Elevated glycolytic metabolism of monocytes limits the generation of HIF1A-driven migratory dendritic cells in tuberculosis
Curation statements for this article:-
Curated by eLife
eLife assessment
This useful study tests the hypothesis that monocytes purified from tuberculosis patients differentiate into dendritic cells with different migratory capacities. The authors conclude that these monocytes are metabolically pre-conditioned to differentiate, with reduced expression of Hif1a and a glycolytically exhaustive phenotype, resulting in low migratory and immunologic potential. Overall, the evidence provided is convincing, advancing the field substantively and providing novel insights.
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
During tuberculosis (TB), migration of dendritic cells (DCs) from the site of infection to the draining lymph nodes is known to be impaired, hindering the rapid development of protective T-cell-mediated immunity. However, the mechanisms involved in the delayed migration of DCs during TB are still poorly defined. Here, we found that infection of DCs with Mycobacterium tuberculosis (Mtb) triggers HIF1A-mediated aerobic glycolysis in a TLR2-dependent manner, and that this metabolic profile is essential for DC migration. In particular, the lactate dehydrogenase inhibitor oxamate and the HIF1A inhibitor PX-478 abrogated Mtb-induced DC migration in vitro to the lymphoid tissue-specific chemokine CCL21, and in vivo to lymph nodes in mice. Strikingly, we found that although monocytes from TB patients are inherently biased toward glycolysis metabolism, they differentiate into poorly glycolytic and poorly migratory DCs compared with healthy subjects. Taken together, these data suggest that because of their preexisting glycolytic state, circulating monocytes from TB patients are refractory to differentiation into migratory DCs, which may explain the delayed migration of these cells during the disease and opens avenues for host-directed therapies for TB.
Article activity feed
-

-

-

-

Author response:
The following is the authors’ response to the previous reviews.
eLife assessment
This useful study tests the hypothesis that Mycobacterium tuberculosis infection increases glycolysis in monocytes, which alters their capacity to migrate to lymph nodes as monocyte-derived dendritic cells. The authors conclude that infected monocytes are metabolically pre-conditioned to differentiate, with reduced expression of Hif1a and a glycolytically exhaustive phenotype, resulting in low migratory and immunologic potential. However, the evidence is incomplete as the use of live and dead mycobacteria still limits the ability to draw firm conclusions. The study will be of interest to microbiologists and infectious disease scientists.
In response to the general eLife assessment, we would like to emphasize that the study did not deal …
Author response:
The following is the authors’ response to the previous reviews.
eLife assessment
This useful study tests the hypothesis that Mycobacterium tuberculosis infection increases glycolysis in monocytes, which alters their capacity to migrate to lymph nodes as monocyte-derived dendritic cells. The authors conclude that infected monocytes are metabolically pre-conditioned to differentiate, with reduced expression of Hif1a and a glycolytically exhaustive phenotype, resulting in low migratory and immunologic potential. However, the evidence is incomplete as the use of live and dead mycobacteria still limits the ability to draw firm conclusions. The study will be of interest to microbiologists and infectious disease scientists.
In response to the general eLife assessment, we would like to emphasize that the study did not deal with “infected monocytes” per se but rather with monocytes purified from patients with active TB. We show that monocytes purified from these TB patients (versus healthy controls) differentiate into DCs with different migratory capacities. In addition, to address the reviewer's comments in this new version of our manuscript, we include a relevant characterization of the migration capacity of DCs infected with Mtb to the plethora of assays already shown with viable bacteria in the previous revised version of our manuscript.
All in all, we believe that our study has significantly improved thanks to the feedback provided by the editor and reviewer panel during the different revision processes. We sincerely hope that this version of our manuscript is deemed fit for publication in this prestigious journal.
Public Reviews:
Reviewer #3 (Public Review):
In the revised manuscript by Maio et al, the authors examined the bioenergetic mechanisms involved in the delayed migration of DC's during Mtb infection. The authors performed a series of in vitro infection experiments including bioenergetic experiments using the Agilent Seahorse XF, and glucose uptake and lactate production experiments. Also, data from SCENITH is included in the revised manuscript as well as some clinical data. This is a well written manuscript and addresses an important question in the TB field. A remaining weakness is the use of dead (irradiated) Mtb in several of the new experiments and claims where iMtb data were used to support live Mtb data. Another notable weakness lies in the author's insistence on asserting that lactate is the ultimate product of glycolysis, rather than acknowledging a large body of historical data in support of pyruvate's role in the process. This raises a perplexing issue highlighted by the authors: if Mtb indeed upregulates glycolysis, one would expect that inhibiting glycolysis would effectively control TB. However, the reality contradicts this expectation. Lastly, the examination of the bioenergetics of cells isolated from TB patients undergoing drug therapy, rather than studying them at their baseline state is a weakness.
We thank the reviewer for this insightful assessment and feedback of our study. With regards to the data obtained with iMtb to support that with live Mtb, we have clarified the use of either iMtb or Mtb for each figure legend in the new version of the manuscript. Furthermore, we included the confirmation of the involvement of TLR2 ligation in the up-regulation of HIF-1α triggered by viable Mtb (new Fig S2E). We also conducted migration assays using (live) Mtb-infected dendritic cells (DCs) treated with either oxamate or PX-478 to validate that the HIF1a/glycolysis axis is indeed essential for DC migration (new Fig 5D).
We respectfully acknowledge the reviewer's statement regarding the potential relationship between glycolysis and the control of TB. However, we find it necessary to elaborate on our stance, as our data offer a nuanced perspective. Our research indicates that DCs exhibit upregulated glycolysis following stimulation or infection by Mtb. This metabolic shift is crucial for facilitating cell migration to the draining lymph nodes, an essential step in mounting an effective immune response. Yet, it remains uncertain whether this glycolytic induction reaches a threshold conducive to generating a protective immune response, a matter that our findings do not definitively address. This aspect is carefully discussed in the manuscript, lines 380-385.
Moreover, analyses of samples from chronic TB patients suggest that the outcome of inhibiting glycolysis may vary depending on factors such as the infection stage, the targeted cell type (e.g., monocytes, DCs), and the affected compartment (systemic versus local). This variability aligns with the concept of "too much, too little" exemplified by the dual roles of IFNγ (PMID: 28646367) and TNFα (PMID: 19275693) in TB, emphasizing the need to maintain an inflammatory equilibrium. In the context of the HIF1α/glycolysis axis, it appears to be a matter of timing: a case of "too early" activation of glycolysis in precursors, which could upset the delicate balance necessary for an effective immune response. We have added these comments in the discussion (pages 19-20, lines 468-485).
In summary, while acknowledging the reviewer's perspective, we believe that a comprehensive understanding of the interplay between Mtb infection and glycolysis in myeloid cells requires further consideration of various contextual conditions, urging caution against oversimplified interpretations.
With regard to the patients' information, as pointed out by the reviewer, according to the inclusion criteria for patient samples in the approved protocol by the Institutional Ethics Committee, we recruit patients who have received less than 15 days of treatment (for sensitive TB, the total treatment duration is at least 6 months). We do not have access to patient sample before they begin the treatment, as starting therapy is the most urgent matter in this case. Following the reviewer's suggestion, we investigated whether the glycolytic activity of monocytes correlated with the initiation of antibiotic treatment within this 15-day period. Our observations did not show any significant impact during the initial 15 days of treatment (see expanded reply below). However, after 2 months of treatment, we found that the glycolytic profile of CD16+ monocytes returned to baseline levels as per our analysis. This suggests that despite the normalization of glycolytic activity with antibiotic therapy, heightened basal glycolysis remains noticeable during the initial two weeks of treatment (time limit to meet the inclusion criteria in our study cohort).
Recommendations for the authors:
Reviewer #3 (Recommendations For The Authors):
(1) In the revised manuscript, the authors addressed concerns related to using irradiated Mtb, a positive development. However, the study predominantly employs 1:1 or 2:1 MOI, representing a low infection model, with no observed statistical distinction between the two MOIs (Fig-1). To enhance the study, inclusion of a higher MOI (e.g., 5:1 or 10:1) would have been more informative. This becomes crucial as prior research on human macrophages indicates that Mtb infection typically hampers glycolysis, a finding inconsistent with the present study.
As the reviewer notes, important work has documented the inhibition of glycolysis in M. tuberculosis-infected macrophages dependent on the MOI (PMID 30444490). For instance, in this study, hMDMs infected at an MOI of 1 showed increased extracellular acidification and glycolytic parameters, as opposed to macrophages infected at higher MOI, or the same MOI but measured in THP1 cells. In light of these findings, we attempted to extend our study with Mo-DCs to higher MOIs, but too much cell death was induced, limiting our ability to obtain reliable metabolic measurements and functional assays from these cultures. Consistent with this, other authors reported that more than 40% of Mo-DC die after 24 hours following infection with H37Rv at an MOI of 10 (PMID 22024399, Fig 2B). We acknowledge that more comprehensive focused in vivo studies would be needed to assess the overall impact of infection. We foresee that in the context of natural infection, DC with different levels of infection will coexist, some with low bacillary load that may be able to trigger glycolysis and migrate, others highly infected and more likely to die. In this case, we are unable to provide a full explanation for the delay in the onset of the adaptive response, an aspect that requires further investigation. From our perspective, the important contribution of our work is more focused on understanding the later stage of infection, when chronic infection is established, where precursors already seem to have a limited capacity to generate DC with a good migratory performance regardless of being confronted with a low bacillary load.
To better clarify the scope and limitations of the work, we added these comments to the discussion (see discussion, lines 405-408).
The study emphasizes that Mtb infection enhances glycolysis in Mo-DCs (Fig-1 and Fig-2). Despite the authors advocating lactate as the end product (citing three reviews/opinions), the historical literature supported by detailed experimentation convincingly favors pyruvate. While the authors' attempt to support an alternate glycolytic paradigm is understandable, it is simply not necessary. This is further supported by the authors' claim that oxamate is an inhibitor of glycolysis (abstract and main text). Oxamate is a pyruvate analogue that directly inhibits the conversion of pyruvate into lactate by lactate dehydrogenase. Simply put, if oxamate was an inhibitor of glycolysis then the cells would have died.
(2) Taking into account the reviewer's suggestions, we changed the text accordingly, referring to oxamate as an LDH inhibitor, including in the abstract.
In Fig-2, clarify the term "bystander DCs." Explain why these MtbRFP- DCs exhibit distinct behavior compared to uninfected DCs, especially considering their similarity to Mtb-infected ones.
(3) To clarify these results, as correctly suggested by the reviewer, we incorporated a sentence in the results section, stating that bystander DCs are cells that are not in direct association with Mtb (Mtb-RFP-DCs), but are rather nearby and exposed to the same environment (page 7, line 145-148). In other words, bystander cells are those exposed to the same secretome and soluble factors as infected cells. Our data indicate that bystander DCs upregulate their state of glycolysis just like infected DCs do, which suggests the presence of soluble mediators induced during infection that are capable of triggering glycolysis even in uninfected cells.
These results are in line with the observation that bacteria lacking infectious capacity (such as the irradiated Mtb) also trigger glycolysis in DCs (Fig 1), likely via TLR2 receptors that are potentially activated by the release of mycobacterial antigens or bacterial debris present in the microenvironment (Fig 3). We incorporated this interpretation in the discussion of the manuscript (lines 403-408).
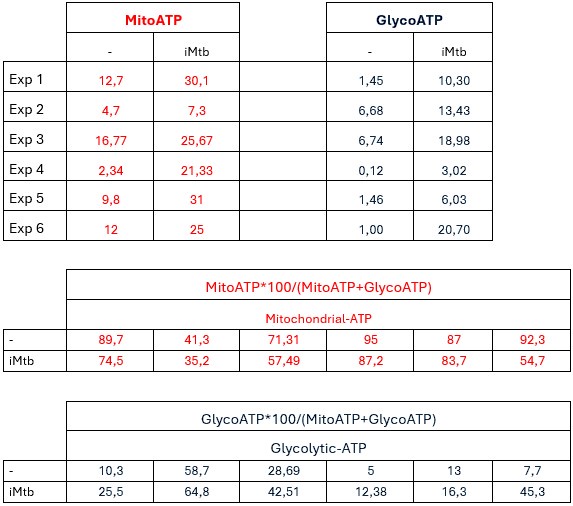
(4) Notably, the authors conducted SCENITH on both iMtb and viable Mtb (Fig-2). However, OCR, PER, and Mito- & Glyco- ATP were solely measured in MO-DCs stimulated by iMtb. Given the distinct glycolytic responses between iMtb and viable Mtb, it is crucial to assess these parameters in Mo-DCs treated with viable Mtb. Moreover, it is unclear as to how the relative ATP in Fig-2F was calculated as both Mito-ATP and Glyco-ATP is significantly high in iMtb-treated Mo-DCs (Fig-2E). Also, figure 2 contains panels with no labeling, which is confusing.
We appreciate the reviewer's suggestion that additional determinations would enrich the bioenergetic profile of DCs during infection. However, due to biosafety considerations and economic-driven limitations, we are currently unable to measure OCR, PER, and Mito- & Glyco- ATP, as these assessments require live cell cultures within BSL3 containment, if live Mtb is to be employed. Regrettably, our BSL3 facility is not equipped with a Seahorse instrument—few facilities in the world have such type of BLS3-driven investment. For this key reason, we employed SCENITH for our BSL3-based experiments.
Concerning the how ATP was calculated, we show below the raw data for Mito-ATP and Glyco-ATP results and calculations of their relative contributions.
Author response table 1.
(5) In Figures 3, 4, & 5, the consistent use of only iMtb was observed. Previous concerns about this approach were raised in the review, with the authors asserting that the use of viable Mtb was beyond the manuscript's scope. However, this claim is inaccurate. Both the authors' findings and literature elsewhere emphasize notable differences not only in host-cell metabolism but also in immune responses when treated with viable Mtb compared to dead or iMtb. Therefore, it is recommended to incorporate viable Mtb in experiments where only iMtb was utilized. Also, in the abstract (3rd sentence), do the authors refer to live or irradiated Mtb? It is imperative to clearly indicate this distinction, as the subsequent conclusions are based only on one of these two scenarios, not both. The contradictory mitochondrial mass results (figure 1; live and dead Mtb showed opposite mitochondrial mass results) clearly illustrate the profound difference live (versus dead) Mtb cells can have on an experiment.
We thank the reviewer for stating this concern. For Figure 3, the involvement of TLR2 ligation on lactate release was also confirmed with live Mtb (shown in Figure S2D). In this current version, we also confirmed the involvement of TLR2 ligation in the up-regulation of HIF-1α triggered by live Mtb (new Fig S2E). As for Figure 4, we agree that performing assays with live Mtb will add complementary information. Indeed, we hope to investigate in the future the impact of the glycolysis/HIF1a axes on the adaptive immune response. We believe that employing live bacteria and considering their active immune evasion strategies will be crucial. However, at present, this is not the focus of the current manuscript and is beyond its scope.
We also agree with the reviewer that confirmation of the migratory behavior of DCs following Mtb infection is a crucial aspect of the study. To comply with this pertinent request, we performed new migration assays using Mtb-infected DCs treated with oxamate or PX-478 to validate that the HIF1a/glycolysis axis; results convincingly demonstrate that this axis is essential for DC migration, particularly in the context of Mtb-infected cells (new Fig 5D). Having observed the same inhibitory effect of HIF1a and LDH inhibition on cell migration in either Mtb-infected or iMtb-stimulated DCs, we consider that the sentence alluded to by the reviewer in the abstract is now applicable to both contexts (page 2, line 34-36). We hope this reviewer agrees.
(6) The discussion and the graphical abstract elucidating the distinctions in glycolysis between CD16+ monocytes of HS and TB patients and iMtb-treated Mo-DCs are currently confusing and require clarification. According to the abstract, monocytes from TB patients exhibit heightened glycolysis, resulting in diminished HIF-a activity and migratory capacity of MO-DCs. This prompts a question: if exacerbated glycolysis in monocytes is associated with adverse outcomes, wouldn't it be logical to consider suppressing glycolysis? If so, how can inhibiting glycolysis, a favored metabolic pathway for pro-inflammatory responses, be beneficial for TB therapy?
We understand the reviewer’s concern about this apparent paradox. As previously mentioned in response to the public review provided by the reviewer, inhibiting glycolysis may yield varying outcomes depending on the stage of infection, as well as the cellular target (e.g., monocytes, DCs) or compartment (systemic versus local). It is imperative to delve deeper into the potential role of the HIF1α/glycolysis axis at the systemic level within the context of chronic inflammation, contrasting with its role in a local setting during the acute phase of infection.
A comprehensive understanding of the interplay between Mtb infection and glycolysis in myeloid cells requires further consideration of various contextual conditions, urging caution against oversimplified interpretations. For instance, one of the objectives of host-directed therapies (HDTs) is to mitigate host-response inflammatory toxicity, which can impede treatment efficacy (doi: 10.3389/fimmu.2021.645485). In this regard, traditional anti-inflammatory drugs such as non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids have been explored as adjunct therapies due to their immunomodulatory properties. Additionally, compounds like vitamin D, phenylbutyrate (PBA), metformin, and thalidomide, among others, have been investigated in the context of TB infections (doi:10.3389/fimmu.2017.00772), highlighting the diverse range of strategies aimed at enhancing TB treatment. These efforts extend beyond bolstering antimicrobial activity to encompass minimizing inflammation and mitigating tissue damage.
(7) I am not convinced that BubbleMap made any significant contribution to the manuscript perhaps because it is poorly described in the figure legends/main text (I am unable to determine what data set is significant or not).
We agree with the reviewer’s comment. To clarify the valuable information gleaned from these analyses, we have added interpretive guidelines on bubble color, bubble size and statistical significance in the legend of Figure 7. We hope these changes may reflect the significant contribution of the BubbleMap analysis approach to this study, which demonstrates a significant enrichment of interferon response gene expression in the monocyte compartment from patients with active TB compared to their control counterparts. Notably, this enrichment does not extend to genes associated with the OXPHOS hallmark.
(8) The use of cells/monocytes from TB patients is a concern in addition to the incomplete demographic table. In the case of the latter, absolute numbers including percentages should be included. Importantly, it appears that cells from TB patients were used, that received anti-TB drug therapy (regimen not stated) up to two weeks post diagnosis and not at baseline. This is important as recent studies have shown that anti-TB drugs modulates the bioenergetics of host cells. Lastly, what were the precise TB symptoms the authors referred to in figure 7C?
We have updated the demographic table and included the absolute numbers. We concur with the reviewer's viewpoint, particularly in light of recent findings illustrating the impact of anti-TB drug treatment on cell metabolism (doi: 10.1128/AAC.00932-21/). Again, this study underscores the complexity of such effects, which exhibit considerable variability influenced by factors such as cell type, drug concentration, and combination therapy.
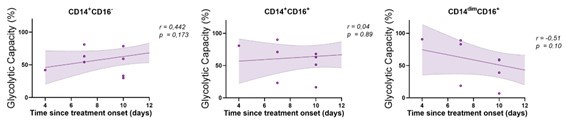
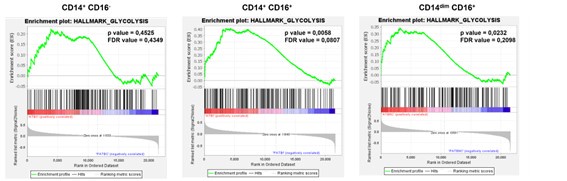
Despite this variability, our analysis involving monocytes from TB patients, who received different antibiotic combinations within short time frames (less than 15 days) reveals a marked increase in glycolysis in CD16+ monocytes compared to healthy counterparts. We did not observe a correlation between monocyte glycolytic capacity and the start time of antibiotic treatment within this 15-day window (see below, Author response image 1). These findings suggest that the antibiotic regimen does not have a significant impact on monocyte glycolytic capacity during the first 15 days. However, we did observe an effect of antibiotic treatment when comparing patients before and 2 months after treatment. Enrichment analysis of various monocyte subsets before and after 2 months of treatment (GEO accession number: GSE185372) showed that CD14dim CD16+ and CD14+ CD16+ populations had higher glycolytic activity before treatment, which is decreased then post-treatment (Author response image 2).
Author response image 1.
Correlation analysis between the baseline glycolytic capacity and the time since treatment onset for each monocyte subset (CD14+CD16-, CD14+CD16+ and CD14dimCD16+, N = 11). Linear regression lines are shown. Spearman’s rank test. The data are represented as scatter plots with each circle representing a single individual.
Author response image 2.
Gene enrichment analysis for glycolytic genes on the pairwise comparisons of each monocyte subset (CD14+CD16-, CD14+CD16+ and CD14dimCD16+) from patients with active TB pre-treatment vs patients with active TB (TB) undergoing treatment for 2 months. Comparisons with a p-value of less than 0.05 and an FDR value of less than 0.25 are considered significantly different.
Overall, our results indicate that while drug treatment does affect cell bioenergetics, this effect is not prominent within the first 15 days of treatment. CD16+ monocytes maintain high basal glycolytic activity that normalizes after treatment, contrasting with the CD16- population (even under the same circulating antibiotic doses). This highlights the intricate interplay between anti-TB drugs and cellular metabolism, underscoring the need for further research to understand the underlying mechanisms and therapeutic implications.
Finally, the term symptoms evolution refers to the time period during which a patient experiences cough and phlegm for more than 2-3 weeks, with or without sputum that may (or not) be bloody, accompanied by symptoms of constitutional illness (e.g, loss of appetite, weight loss, night sweats, general malaise). As requested, this definition has been included in the method section (page 28-29, lines 705-709).
Minor:
(1) Incorporate the abbreviation for tuberculosis "(TB)" in the first line of the abstract and similarly introduce the abbreviation for Mycobacterium tuberculosis when it is first mentioned in the abstract.
Thank you, we have amended it accordingly.
(2) As the majority of experiments are in vitro, the authors should specify the number of times each experiment was conducted for every figure.
We have included this information in each figure legend (see N for each panel). Since the majority of our approaches are conducted in vitro using primary cell cultures (specifically, human monocyte-derived DCs), we utilized samples from four to ten independent donors, not replicates, in order to account for the variability seen between donors.
(3) Rename Fig-2. Ensure consistent labeling for the metabolic dependency of uninfected, Mtb-infected, and the Bystander panel, aligning with the format used in panels A & B. Similarly, replace '-' with 'uninfected'.
We have modified the figure following most of the reviewer’s suggestions. However, we decided to keep the nomenclature “-” to denote a control condition, which can be unstimulated (panels A-B, fig 2) or uninfected cells (panels C-D, fig 2) depending on the experimental design.
(4) Discussion: It is unclear what the authors mean by 'some sort of exhausted glycolytic capacity'.
We have slightly modified the phrase.
-

eLife assessment
This useful study tests the hypothesis that monocytes purified from tuberculosis patients differentiate into dendritic cells with different migratory capacities. The authors conclude that these monocytes are metabolically pre-conditioned to differentiate, with reduced expression of Hif1a and a glycolytically exhaustive phenotype, resulting in low migratory and immunologic potential. Overall, the evidence provided is convincing, advancing the field substantively and providing novel insights.
-

Reviewer #2 (Public Review):
In the manuscript by Maio et al, the authors examined the bioenergetic mechanisms involved in the delayed migration of DC's during Mtb infection. The authors performed a series of in vitro infection experiments including bioenergetic experiments using the Agilent Seahorse XF, and glucose uptake and lactate production experiments. Also, data from SCENITH is included in the revised manuscript as well as some clinical data. This is a well written manuscript and addresses an important question in the TB field.
-

-

Author Response
The following is the authors’ response to the original reviews.
Reviewer #1 (Recommendations For The Authors):
Firstly, the authors place a great deal of emphasis on the impact of the Hif1-a inhibitor PX-478. The literature surrounding this inhibitor and its mode of action indicates that it is not a direct inhibitor of activity but that its greatest impact is on the production of Hif1-a. The authors do include another inhibitor as a control, Echinomycin, but it does not appear to be as biologically active and the panel of experiments conducted with this is extremely limited. I would be more comfortable with a full Seahorse experimental panel for Echinomycin, similar to SFig 2.G as performed with PX-478.
We thank the reviewer for their comment highlighting the different mechanisms of action of the HIF-1α inhibitors …
Author Response
The following is the authors’ response to the original reviews.
Reviewer #1 (Recommendations For The Authors):
Firstly, the authors place a great deal of emphasis on the impact of the Hif1-a inhibitor PX-478. The literature surrounding this inhibitor and its mode of action indicates that it is not a direct inhibitor of activity but that its greatest impact is on the production of Hif1-a. The authors do include another inhibitor as a control, Echinomycin, but it does not appear to be as biologically active and the panel of experiments conducted with this is extremely limited. I would be more comfortable with a full Seahorse experimental panel for Echinomycin, similar to SFig 2.G as performed with PX-478.
We thank the reviewer for their comment highlighting the different mechanisms of action of the HIF-1α inhibitors used in this article. While echinomycin inhibits the binding of HIF-1α to the hypoxia response element (HRE) thereby blocking HIF-1a DNA binding capability, PX-478 inhibits HIF-1α deubiquitination, decreases HIF-1α mRNA expression, and reduces HIF-1α translation. We have included a paragraph explaining this phenomenon in the new version of the manuscript (page 9). In addition, we extended the panel of experiments performed with echinomycin, which confirmed a marked inhibition of the glycolytic pathway when DCs were stimulated with irradiated Mtb in the presence of echinomycin as assessed by SCENITH (new Figure S3H).
Similarly, it would be of value to have Seahorse profiling that directly excludes FAO from the metabolic profile through the use of Etomoxir as an inhibitor of fatty acid oxidation, which one would assume would have no impact on the metabolic response.
In order to estimate the contribution of FAO towards fueling protein synthesis in DCs stimulated with iMtb, the FAO inhibitor etomoxir was incorporated to the SCENITH method as previously described (Adamik et al., 2022). Overall, FAO dependence was found to be less than 10% in DCs, regardless of their activation state. While mitochondrial dependence is reduced after iMtb stimulation, there is no difference in FAO dependence, suggesting that OXPHOS is primarily driven by glucose in iMtb-stimulated cells. This is consistent with HIF1α-induced increase of glucose metabolism-related genes. We have adjusted the results section to include this new result (new Figure S1).
Aside from these minor points, I believe this to be a rigorous study.
Reviewer #2 (Recommendations For The Authors):
In Fig. 1 and Fig. 2, the authors conclude that Mtb rewires the metabolism of Mo-DCs and induces both glycolysis and OXPHOS. The data shows that infection with iMtb or Mtb increases glucose uptake and lactate release, suggesting an increase in glycolysis. However, an increase in lactate is not a measure of glycolysis. Lactate is a byproduct of glycolysis; the end product of glycolysis is pyruvate.
We are grateful for the reviewer's comment, as it gives us the opportunity to explain the conceptual framework on which we based our study. Traditionally, pyruvate has been considered to be the end product of glycolysis when oxygen is present and lactate the end product under hypoxic conditions. Numerous studies have shown that lactate is produced even under aerobic conditions (Brooks, 2018). Therefore, we frame this work in accordance with this view that states that glycolysis begins with glucose as its substrate and terminates with the production of lactate as its main end product (Rogatzki, Ferguson, Goodwin, & Gladden, 2015; Schurr, 2023; Schurr & Schurr, 2017).
Secondly, since the authors have access to the Agilent Extracellular Flux Analyzer, they should have performed detailed ECAR/OCR measurements to conclusively demonstrate that both glycolysis and OXPHOS are increased in Mo. This is especially important for OXPHOS because the only readout shown for OXPHOS is an increase in mitochondrial mass (figure 1 G, H), which is not acceptable. Overall, the data does not indicate that Mtb triggers OXPHOS in the dendritic cells. It only indicates dead iMtb increases the mass of mitochondria in DCs.
The reviewer’s advice is well appreciated. However, we would like to clarify what may be a misunderstanding; that is, the assays alluded to by the reviewer were not performed on monocytes but on DCs. As advised by the reviewer, we now include the OCR measurements by Seahorse and describe the figures according to their order of appearance in the new version of the manuscript.
What happens to the mitochondrial mass when infected with live Mtb?
In response to the reviewer’s question, we determined the mitochondrial mass in infected DCs with live Mtb. In contrast to DCs treated with irradiated Mtb, those infected with live bacteria showed a clear reduction of their mitochondrial mass (modified Figure 1G). This result indicates that, although both Mtb-infected and irradiated Mtb-exposed DCs show a clear increase in their glycolytic activity, divergent responses are observed in terms of mitochondrial mass.
It will be best if the authors indicate in the figure headings that dead Mtb was used.
We agree with the reviewer. For figures 1-3, we applied the term “Mtb” in the figure headings since both irradiated and viable bacteria were used for the corresponding experiments. In figures 4-5, the term “iMtb” (alluding to irradiated Mtb) was used in the figure headings as suggested by the reviewer. For the remaining figures, the term “iMtb” was indicated in their legends when dead bacteria weres used to stimulate DCs.
E.g., Figure 1F; what does live Mtb do to GLUT1 levels etc etc?
In response to the reviewer’s question, we included new data about Glut1 expression in DCs infected with live Mtb in the latest version of the manuscript. In line with the increase in glucose uptake shown in figure 1B, we observed an increase in the percentage of Glut1 positive DCs upon Mtb infection (new Figure 1F, lower panels). The increase in Glut1 expression strengthens the notion that DCs activates their glycolytic activity in response to the infection, as demonstrated by the elevated release of lactate, glucose consumption, HIF-1α expression, LDHA expression (Figure 1) and glycolytic activity (Figure 2, SCENITH results with viable Mtb). Therefore, these data strongly support the induction of glycolysis by Mtb (either viable or irradiated) in DCs.
Also, we found that they were still able to activate CD4+ T cells from PPD+ donors in response to iMtb. This activation of CD4 T cells with iMtb in the presence of a HIF-1alpha inhibitor is expected, as iMtb is dead and not virulent. What happens when the cells are infected with live virulent Mtb?
We would like to clarify the main purpose of the DC-T cells co-culture assays in the presence of the HIF-1α inhibitors. To characterize the impact of HIF-1α on DC functionality, we assessed the capacity of DCs to activate autologous CD4+ T cells when stimulated with iMtb in the presence of HIF-1α inhibitors. To this end, we used iMtb merely as a source of antigens to load DCs and evaluate the effect of HIF-1α inhibition on the activation of antigen-specific T cell. The use of viable Mtb may introduce confounding factors, such as pathogen-triggered inhibitory mechanisms (e.g., EsxH secretion by Mtb, (Portal-Celhay et al., 2016)), which would prevent us from reaching conclusions about the role of HIF-1α. Thus, we consider that the use of live bacteria for this experiment is out of the scope of this manuscript.
The authors demonstrated that CD16+ monocytes from TB patients have higher glycolytic capacity than healthy controls Fig 7. The authors should differentiate TB patient monocytes into DCs and measure their bioenergetics to test if infection alters their glycolysis and OXPHOS.
In agreement with the reviewer, the determination of metabolic pathways in DCs differentiated from monocytes of TB patients is a key aspect of this work. Accordingly, the bioenergetic determinations of DCs generated from monocytes from TB patients versus healthy subjects are now illustrated in Figures 6F (lactate release) and 6G (SCENITH profile).
In the discussion, the authors state that "pathologically active glycolysis in monocytes from TB patients leads to poor glycolytic induction and migratory capacities of monocyte-derived DCs." However, the data from Fig. 1 and 2 show that treatment with iMtb or Mtb induces glycolysis in MoDCs. How do the authors explain these contrasting results?
We thank the reviewer for pointing out this issue. Figures 1 and 2 show DCs differentiated from monocytes of healthy donors (HS). In this case, DCs from HS respond to Mtb by inducing a glycolytic and migratory profile. Yet, in the case of monocytes isolated from TB patients, these cells exhibit an early glycolytic profile from the beginning of differentiation, ultimately yielding DCs with low glycolytic capacity and low migratory activity in response to Mtb. We included this explanation in the discussion (page 18) to better clarify this issue.
Also, the term "pathological" active glycolysis (Introduction and Discussion) is an inappropriate term.
As requested by the reviewer, we excluded the term “pathological” to describe the phenomenon reported in this study.
Lastly, it should be shown whether the DCs generated from CD16+ monocyte from TB patients generate tolerogenic and/or aberrant DCs, which have lower glycolytic and migration capacity compared to the CD16- monocyte population. In Figure 7B, the authors should discuss why the CD16+ monocyte population has lower glycolytic capacity compared to CD16- monocytes in healthy donors. Furthermore, in contrast to the TB patients, do DCs generated from CD16+ monocyte in healthy donors have increased glycolytic and migration capacity compared to CD16- monocyte (because these monocytes showed lower glycolytic capacity)? Furthermore, if there is no difference in glycolytic capacity among the three monocyte populations in TB patients, on what basis was it concluded that DCs generated only from the CD16+ monocyte population may be the cause of lower migration capacity? The authors state in Figure 7F that the DMOG pretreatment matches the situation where the Mo-DCs from TB patients showed reduced migration. Did the authors check the Hif-1alpha levels in monocytes obtained from TB patients?
We appreciate this in-depth analysis by the reviewer because it allows us to clarify some interpretations of the SCENITH results in Figure 7B. It is important to keep in mind that with the SCENITH technique we can only infer about the relative contributions between the metabolic pathways, without alluding to the absolute magnitudes of such contributions. In this regard, it is key to note that the amount of lactate released during the first hours of the TB monocyte culture is much higher than that released by monocytes from healthy subjects (HS, Figure 7A), even when most of monocytes, which are CD14+ CD16-, have comparable glycolytic capacities between HS and TB. Another example to illustrate how to interpret SCENITH results can be found in Figure 2, where a lower mitochondrial dependence is observed in iMtb-stimulated DCs (Figure 2A), while the absolute ATP production associated to OXPHOS is indeed higher as measured by Seahorse (Figure 2D). Therefore, the glycolytic capacity is not a direct readout of the magnitude of glycolysis, but of its contribution to total metabolism. The low levels of lactate released from HS monocytes likely reflects their low activation state and low metabolic activity compared to TB monocytes. In this regard, we have previously demonstrated that monocytes from pulmonary TB patients display an activated phenotype (Balboa et al., 2011). The fact that there is no difference between the glycolytic capacities of TB and HS CD16- monocytes indicates that their proportional contributions to protein synthesis are comparable (again, without inferring about their absolute values, which may be very different).
Beyond the previous clarification, the reviewer's proposal to isolate subsets of monocytes is a very interesting idea. However, the experimental approach is very difficult based on the amount of blood we can obtain from patients. The cohort of patients included in this work comprises very severe patients and we are given up to 15-20 ml of peripheral blood from each. This volume of blood yields up to 10 million PBMC with approximately 1 million monocytes. If we separate the monocyte subsets, the recovered cells per condition will be insufficient to perform the intended assays.
Nevertheless, we incorporate new evidence that TB disease is associated with an increased activation and glycolytic profile of circulating CD16+ monocytes.
i) First, we show that the baseline glycolytic capacity of CD16+ monocytes correlates with time since the onset of TB-related symptoms (new Figure 7C).
ii) Second, we performed high-throughput GeneSet Enrichment Analysis (GSEA) on transcriptomic data (GEO accession number: GSE185372) of CD14+CD16-, CD14+CD16+ and CD14dimCD16+ monocytes isolated from individuals with active TB, latent TB (IGRA+), as well as from TB negative healthy controls (IGRA-). We found enrichments that, unlike oxidative phosphorylation, glycolysis tends to increase in active TB in both CD14+CD16+ and CD14dimCD16+ monocytes (new Figure 7D).
iii) We measured the expression of HIF-1α in monocyte subsets by FACS and found that this transcription factor is expressed at higher levels in CD16+ monocyte subsets from TB patients compared to their counterparts from healthy donors (new Figure 8 A). We consider this result justifies the assays shown in Figure 8B-C, in which we prematurely activated HIF-1α in healthy donor monocytes during early differentiation to DCs and measured its impact on the migration of the generated DCs.
In the Discussion, the authors mention that circulating monocytes from TB patients differentiate from DCs with low immunogenic potential. However, the authors have not shown any immunological defect in any of their data with monocytes from TB patients. In the proxy model mentioned in Figure 7, they have in fact shown that these preconditioned DCs have higher CD86 expression. Can the authors explain/show data to justify the statement in the first paragraph of the Discussion?
We agree with the reviewer on this observation. Our findings are limited to the generation of DCs with low migratory potential (low chemotactic activity towards CCL21 of DC differentiated from TB patient monocytes shown in figure 6H and of DC generated from pre-conditioned monocytes shown in figure 8C). We have modified that part of the discussion to better clarify this point, replacing migratory with immunogenic.
The authors should note that oxamate is a competitive inhibitor of the enzyme lactate dehydrogenase and not glycolysis. Also, LDHA catalyzes the conversion from pyruvate to lactate and not the other way around (Results, page 6).
This comment relates to the first one by the reviewer, in which the dogma of glycolysis was discussed. According to the new conception of glycolysis, it begins with glucose as its substrate and terminates with the production of lactate as its main end product.
The following statements by the authors on page 6 are incorrect: "Because irradiated and viable Mtb induced comparable activation of glycolysis, we subsequently performed all our assays with irradiated Mtb only in the rest of the study due to biosafety reasons." and: "To our knowledge, this is the first study addressing the metabolic status and migratory activity of Mo-DCs from TB patients."
We deleted the first sentence and reworded the second sentence as "To our knowledge, this is the first study to address how the metabolic status of monocytes from TB patients influences the migratory activity of further differentiated DCs".
The Discussion reads as if live Mtb was used in the experiments, which is not the case. This should be corrected.
We changed Mtb for iMtb when it was the case in the discussion. In some cases, Mtb stimulation was used instead of Mtb infection.
Minor Comments:
(1) In Figure 1F legend "Quantification of Glut1+ cells plotted to the right". The underlined part should be "plotted below".
It was corrected.
(2) In Figure 1H. Please describe the quantitation method and describe how many cells or the number/size of fields were used to quantitate mitochondria.
For mitochondrial morphometric analysis, TEM images were quantified with the ImageJ “analyze particles” plugin in thresholded images, with size (μm2) settings from 0.001 to infinite. For quantification, 8–10 cells of random fields (1000x magnification) per condition were analyzed. We included this information in the methods section of the new version of the manuscript.
(3) Please mention the number of independent experimental repeats for each experimental data set and figure.
In each figure, the number of independent experiments is indicated by individual dots.
(4) In Figure 2A legend, "PER; left panel" should be PER; lower panel and "OCR; right panel" should be OCR; upper panel.
It was corrected.
References for reviewers
Adamik, J., Munson, P. V., Hartmann, F. J., Combes, A. J., Pierre, P., Krummel, M. F., … Butterfield, L. H. (2022). Distinct metabolic states guide maturation of inflammatory and tolerogenic dendritic cells. Nature Communications 2022 13:1, 13(1), 1–19. https://doi.org/10.1038/s41467-022-32849-1
Balboa, L., Romero, M. M., Basile, J. I., Sabio y Garcia, C. A., Schierloh, P., Yokobori, N., … Aleman, M. (2011). Paradoxical role of CD16+CCR2+CCR5+ monocytes in tuberculosis: efficient APC in pleural effusion but also mark disease severity in blood. Journal of Leukocyte Biology. https://doi.org/10.1189/jlb.1010577
Brooks, G. A. (2018). Cell Metabolism The Science and Translation of Lactate Shuttle Theory. Cell Metab. https://doi.org/10.1016/j.cmet.2018.03.008
Portal-Celhay, C., Tufariello, J. M., Srivastava, S., Zahra, A., Klevorn, T., Grace, P. S., … Philips, J. A. (2016). Mycobacterium tuberculosis EsxH inhibits ESCRT-dependent CD4+ T-cell activation. Nature Microbiology, 2, 16232. https://doi.org/10.1038/NMICROBIOL.2016.232
Rogatzki, M. J., Ferguson, B. S., Goodwin, M. L., & Gladden, L. B. (2015). Lactate is always the end product of glycolysis. Frontiers in Neuroscience, 9(FEB), 125097. https://doi.org/10.3389/FNINS.2015.00022/BIBTEX
Schurr, A. (2023). From rags to riches: Lactate ascension as a pivotal metabolite in neuroenergetics. Frontiers in Neuroscience, 17, 1145358. https://doi.org/10.3389/FNINS.2023.1145358/BIBTEX
Schurr, A., & Schurr, A. (2017). Lactate, Not Pyruvate, Is the End Product of Glucose Metabolism via Glycolysis. Carbohydrate. https://doi.org/10.5772/66699
-

eLife assessment
This useful study tests the hypothesis that Mycobacterium tuberculosis infection increases glycolysis in monocytes, which alters their capacity to migrate to lymph nodes as monocyte-derived dendritic cells. The authors conclude that infected monocytes are metabolically pre-conditioned to differentiate, with reduced expression of Hif1a and a glycolytically exhaustive phenotype, resulting in low migratory and immunologic potential. However, the evidence is incomplete as the use of live and dead mycobacteria still limits the ability to draw firm conclusions. The study will be of interest to microbiologists and infectious disease scientists.
-

Reviewer #3 (Public Review):
In the revised manuscript by Maio et al, the authors examined the bioenergetic mechanisms involved in the delayed migration of DC's during Mtb infection. The authors performed a series of in vitro infection experiments including bioenergetic experiments using the Agilent Seahorse XF, and glucose uptake and lactate production experiments. Also, data from SCENITH is included in the revised manuscript as well as some clinical data. This is a well written manuscript and addresses an important question in the TB field. A remaining weakness is the use of dead (irradiated) Mtb in several of the new experiments and claims where iMtb data were used to support live Mtb data. Another notable weakness lies in the author's insistence on asserting that lactate is the ultimate product of glycolysis, rather than …
Reviewer #3 (Public Review):
In the revised manuscript by Maio et al, the authors examined the bioenergetic mechanisms involved in the delayed migration of DC's during Mtb infection. The authors performed a series of in vitro infection experiments including bioenergetic experiments using the Agilent Seahorse XF, and glucose uptake and lactate production experiments. Also, data from SCENITH is included in the revised manuscript as well as some clinical data. This is a well written manuscript and addresses an important question in the TB field. A remaining weakness is the use of dead (irradiated) Mtb in several of the new experiments and claims where iMtb data were used to support live Mtb data. Another notable weakness lies in the author's insistence on asserting that lactate is the ultimate product of glycolysis, rather than acknowledging a large body of historical data in support of pyruvate's role in the process. This raises a perplexing issue highlighted by the authors: if Mtb indeed upregulates glycolysis, one would expect that inhibiting glycolysis would effectively control TB. However, the reality contradicts this expectation. Lastly, the examination of the bioenergetics of cells isolated from TB patients undergoing drug therapy, rather than studying them at their baseline state is a weakness.
-

-

Author Response
On behalf of the authors of the article "Elevated glycolytic metabolism of monocytes limits the generation of HIF-1α-driven migratory dendritic cells in tuberculosis", I would like to provide interim responses noting some relevant points about eLife assessment and public reviews,
eLife assessment
This useful study tests the hypothesis that Mycobacterium tuberculosis infection increases glycolysis in monocytes, which alters their capacity to migrate to lymph nodes as monocyte-derived dendritic cells. The authors conclude that infected monocytes are metabolically pre-conditioned to differentiate, with reduced expression of Hif1a and a glycolytically exhaustive phenotype, resulting in low migratory and immunologic potential. Unfortunately, the evidence for the conclusions is currently incomplete, as the use of dead …
Author Response
On behalf of the authors of the article "Elevated glycolytic metabolism of monocytes limits the generation of HIF-1α-driven migratory dendritic cells in tuberculosis", I would like to provide interim responses noting some relevant points about eLife assessment and public reviews,
eLife assessment
This useful study tests the hypothesis that Mycobacterium tuberculosis infection increases glycolysis in monocytes, which alters their capacity to migrate to lymph nodes as monocyte-derived dendritic cells. The authors conclude that infected monocytes are metabolically pre-conditioned to differentiate, with reduced expression of Hif1a and a glycolytically exhaustive phenotype, resulting in low migratory and immunologic potential. Unfortunately, the evidence for the conclusions is currently incomplete, as the use of dead mycobacteria will affect bioenergetic readouts. The study will be of interest to microbiologists and infectious disease scientists.
We would like to clarify what may be a misunderstanding. Indeed, the study did not deal with “infected monocytes” per se, but rather with the ability of monocytes purified from TB patients vs. healthy control to differentiate into DCs with different migratory capacities upon Mtb infection or stimulation. Since there is no evidence for the presence of Mtb in the patient’s blood, the metabolic effects we observed are likely a consequence of systemic pulmonary disease rather than of direct interaction of monocytes with Mtb. Although irradiated Mtb was used in most experiments, in particular because Seahorse and other technologies cannot be used in our BSL3 laboratory, we provide evidence (Figure 1) that infecting DCs with live Mtb or stimulating DCs with irradiated Mtb generates comparable glycolytic profiles (release of lactate, glucose consumption, HIF1a expression and LDHA expression). To strengthen the relevance of our data, we will characterize the metabolism of DCs infected with live Mtb using SCENITH.
Reviewer #1 (Public Review):
The manuscript by Maio and colleagues looks at the impact of the heightened glycolytic activity induced by Mtb in monocytes, and its impact on Hif1-a dependent migration of DCs.
Data concerning the biological significance of the impact of enhanced glycolysis on DC migration is strong and convincing. While Hif1-a is obviously a key factor, the evidence that it is a linear component in the cascade falls a little short as the main inhibitor used PX-478 does not have a clear, single mode of action. Additional characterization with the alternative inhibitor (Echinomycin) would make the argument more convincing.
We would like to thank the reviewer for their positive assessment of our manuscript. Although Echinomycin has been used for validating some of the representative experiments performed in our study (see supplementary figure 2E-F), we agree with the reviewer’s suggestion. Therefore, additional experiments using echinomycin will be carried out to confirm our results.
Reviewer #2 (Public Review):
The manuscript by Maio et al attempts to examine the bioenergetic mechanisms involved in the delayed migration of DC's during Mtb infection. The authors performed a series of in vitro infection experiments including bioenergetic experiments using the Agilent Seahorse XF, and glucose uptake and lactate production experiments. This is a well-written manuscript and addresses an important question in the TB field. A major weakness is the use of dead Mtb in virtually all the experiments. Unfortunately, the authors did not attempt to address this critical confounding factor. As a result, data was interpreted, and conclusions were made as if live Mtb was used. Also, previous studies (PMID: 30444490 and PMID: 31914380) have shown that live Mtb suppresses glycolysis, which contradicts findings in this study, perhaps because dead Mtb was used here. For these reasons, obtaining any pertinent conclusions from the study is not possible, which diminishes the significance of the work.
We thank the reviewer for their evaluation of our study. We agree that using live Mtb in all experiments would have been ideal. However, we do not have a Seahorse Analyzer in our BSL3 facility. Thus, we will characterize the metabolism of DCs infected with live Mtb using SCENITH during revision of our manuscript.
With regard to the differences between our results and those of previous studies showing Mtb-induced suppression of glycolysis, they could be explained by the use of different Mtb strains, different multiplicity of infection (MOI), macrophages of different origins, and different measurement timepoints, as discussed in one of these publications (PMID 30444490). For instance, in PMID 30444490, hMDMs infected at an MOI of 1 showed increased extracellular acidification and glycolytic parameters, as opposed to higher MOI or the same MOI but measured in THP1 cells. Importantly, the aforementioned articles studied macrophage and not DC metabolism. These aspects will be discussed in a revised manuscript.
-

eLife assessment
This useful study tests the hypothesis that Mycobacterium tuberculosis infection increases glycolysis in monocytes, which alters their capacity to migrate to lymph nodes as monocyte-derived dendritic cells. The authors conclude that infected monocytes are metabolically pre-conditioned to differentiate, with reduced expression of Hif1a and a glycolytically exhaustive phenotype, resulting in low migratory and immunologic potential. Unfortunately, the evidence for the conclusions is currently incomplete, as the use of dead mycobacteria will affect bioenergetic readouts. The study will be of interest to microbiologists and infectious disease scientists.
-

Reviewer #1 (Public Review):
The manuscript by Maio and colleagues looks at the impact of the heightened glycolytic activity induced by Mtb in monocytes, and its impact on Hif1- dependent migration of DCs.
Data concerning the biological significance of the impact of enhanced glycolysis on DC migration is strong and convincing. While Hif1-a is obviously a key factor, the evidence that it is a linear component in the cascade falls a little short as the main inhibitor used PX-478 does not have a clear, single mode of action. Additional characterization with the alternative inhibitor (Echinomycin) would make the argument more convincing.
-

Reviewer #2 (Public Review):
The manuscript by Maio et al attempts to examine the bioenergetic mechanisms involved in the delayed migration of DC's during Mtb infection. The authors performed a series of in vitro infection experiments including bioenergetic experiments using the Agilent Seahorse XF, and glucose uptake and lactate production experiments. This is a well-written manuscript and addresses an important question in the TB field. A major weakness is the use of dead Mtb in virtually all the experiments. Unfortunately, the authors did not attempt to address this critical confounding factor. As a result, data was interpreted, and conclusions were made as if live Mtb was used. Also, previous studies (PMID: 30444490 and PMID: 31914380) have shown that live Mtb suppresses glycolysis, which contradicts findings in this study, perhaps …
Reviewer #2 (Public Review):
The manuscript by Maio et al attempts to examine the bioenergetic mechanisms involved in the delayed migration of DC's during Mtb infection. The authors performed a series of in vitro infection experiments including bioenergetic experiments using the Agilent Seahorse XF, and glucose uptake and lactate production experiments. This is a well-written manuscript and addresses an important question in the TB field. A major weakness is the use of dead Mtb in virtually all the experiments. Unfortunately, the authors did not attempt to address this critical confounding factor. As a result, data was interpreted, and conclusions were made as if live Mtb was used. Also, previous studies (PMID: 30444490 and PMID: 31914380) have shown that live Mtb suppresses glycolysis, which contradicts findings in this study, perhaps because dead Mtb was used here. For these reasons, obtaining any pertinent conclusions from the study is not possible, which diminishes the significance of the work.
-