Efficient differentiation of human primordial germ cells through geometric control reveals a key role for Nodal signaling
Curation statements for this article:-
Curated by eLife
Evaluation Summary:
This manuscript uses a micropatterned cell culture system to explore the mechanism of human primordial germ cell-like cell specification from human pluripotent stem cells, and proposes a previously unrecognized role of NODAL signaling operating downstream of BMP signaling. The strength of the manuscript is the development of a simple and efficient in vitro system that is potentially suitable for exploring the mechanism of human primordial germ cell-like cell specification. With a more rigorous validation of the identity of the studied cells and more discussion relating the findings to developmental mechanisms in vivo, this study will be of interest to stem cell and developmental biologists.
(This preprint has been reviewed by eLife. We include the public reviews from the reviewers here; the authors also receive private feedback with suggested changes to the manuscript. The reviewers remained anonymous to the authors.)
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
Human primordial germ cells (hPGCs) form around the time of implantation and are the precursors of eggs and sperm. Many aspects of hPGC specification remain poorly understood because of the inaccessibility of the early postimplantation human embryo for study. Here, we show that micropatterned human pluripotent stem cells (hPSCs) treated with BMP4 give rise to hPGC-like cells (hPGCLC) and use these as a quantitatively reproducible and simple in vitro model to interrogate this important developmental event. We characterize micropatterned hPSCs up to 96 hr and show that hPGCLC populations are stable and continue to mature. By perturbing signaling during hPGCLC differentiation, we identify a previously unappreciated role for Nodal signaling and find that the relative timing and duration of BMP and Nodal signaling are critical parameters controlling the number of hPGCLCs. We formulate a mathematical model for a network of cross-repressive fates driven by Nodal and BMP signaling, which predicts the measured fate patterns after signaling perturbations. Finally, we show that hPSC colony size dictates the efficiency of hPGCLC specification, which led us to dramatically improve the efficiency of hPGCLC differentiation.
Article activity feed
-

Author Response:
Reviewer #1 (Public Review):
Jo et al. use a combination of micropatterned differentiation, single cell RNA sequencing and pharmacological treatments to study primordial germ cell (PGC) differentiation starting from human pluripotent stem cells. Geometrical confinement in conjunction with a pre-differentiation step allowed the authors to reach remarkable differentiation efficiencies. While Minn et al. already reported the presence of PGC-like cells in micropatterned differentiating human cultures by scRNA-Seq (as acknowledged by the authors), the careful characterization of the PGC-like population using immunostainings and scRNA-Seq is a strength of the manuscript. The attempt at mechanistically dissecting the signaling pathways required for PGC fate specification is somehow weaker. The authors do not present …
Author Response:
Reviewer #1 (Public Review):
Jo et al. use a combination of micropatterned differentiation, single cell RNA sequencing and pharmacological treatments to study primordial germ cell (PGC) differentiation starting from human pluripotent stem cells. Geometrical confinement in conjunction with a pre-differentiation step allowed the authors to reach remarkable differentiation efficiencies. While Minn et al. already reported the presence of PGC-like cells in micropatterned differentiating human cultures by scRNA-Seq (as acknowledged by the authors), the careful characterization of the PGC-like population using immunostainings and scRNA-Seq is a strength of the manuscript. The attempt at mechanistically dissecting the signaling pathways required for PGC fate specification is somehow weaker. The authors do not present sufficient evidence supporting the ability to specify PGC fate in the absence of Wnt signaling and the importance of the relative signaling levels of BMP to Nodal pathways; the wording of the text should be amended to better reflect the presented evidence or the authors should perform additional experiments to support these claims.
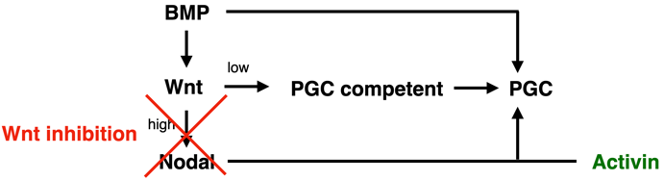
We thank the reviewer for this comment. As described in more detail in the responses below, we have significantly strengthened the evidence for the rescue of Wnt inhibition by exogenous Activin treatment and have nuanced our interpretation. We believe that our data suggest low levels of Wnt may be required directly for PGC competence, while much higher levels are required indirectly to induce Nodal, with Nodal signaling being the limiting factor for PGC specification under the reference condition with BMP4 treatment only. We describe this in detail in the manuscript but summarize it here in a simplified diagram:
We have also carried out additional experiments that match model predictions demonstrating the importance of relative BMP and Nodal signaling levels and amended the text to reflect the evidence as suggested. More details are provided below.
The molecular characterization of why colonies confined to small areas differentiate much better would greatly increase the biological significance of the manuscript (the technical achievement of reaching such efficiency is impressive on its own).
We believe the mechanism by which cells confined to small colonies differentiate to PGCLCs more efficiently is explained by a larger fraction of the cells being exposed to the necessary levels of BMP and Nodal signaling. In large colonies BMP signaling was shown to be restricted to a distance of 50-100 um from the colony edge through receptor localization and secretion of inhibitors (Etoc et al, Dev Cell 2016). From this one would expect that BMP signaling extends a similar distance from the edge in small colonies, so that a larger fraction of cells are receiving the BMP signal needed to differentiate to PGCLCs. Because it was not previously shown that the length scale of BMP signaling and downstream signals are preserved as colony size is reduced, we have now included an analysis of BMP signaling (pSmad1 levels) and Nodal signaling (nuclear Smad2/3 levels) as a function of colony size (Figure 5i-k). This confirms our hypothesis and provides a potential mechanism.
The authors propose a mathematical model based on BMP and Nodal signaling that qualitatively recapitulate their experimental data. While the authors should be commended for providing examples of other simple models that do not fully recapitulate their data, it would have been nice to see an attempt at challenging quantitatively the model. In particular, the authors do not take advantage of the ability to explore in a more systematic manner the BMP/Nodal phase space with their system.
We thank the reviewer for this suggestion. Experimentally we have now tested the effect of 5x5 = 25 different combinations of BMP and Activin doses on PGCLC differentiation. We then challenged the mathematical model to predict the ‘phase diagram’ corresponding to this data with good agreement (Figure 6f). It is important to note here that the model was fit using only data with 50ng/ml of BMP, making this a true prediction. We also point out that the phase diagram predicted in this way is different from the one shown in Figure 6d, not only because of the lower resolution, but because Figure 6f shows the steady state after uniform stimulation in space and time (i.e. the response on the very edge), whereas the predicted phase diagram shows average expression at 42h in a 100um range from the colony edge using the previously measured spatiotemporal gradients of BMP and Activin response. Finally, the data in Figure 6f shows mean expression levels as opposed to the percentage double positive cells for the same data in Figure 4q because our model does not simulate individual cells and noise, only allowing us to compare mean expression. We explain all this in the text now. As a minor change to facilitate comparison of data and model we have now plotted the concentrations of BMP and Activin in Figure 6 rather than the scaled model parameters from 0 to 1, we also further optimized the model parameters without qualitative changes.
The authors' claim that PGCLC formation can be rescued by exogenous Activin when blocking endogenous Wnt production is surprising given the literature. The authors only show that they can restore a TFAP2C+SOX17+ population but do not actually stain for an established germ cell marker. It appears essential to perform a PRDM1 staining in these conditions (Figure 4A) to unambiguously identify this population.
We have significantly extended our analysis of the effect of WNT inhibition and subsequent rescue of PGCs by Activin treatment. This includes staining for TFAP2C,NANOG,PRDM1 and staining for LEF1 as a measure of WNT signaling. Figure 4 and Figure 4—figure supplement 1 now also include treatment with IWR-1, a different small molecule inhibitor of WNT signaling, as well inhibition by IWR-1 and IWP2 at different times and different doses.
The authors only provide weak evidence that the fates depend on the relative signaling levels of BMP and Nodal. Indeed, fewer cells acquire a fate the lower BMP concentration they use, including the fates marked by Sox17 expression. It would more convincing to show the assay of Figure 4F for a range of BMP concentrations at which the overall differentiation works sufficiently well.
As suggested, we have now included a range of BMP concentrations. The reduction in PGCs at lower BMP doses is in line with our model and does not contradict a dependence on the relative signaling levels of BMP and Nodal by which we mean that optimal dose of Activin for PGCLC specification depends on the level of BMP and vice versa. We have amended the text to state this more clearly.
References
Chen, Di, Na Sun, Lei Hou, Rachel Kim, Jared Faith, Marianna Aslanyan, Yu Tao, et al. 2019. “Human Primordial Germ Cells Are Specified From Lineage-Primed Progenitors..” Cell Reports 29 (13): 4568–4582.e5. doi:10.1016/j.celrep.2019.11.083.
Etoc, Fred, Jakob Metzger, Albert Ruzo, Christoph Kirst, Anna Yoney, M Zeeshan Ozair, Ali H Brivanlou, and Eric D Siggia. 2016. “A Balance Between Secreted Inhibitors and Edge Sensing Controls Gastruloid Self-Organization..” Developmental Cell 39 (3): 302–15. doi:10.1016/j.devcel.2016.09.016.
Kobayashi, Toshihiro, Haixin Zhang, Walfred W C Tang, Naoko Irie, Sarah Withey, Doris Klisch, Anastasiya Sybirna, et al. 2017. “Principles of Early Human Development and Germ Cell Program From Conserved Model Systems..” Nature 546 (7658): 416–20. doi:10.1038/nature22812.
Kojima, Yoji, Kotaro Sasaki, Shihori Yokobayashi, Yoshitake Sakai, Tomonori Nakamura, Yukihiro Yabuta, Fumio Nakaki, et al. 2017. “Evolutionarily Distinctive Transcriptional and Signaling Programs Drive Human Germ Cell Lineage Specification From Pluripotent Stem Cells..” Cell Stem Cell 21 (4): 517–532.e5. doi:10.1016/j.stem.2017.09.005.
Sasaki, Kotaro, Tomonori Nakamura, Ikuhiro Okamoto, Yukihiro Yabuta, Chizuru Iwatani, Hideaki Tsuchiya, Yasunari Seita, et al. 2016. “The Germ Cell Fate of Cynomolgus Monkeys Is Specified in the Nascent Amnion..” Developmental Cell 39 (2): 169–85. doi:10.1016/j.devcel.2016.09.007.
Tyser, R.C.V., Mahammadov, E., Nakanoh, S. et al. Single-cell transcriptomic characterization of a gastrulating human embryo. Nature 600, 285–289 (2021). https://doi.org/10.1038/s41586-021-04158-y
-

Evaluation Summary:
This manuscript uses a micropatterned cell culture system to explore the mechanism of human primordial germ cell-like cell specification from human pluripotent stem cells, and proposes a previously unrecognized role of NODAL signaling operating downstream of BMP signaling. The strength of the manuscript is the development of a simple and efficient in vitro system that is potentially suitable for exploring the mechanism of human primordial germ cell-like cell specification. With a more rigorous validation of the identity of the studied cells and more discussion relating the findings to developmental mechanisms in vivo, this study will be of interest to stem cell and developmental biologists.
(This preprint has been reviewed by eLife. We include the public reviews from the reviewers here; the authors also receive …
Evaluation Summary:
This manuscript uses a micropatterned cell culture system to explore the mechanism of human primordial germ cell-like cell specification from human pluripotent stem cells, and proposes a previously unrecognized role of NODAL signaling operating downstream of BMP signaling. The strength of the manuscript is the development of a simple and efficient in vitro system that is potentially suitable for exploring the mechanism of human primordial germ cell-like cell specification. With a more rigorous validation of the identity of the studied cells and more discussion relating the findings to developmental mechanisms in vivo, this study will be of interest to stem cell and developmental biologists.
(This preprint has been reviewed by eLife. We include the public reviews from the reviewers here; the authors also receive private feedback with suggested changes to the manuscript. The reviewers remained anonymous to the authors.)
-

Reviewer #1 (Public Review):
Jo et al. use a combination of micropatterned differentiation, single cell RNA sequencing and pharmacological treatments to study primordial germ cell (PGC) differentiation starting from human pluripotent stem cells. Geometrical confinement in conjunction with a pre-differentiation step allowed the authors to reach remarkable differentiation efficiencies. While Minn et al. already reported the presence of PGC-like cells in micropatterned differentiating human cultures by scRNA-Seq (as acknowledged by the authors), the careful characterization of the PGC-like population using immunostainings and scRNA-Seq is a strength of the manuscript. The attempt at mechanistically dissecting the signaling pathways required for PGC fate specification is somehow weaker. The authors do not present sufficient evidence …
Reviewer #1 (Public Review):
Jo et al. use a combination of micropatterned differentiation, single cell RNA sequencing and pharmacological treatments to study primordial germ cell (PGC) differentiation starting from human pluripotent stem cells. Geometrical confinement in conjunction with a pre-differentiation step allowed the authors to reach remarkable differentiation efficiencies. While Minn et al. already reported the presence of PGC-like cells in micropatterned differentiating human cultures by scRNA-Seq (as acknowledged by the authors), the careful characterization of the PGC-like population using immunostainings and scRNA-Seq is a strength of the manuscript. The attempt at mechanistically dissecting the signaling pathways required for PGC fate specification is somehow weaker. The authors do not present sufficient evidence supporting the ability to specify PGC fate in the absence of Wnt signaling and the importance of the relative signaling levels of BMP to Nodal pathways; the wording of the text should be amended to better reflect the presented evidence or the authors should perform additional experiments to support these claims. The molecular characterization of why colonies confined to small areas differentiate much better would greatly increase the biological significance of the manuscript (the technical achievement of reaching such efficiency is impressive on its own).
The authors propose a mathematical model based on BMP and Nodal signaling that qualitatively recapitulate their experimental data. While the authors should be commended for providing examples of other simple models that do not fully recapitulate their data, it would have been nice to see an attempt at challenging quantitatively the model. In particular, the authors do not take advantage of the ability to explore in a more systematic manner the BMP/Nodal phase space with their system.
The authors' claim that PGCLC formation can be rescued by exogenous Activin when blocking endogenous Wnt production is surprising given the literature. The authors only show that they can restore a TFAP2C+SOX17+ population but do not actually stain for an established germ cell marker. It appears essential to perform a PRDM1 staining in these conditions (Figure 4A) to unambiguously identify this population.
The authors only provide weak evidence that the fates depend on the relative signaling levels of BMP and Nodal. Indeed, fewer cells acquire a fate the lower BMP concentration they use, including the fates marked by Sox17 expression. It would more convincing to show the assay of Figure 4F for a range of BMP concentrations at which the overall differentiation works sufficiently well.
-

Reviewer #2 (Public Review):
This manuscript uses a micropatterned differentiation system to explore the mechanism of human primordial germ cell-like cell (hPGCLC) specification and proposes a previously un-recognized role of NODAL signaling operating downstream of BMP signaling. The strength of the manuscript is the development of a simple in vitro system that is potentially suitable for exploring the mechanism of human primordial germ cell-like cell (hPGCLC) specification. The weakness of the manuscript is the lack of rigorous validation of the identity of the hPGCLCs appeared in the micropatterned differentiation system and the lack of the discussion relevant to the in vivo developmental mechanisms.
-

-