. What do the authors make of this?
We were not clear. The second classifier was predicting response time. This was confusing and we will remove it.
Impact:
The task and data presented by the authors are very intriguing, and there are many groups interested in how striatal activity contributes to the neural perception of time. The authors perform a wide variety of experiments and analysis to examine how DMS activity influences time perception during an interval-timing task, allowing for insight into this process. However, the significance of the key finding - that D2/D1 activity increases/ decreases with time - remains somewhat ambiguous to me. This arises from a lack of clarity regarding the initial hypothesis and the implications of this finding for advancing our understanding of striatal functions.
Again, we are grateful for the constructive and very insightful comments that we look forward to clarifying in a full revision.
Reviewer #2 (Public Review):
Summary:
In the present study, the authors investigated the neural coding mechanisms for D1- and D2-expressing striatal direct and indirect pathway MSNs in interval timing by using multiple strategies. They concluded that D2-MSNs and D1-MSNs have opposing temporal dynamics yet disrupting either type produced similar effects on behavior, indicating the complementary roles of D1- and D2- MSNs in cognitive processing. However, the data was incomplete to fully support this major finding. One major reason is the heterogenetic responses within the D1-or D2-MSN populations. In addition, there are additional concerns about the statistical methods used. For example, the majority of the statistical tests are based on the number of neurons, but not the number of mice. It appears that the statistical difference was due to the large sample size they used (n=32 D2-MSNs and n=41 D1-MSNs), but different neurons recorded in the same mouse cannot be treated as independent samples; they should use independent mouse-based statistical analysis.
Strengths:
The authors used multiple approaches including awake mice behavior training, optogenetic-assistant cell-type specific recording, optogenetic or pharmacological manipulation, neural computation, and modeling to study neuronal coding for interval timing.
We appreciate the reviewer’s careful read recognizing the breadth of our approach.
Weaknesses:
(1) More detailed behavior results should be shown, including the rate of the success switches, and how long it takes to wait in the second nose poke to get a reward. For line 512 and the Figure 1 legend, the reviewer is not clear about the reward delivery. The methods appear to state that the mouse had to wait for 18s, then make nose pokes at the second port to get the reward. What happens if the mouse made the second nose poke before 18 seconds, but then exited? Would the mouse still get the reward at 18 seconds? Similarly, what happens if the mice made the third or more nosepokes within 18 seconds? It is important to clarify because, according to the method described, if the mice made a second nose poke before 18 seconds, this already counted as the mouse making the "switch." Lastly, what if the mice exited before 6s in the first nosepoke?
We agree. These were presented in detail in our prior work (Bruce et al., 2021; Larson et al., 2022; and Weber et al., 2023) and work from others (Balci et al 2008; Tosun et al., 2016. However, we will work on a detailed behavioral schematic in the revision and move supplementary behavioral data in Figure S1 to the main manuscript.
(2) There are a lot of time parameters in this behavior task, the description of those time parameters is mentioned in several parts, in the figure legend, supplementary figure legend, and methods, but was not defined clearly in the main text. It is inconvenient, sometimes, confusing for the readers. The authors should make a schematic diagram to illustrate the major parameters and describe them clearly in the main text.
This is a great suggestion – we will do this – and clarify in the above schematic.
(3) In Line 508, the reviewer suggests the authors pay attention to those trials without "switch". It would be valuable to compare the MSN activity between those trials with or without a "switch".
We analyzed MSN activity on errors in detail Figure 6 of Bruce et al., 2021. These errors are infrequent and inconsistent – we will discuss this in the revision.
(4) The definition of interval is not very clear. It appears that the authors used a 6-second interval in analyzing the data in Figure 2 and Figure 3. But from my understanding, the interval should be the time from time "0" to the "switch", when the mice start to exit from the first nose poke.
We agree. The switch time can be vastly different on some trials, making it challenging to compare different lengths and slopes. However, we will clarify the interval as noted above, and we have a few ideas on how to do the analysis the reviewer suggests.
(5) For Figure 2 C-F, the authors only recorded 32 D2-MSNs in 4 mice, and 41 D1-MSNs in 5 mice. The sample size is too small compared to the sample size usually used in the field. In addition to the small sample size, the single-cell activity exhibited heterogeneity, which created potential issues. For both D1 and D2 MSNs, the authors tried to make conclusions on the "trend" of increasing in D2-MSNs and decreasing in D1-MSNs populations, respectively, during the interval. However, such a conclusion is not sufficiently supported by the data presented. It looks like the single-cell activity patterns can be separated into groups: one is a decreasing activity group, one is an increasing activity group and a small group for on and off response. Because of the small sample size, the author should pay attention to the variance across different mice (which needs to be clearly presented in the manuscript), instead of pooling data together and analyzing the mean activity.
We were not clear – we did this analysis exactly the reviewer suggested. We are not pooling any data – instead – as we state on line 620 – we are using linear-mixed effects models to account for mouse-specific and neuron-specific variance. This approach was developed with our statistics core for exactly the reasons the reviewer suggested. Furthermore, we will add to this analysis demonstrative that it is resistant to outliers. Finally, we will include measures of effect size noting that it is a medium to large effect.
It’s a helpful idea to plot data individually by mice, and we will do so in the revision.
(6) For Figure 2, from the activity in E and F, it seems that the activity already rose before the trial started, the authors should add some longer baseline data before time zero for clarification and comparison, and show the timing of the actual start of the activity with the corresponding behavior. What behavior states are the mice in when initiating the activity?
We can certainly include a longer baseline. We can clarify in the revision that mice initiate trials at the rear nosepoke, and this is what initiates the task cues and the temporal interval.
(7) The authors were focused on the "switch " behavior in the task, but they used an arbitrary 6s time window to analyze the activity, and tried to correlate the decreasing or increasing activities of MSNs to the neural coding for time. A better way to analyze is to sort the activity according to the "switch" time, from short to long intervals. This way, the authors could see and analyze whether the activity of D1 or D2 MSNs really codes for the different length of interval, instead of finding a correlation between average activity trends and the arbitrary 6s time window.
This is a great idea, and we have some ideas on how to adapt the GLM analysis to perform this analysis.
Reviewer #3 (Public Review):
Summary:
The cognitive striatum, also known as the dorsomedial striatum, receives input from brain regions involved in high-level cognition and plays a crucial role in processing cognitive information. However, despite its importance, the extent to which different projection pathways of the striatum contribute to this information processing remains unclear. In this paper, Bruce et al. conducted a study using a range of causal and correlational techniques to investigate how these pathways collectively contribute to interval timing in mice. Their results were consistent with previous research, showing that the direct and indirect striatal pathways perform opposing roles in processing elapsed time. Based on their findings, the authors proposed a revised computational model in which two separate accumulators track evidence for elapsed time in opposing directions. These results have significant implications for understanding the neural mechanisms underlying cognitive impairment in neurological and psychiatric disorders, as disruptions in the balance between direct and indirect pathway activity are commonly observed in such conditions.
Strengths:
The authors employed a well-established approach to study interval timing and employed optogenetic tagging to observe the behavior of specific cell types in the striatum. Additionally, the authors utilized two complementary techniques to assess the impact of manipulating the activity of these pathways on behavior. Finally, the authors utilized their experimental findings to enhance the theoretical comprehension of interval timing using a computational model.
We are grateful for the reviewer’s consideration of our work and recognizing the strengths of our approach.
Weaknesses:
The behavioral task used in this study is best suited for investigating elapsed time perception, rather than interval timing. Timing bisection tasks are often employed to study interval timing in humans and animals.
This is certainly valid, and we will include these points in the revision.
The main results from unit recording (opposing slopes of D1/D2 cell firing rate, as shown in Figure 3D) appear to be very sensitive to a couple of outlier cells, and the predictive power of ensemble recording seems to be only slightly above chance levels.
We are glad that the reviewer raised this. We will add to this analysis demonstrative that it is resistant to outliers. Finally, we will include measures of effect size noting that it is a medium to large effect. Thus, it is significantly above chance, and rather reliable, and supported by our PCA results in Figure 3C.
In the optogenetic experiment, the laser was kept on for too long (18 seconds) at high power (12 mW). This has been shown to cause adverse effects on population activity (for example, through heating the tissue) that are not necessarily related to their function during the task epochs.
Again, this is an important point. We are well aware of heating effects with optogenetics. For the exact reasons noted by the reviewer, we had opsin-negative controls –when the laser was on the exact same time course and parameters – in Figure S5. There were no behavioral effects in controls with identical heating and other effects of the laser. Furthermore, these effects are similar to pharmacological effects in this manuscript and in our prior work (De Corte et al., 2019; Stutt et al., 2023). We will better highlight these issues in the revision.
Given the systemic delivery of pharmacological interventions, it is difficult to conclude that the effects are specific to the dorsomedial striatum. Future studies should use the local infusion of drugs into the dorsomedial striatum.
This is a great point - we did exactly this experiment in De Corte et al, 2019 with local drug infusions. This earlier study was the departure point for this experiment, although it is challenging to combine focal pharmacological inactivation with recordings in mice (we have extensive experience with this in rats in Parker et al., 2015 and Parker et al, 2015). Furthermore, we have similar local optogenetics effects in this paper. We will include these points in the revised manuscript.
-

eLife assessment
This valuable study examines the activity and function of dorsomedial striatal neurons in estimating time. The authors examine striatal activity as a function of time and the impact of optogenetic striatal manipulation on the animal's ability to estimate a time interval. However, the task's design and methodology present several confounding factors that mean the evidence in support of the authors' claims is incomplete. With these limitations addressed, the work would be of interest to neuroscientists examining how striatum contributes to behavior.
-

Reviewer #1 (Public Review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nose poke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Critically, this task thus requires animals to estimate if at least 6 seconds have passed after the first nose poke - this is the key aspect of the task focused on here. After verifying that animals reliably estimate the passage of 6 seconds by leaving on average after 9 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2-MSNs increase activity, throughout this interval. They suggest that this activity follows a drift-diffusion model, in …
Reviewer #1 (Public Review):
Summary:
In this work, the authors examine the activity and function of D1 and D2 MSNs in dorsomedial striatum (DMS) during an interval timing task. In this task, animals must first nose poke into a cued port on the left or right; if not rewarded after 6 seconds, they must switch to the other port. Critically, this task thus requires animals to estimate if at least 6 seconds have passed after the first nose poke - this is the key aspect of the task focused on here. After verifying that animals reliably estimate the passage of 6 seconds by leaving on average after 9 seconds, the authors examine striatal activity during this interval. They report that D1-MSNs tend to decrease activity, while D2-MSNs increase activity, throughout this interval. They suggest that this activity follows a drift-diffusion model, in which activity increases (or decreases) to a threshold after which a decision (to leave) is made. The authors next report that optogenetically inhibiting D1 or D2 MSNs, or pharmacologically blocking D1 and D2 receptors, increased the average wait time of the animals to 10 seconds on average. This suggests that both D1 and D2 neurons contribute to the estimate of time, with a decrease in their activity corresponding to a decrease in the rate of 'drift' in their drift-diffusion model. Lastly, the authors examine MSN activity while pharmacologically inhibiting D1 or D2 receptors. The authors observe most recorded MSNs neurons decrease their activity over the interval, with the rate decreasing with D1/D2 receptor inhibition.
Major strengths:
The study employs a wide range of techniques - including animal behavioral training, electrophysiology, optogenetic manipulation, pharmacological manipulations, and computational modeling. The behavioral task used by the authors is quite interesting and a nice way to probe interval timing in rodents. The question posed by the authors - how striatal activity contributes to interval timing - is of importance to the field and has been the focus of many studies and labs; thus, this paper can meaningfully contribute to that conversation. The data within the paper is presented very clearly, and the authors have done a nice job presenting the data in a transparent manner (e.g., showing individual cells and animals). Overall, the manuscript is relatively easy to read and clear, with sufficient detail given in most places regarding the experimental paradigm or analyses used.
Major weaknesses:
I perceive two major weaknesses. The first is the impact or contextualization of their results in terms of the results of the field more broadly. More specifically, it was not clear to me how the authors are interpreting the striatal activity in the context of what others have observed during interval timing tasks. In other words - what was the hypothesis going into this experiment? Does observing increasing/decreasing activity in D2 versus D1 support one model of interval timing over another, or does it further support a more specific idea of how DMS contributes to interval timing? Or was the main question that we didn't know if D2 or D1 neurons had differential activity during interval timing?
In the second, I felt that some of the conclusions suggested by the authors don't seem entirely supported by the data they present, or the data presented suggests a slightly more complicated story. Below I provide additional detail on some of these instances.
Regarding the results presented in Figures 2 and 3:
I am not sure the PC analysis adds much to the interpretation, and potentially unnecessarily complicates things. In particular, running PCA on a matrix of noisy data that is smoothed with a Gaussian will often return PCs similar to what is observed by the authors, with the first PC being a line up/down, the 2nd PC being a parabola that is up/down, etc. Thus, I'm not sure that there is much to be interpreted by the specific shape of the PCs here. I think an alternative analysis that might be both easier and more informative is to compute the slope of the activity of each neuron across the 6 seconds. This would allow the authors to quantify how many neurons increase or decrease their activity much like what is shown in Figure 2.
Relatedly, it seems that the data shown in Figure 2D *doesn't* support the authors' main claim regarding D2/D1 MSNs increasing/decreasing their activity, as the trial-by-trial slope is near 0 for both cell types.
Regarding the results in Figure 4:
The authors suggest that their data is consistent with a drift-diffusion model. However, it is unclear how well the output from the model fits the activity from neurons the authors recorded. Relatedly, it is unclear how the parameters were chosen for the D1/D2 versions of this model. I think that an alternate approach that would answer these questions is to fit the model to each cell, and then examine the best-fit parameters, as well as the ability of the model to predict activity on trials held out from the fitting process. This would provide a more rigorous method to identify the best parameters and would directly quantify how well the model captures the data.
Relatedly, looking at the raw data in Figure 2, it seems that many neurons either fire at the beginning or end of the interval, with more neurons firing at the end, and more firing at the beginning, for D2/D1 neurons respectively. Thus, it's not clear to me whether the drift-diffusion model is a good model of activity. Or, perhaps the model is supposed to be related to the aggregate activity of all D1/D2 neurons? (If so, this should be made more explicit. The comment about fitting the model directly to the data also still stands).
Further, it's unclear to me how, or why, the authors changed the specific parameters they used to model the optogenetic manipulation. Were these parameters chosen because they fit the manipulation data? This I don't think is in itself an issue, but perhaps should be clearly stated, because otherwise it sounds a bit odd given the parameter changes are so specific. It is also not clear to me why the noise in the diffusion process would be expected to change with increased inhibition.
Regarding the results in Figure 6:
My comments regarding the interpretation of PCs in Figure 2 apply here as well. In addition, I am not sure that examining PC2 adds much here, given that the authors didn't examine such nonlinear changes earlier in the paper.
A larger concern though that seems potentially at odds with the authors' interpretation is that there seems to be very little change in the firing pattern after D1 or D2 blockade. I see that in Figure 6F the authors suggest that many cells slope down (and thus, presumably, they are recoding more D1 cells), and that this change in slope is decreased, but this effect is not apparent in Figure 6C, and Figure 6B shows an example of a cell that seems to fire in the opposite direction (increase activity). I think it would help to show some (more) individual examples that demonstrate the summary effect shown by the authors, and perhaps the authors can comment on the robustness (or the variability) of this result.
Also, it seems that if the authors want to claim that this manipulation lowers the drift rate. I think to make this claim, they could fit the DDM model and examine whether D is significantly lower.
Regarding the results in Figure 7:
I am overall a bit confused about what the authors are trying to claim here. In Figure 7, they present data suggesting that D1 or D2 blockade disrupts their ability to decode time in the interval of interest (0-6 seconds). However, in the final paragraph of the results, the authors seem to say that by using another technique, they didn't see any significant change in decoding accuracy after D1 or D2 blockade. What do the authors make of this?
Impact:
The task and data presented by the authors are very intriguing, and there are many groups interested in how striatal activity contributes to the neural perception of time. The authors perform a wide variety of experiments and analysis to examine how DMS activity influences time perception during an interval-timing task, allowing for insight into this process. However, the significance of the key finding - that D2/D1 activity increases/ decreases with time - remains somewhat ambiguous to me. This arises from a lack of clarity regarding the initial hypothesis and the implications of this finding for advancing our understanding of striatal functions.
-

Reviewer #2 (Public Review):
Summary:
In the present study, the authors investigated the neural coding mechanisms for D1- and D2-expressing striatal direct and indirect pathway MSNs in interval timing by using multiple strategies. They concluded that D2-MSNs and D1-MSNs have opposing temporal dynamics yet disrupting either type produced similar effects on behavior, indicating the complementary roles of D1- and D2- MSNs in cognitive processing. However, the data was incomplete to fully support this major finding. One major reason is the heterogenetic responses within the D1-or D2-MSN populations. In addition, there are additional concerns about the statistical methods used. For example, the majority of the statistical tests are based on the number of neurons, but not the number of mice. It appears that the statistical difference was due …
Reviewer #2 (Public Review):
Summary:
In the present study, the authors investigated the neural coding mechanisms for D1- and D2-expressing striatal direct and indirect pathway MSNs in interval timing by using multiple strategies. They concluded that D2-MSNs and D1-MSNs have opposing temporal dynamics yet disrupting either type produced similar effects on behavior, indicating the complementary roles of D1- and D2- MSNs in cognitive processing. However, the data was incomplete to fully support this major finding. One major reason is the heterogenetic responses within the D1-or D2-MSN populations. In addition, there are additional concerns about the statistical methods used. For example, the majority of the statistical tests are based on the number of neurons, but not the number of mice. It appears that the statistical difference was due to the large sample size they used (n=32 D2-MSNs and n=41 D1-MSNs), but different neurons recorded in the same mouse cannot be treated as independent samples; they should use independent mouse-based statistical analysis.
Strengths:
The authors used multiple approaches including awake mice behavior training, optogenetic-assistant cell-type specific recording, optogenetic or pharmacological manipulation, neural computation, and modeling to study neuronal coding for interval timing.
Weaknesses:
(1) More detailed behavior results should be shown, including the rate of the success switches, and how long it takes to wait in the second nose poke to get a reward. For line 512 and the Figure 1 legend, the reviewer is not clear about the reward delivery. The methods appear to state that the mouse had to wait for 18s, then make nose pokes at the second port to get the reward. What happens if the mouse made the second nose poke before 18 seconds, but then exited? Would the mouse still get the reward at 18 seconds? Similarly, what happens if the mice made the third or more nosepokes within 18 seconds? It is important to clarify because, according to the method described, if the mice made a second nose poke before 18 seconds, this already counted as the mouse making the "switch." Lastly, what if the mice exited before 6s in the first nosepoke?
(2) There are a lot of time parameters in this behavior task, the description of those time parameters is mentioned in several parts, in the figure legend, supplementary figure legend, and methods, but was not defined clearly in the main text. It is inconvenient, sometimes, confusing for the readers. The authors should make a schematic diagram to illustrate the major parameters and describe them clearly in the main text.
(3) In Line 508, the reviewer suggests the authors pay attention to those trials without "switch". It would be valuable to compare the MSN activity between those trials with or without a "switch".
(4) The definition of interval is not very clear. It appears that the authors used a 6-second interval in analyzing the data in Figure 2 and Figure 3. But from my understanding, the interval should be the time from time "0" to the "switch", when the mice start to exit from the first nose poke.
(5) For Figure 2 C-F, the authors only recorded 32 D2-MSNs in 4 mice, and 41 D1-MSNs in 5 mice. The sample size is too small compared to the sample size usually used in the field. In addition to the small sample size, the single-cell activity exhibited heterogeneity, which created potential issues. For both D1 and D2 MSNs, the authors tried to make conclusions on the "trend" of increasing in D2-MSNs and decreasing in D1-MSNs populations, respectively, during the interval. However, such a conclusion is not sufficiently supported by the data presented. It looks like the single-cell activity patterns can be separated into groups: one is a decreasing activity group, one is an increasing activity group and a small group for on and off response. Because of the small sample size, the author should pay attention to the variance across different mice (which needs to be clearly presented in the manuscript), instead of pooling data together and analyzing the mean activity.
(6) For Figure 2, from the activity in E and F, it seems that the activity already rose before the trial started, the authors should add some longer baseline data before time zero for clarification and comparison, and show the timing of the actual start of the activity with the corresponding behavior. What behavior states are the mice in when initiating the activity?
(7) The authors were focused on the "switch " behavior in the task, but they used an arbitrary 6s time window to analyze the activity, and tried to correlate the decreasing or increasing activities of MSNs to the neural coding for time. A better way to analyze is to sort the activity according to the "switch" time, from short to long intervals. This way, the authors could see and analyze whether the activity of D1 or D2 MSNs really codes for the different length of interval, instead of finding a correlation between average activity trends and the arbitrary 6s time window.
-

Reviewer #3 (Public Review):
Summary:
The cognitive striatum, also known as the dorsomedial striatum, receives input from brain regions involved in high-level cognition and plays a crucial role in processing cognitive information. However, despite its importance, the extent to which different projection pathways of the striatum contribute to this information processing remains unclear. In this paper, Bruce et al. conducted a study using a range of causal and correlational techniques to investigate how these pathways collectively contribute to interval timing in mice. Their results were consistent with previous research, showing that the direct and indirect striatal pathways perform opposing roles in processing elapsed time. Based on their findings, the authors proposed a revised computational model in which two separate accumulators …
Reviewer #3 (Public Review):
Summary:
The cognitive striatum, also known as the dorsomedial striatum, receives input from brain regions involved in high-level cognition and plays a crucial role in processing cognitive information. However, despite its importance, the extent to which different projection pathways of the striatum contribute to this information processing remains unclear. In this paper, Bruce et al. conducted a study using a range of causal and correlational techniques to investigate how these pathways collectively contribute to interval timing in mice. Their results were consistent with previous research, showing that the direct and indirect striatal pathways perform opposing roles in processing elapsed time. Based on their findings, the authors proposed a revised computational model in which two separate accumulators track evidence for elapsed time in opposing directions. These results have significant implications for understanding the neural mechanisms underlying cognitive impairment in neurological and psychiatric disorders, as disruptions in the balance between direct and indirect pathway activity are commonly observed in such conditions.
Strengths:
The authors employed a well-established approach to study interval timing and employed optogenetic tagging to observe the behavior of specific cell types in the striatum. Additionally, the authors utilized two complementary techniques to assess the impact of manipulating the activity of these pathways on behavior. Finally, the authors utilized their experimental findings to enhance the theoretical comprehension of interval timing using a computational model.
Weaknesses:
The behavioral task used in this study is best suited for investigating elapsed time perception, rather than interval timing. Timing bisection tasks are often employed to study interval timing in humans and animals. The main results from unit recording (opposing slopes of D1/D2 cell firing rate, as shown in Figure 3D) appear to be very sensitive to a couple of outlier cells, and the predictive power of ensemble recording seems to be only slightly above chance levels. In the optogenetic experiment, the laser was kept on for too long (18 seconds) at high power (12 mW). This has been shown to cause adverse effects on population activity (for example, through heating the tissue) that are not necessarily related to their function during the task epochs. Given the systemic delivery of pharmacological interventions, it is difficult to conclude that the effects are specific to the dorsomedial striatum. Future studies should use the local infusion of drugs into the dorsomedial striatum.
-