Differential dopaminergic modulation of spontaneous cortico–subthalamic activity in Parkinson’s disease
Curation statements for this article:-
Curated by eLife
Evaluation Summary:
This manuscript is of broad interest to readers in the field of clinical neuroscience and movement disorders. The identification and characterization of dynamic networks that are differentially affected by dopaminergic medication in patients with Parkinson's disease is an important contribution to our understanding of physiologic and pathologic brain activities. The used methods provide the potential to uncover spectral, local, and temporal properties of dynamic neural systems. Overall, the data are properly analyzed, although many aspects of reporting the results could be worked out better.
(This preprint has been reviewed by eLife. We include the public reviews from the reviewers here; the authors also receive private feedback with suggested changes to the manuscript. Reviewer #1 and Reviewer #2 agreed to share their names with the authors.)
This article has been Reviewed by the following groups
Discuss this preprint
Start a discussion What are Sciety discussions?Listed in
- Evaluated articles (eLife)
Abstract
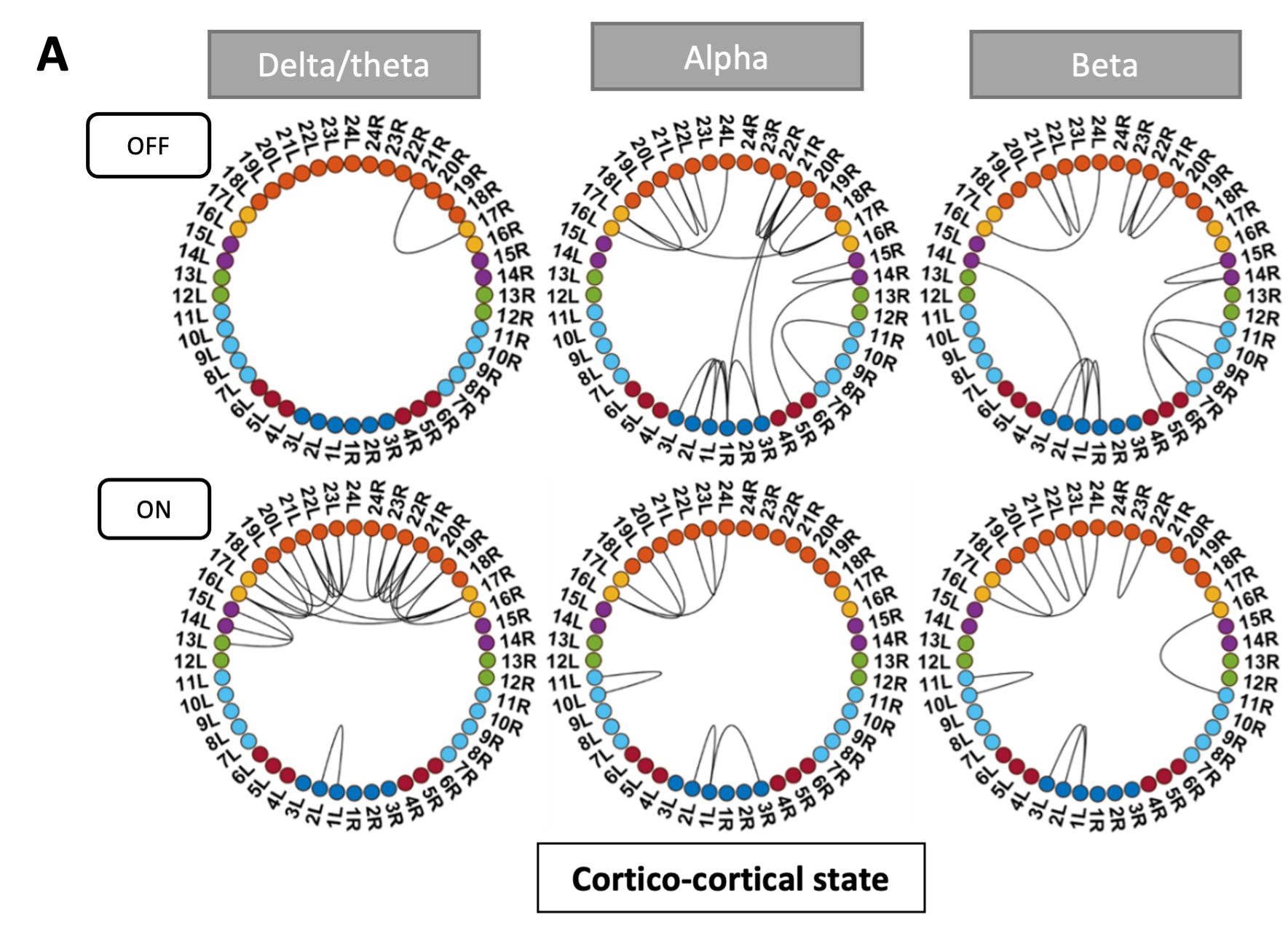
Pathological oscillations including elevated beta activity in the subthalamic nucleus (STN) and between STN and cortical areas are a hallmark of neural activity in Parkinson’s disease (PD). Oscillations also play an important role in normal physiological processes and serve distinct functional roles at different points in time. We characterised the effect of dopaminergic medication on oscillatory whole-brain networks in PD in a time-resolved manner by employing a hidden Markov model on combined STN local field potentials and magnetoencephalography (MEG) recordings from 17 PD patients. Dopaminergic medication led to coherence within the medial and orbitofrontal cortex in the delta/theta frequency range. This is in line with known side effects of dopamine treatment such as deteriorated executive functions in PD. In addition, dopamine caused the beta band activity to switch from an STN-mediated motor network to a frontoparietal-mediated one. In contrast, dopamine did not modify local STN–STN coherence in PD. STN–STN synchrony emerged both on and off medication. By providing electrophysiological evidence for the differential effects of dopaminergic medication on the discovered networks, our findings open further avenues for electrical and pharmacological interventions in PD.
Article activity feed
-

-

Author Response:
Reviewer #1:
The largest concern with the manuscript is its use of resting-state recordings in Parkinson's Disease patients on and off levodopa, which the authors interpret as indicative of changes in dopamine levels in the brain but not indicative of altered movement and other neural functions. For example, when patients are off medication, their UPDRS scores are elevated, indicating they likely have spontaneous movements or motor abnormalities that will likely produce changed activations in MEG and LFP during "rest". Authors must address whether it is possible to study a true "resting state" in unmedicated patients with severe PD. At minimum this concern must be discussed in the manuscript.
We agree that Parkinson’s disease can lead to unwanted movements such as tremor as well as hyperkinesias. This would of …
Author Response:
Reviewer #1:
The largest concern with the manuscript is its use of resting-state recordings in Parkinson's Disease patients on and off levodopa, which the authors interpret as indicative of changes in dopamine levels in the brain but not indicative of altered movement and other neural functions. For example, when patients are off medication, their UPDRS scores are elevated, indicating they likely have spontaneous movements or motor abnormalities that will likely produce changed activations in MEG and LFP during "rest". Authors must address whether it is possible to study a true "resting state" in unmedicated patients with severe PD. At minimum this concern must be discussed in the manuscript.
We agree that Parkinson’s disease can lead to unwanted movements such as tremor as well as hyperkinesias. This would of course be a deviation from a resting state in healthy subjects. However, such movements are part of the disease and occur unwillingly. The main tremor in Parkinson’s disease is a rest tremor and - as the name already suggests – it occurs while not doing anything. Therefore, such movements can arguably be considered part of the resting state of Parkinson’s disease. Resting state activity with and without medication is therefore still representative for changes in brain activity in Parkinson’s patients and indicative of alterations due to medication.
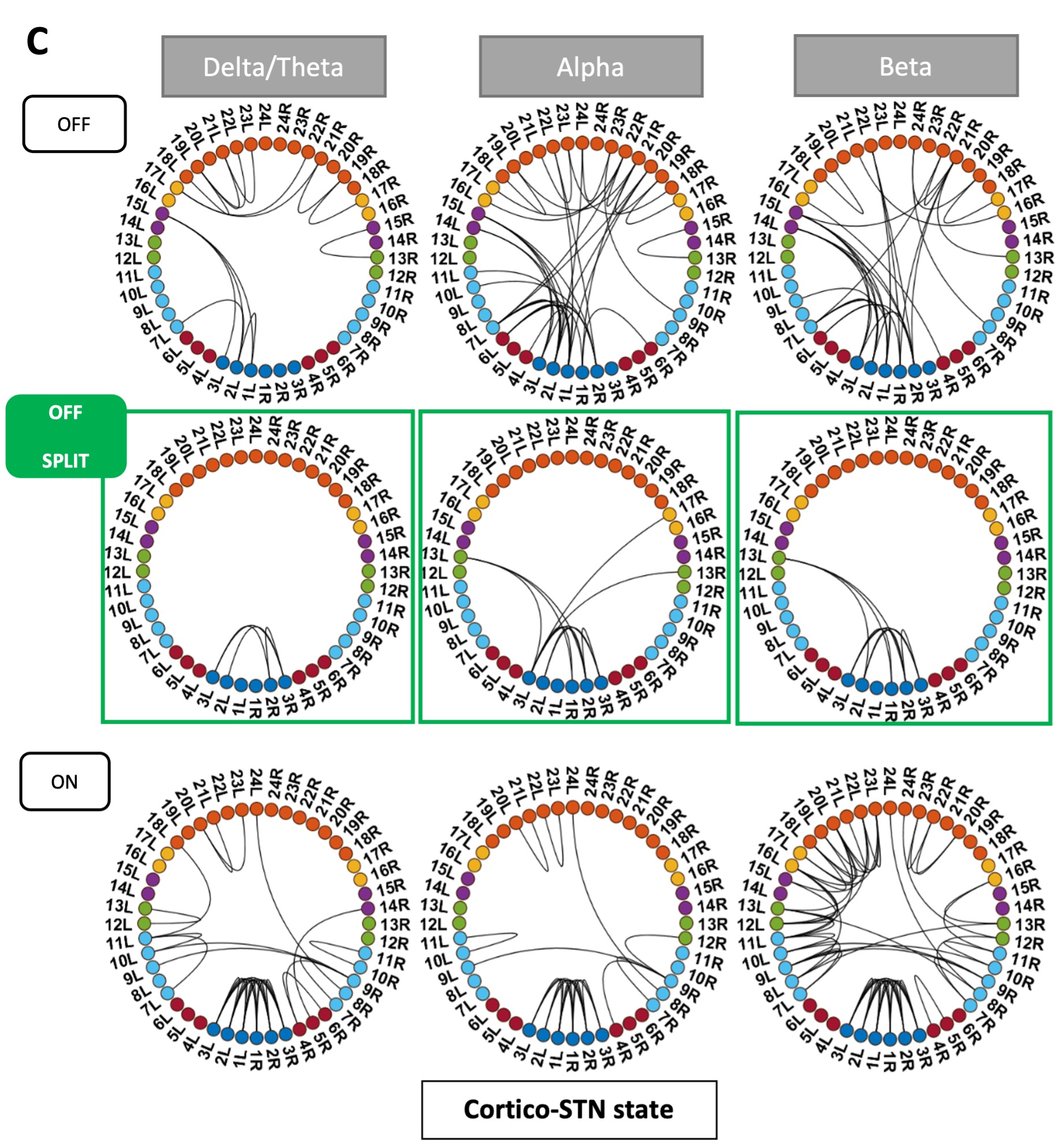
To further investigate the effect of movement in our patients, we subdivided the UPDRS part 3 score into tremor and non-tremor subscores. For the tremor subscore we took the mean of item 15 and 17 of the UPDRS, whereas for the non-tremor subscore items 1, 2, 3, 9, 10, 12, 13, and 14 were averaged. Following Spiegel et al., 2007, we classified patients as akinetic-rigid (non-tremor score at least twice the tremor score), tremor-dominant (tremor score at least twice as large as the non-tremor score), and mixed type (for the remaining scores). Of the 17 patients, 1 was tremor dominant and 1 was classified as mixed type (his/her non-tremor score was greater than tremor score). None of our patients exhibited hyperkinesias during the recording. To exclude that our results are driven by tremor-related movement, we re-ran the HMM without the tremor-dominant and the mixed-type patient (see Figure R1 response letter).
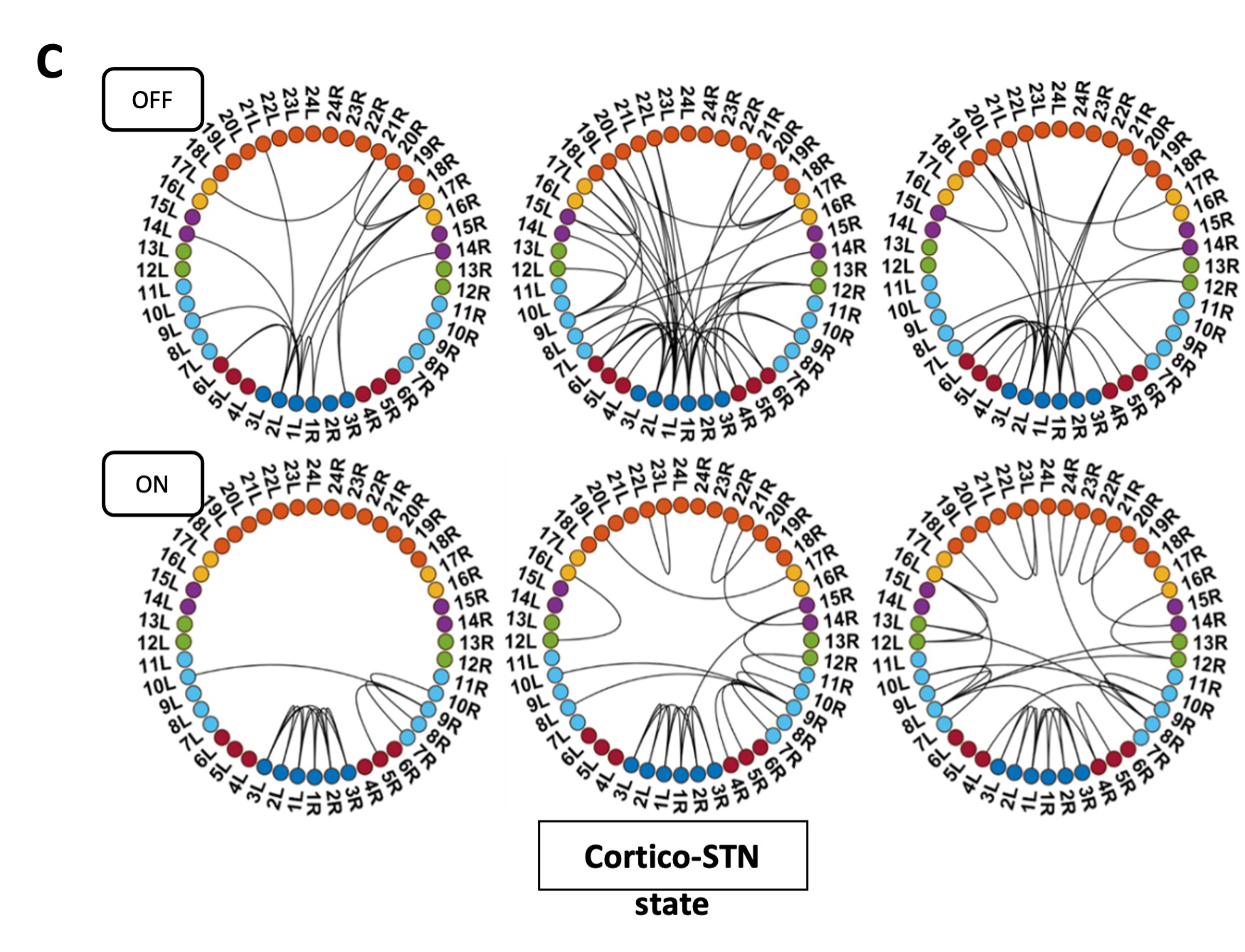
ON medication results for all HMM states remained the same. OFF medication results for the Ctx-Ctx and STN-STN state remained the same as well. The Ctx-STN state OFF medication was split into two states: Sensorimotor-STN connectivity was captured in one state and all other types of Ctx-STN connections were captured in another state (see Figure 1 response letter. The important point is that the biological conclusions stand across these solutions. Regardless, both with and without the two subjects a stable covariance matrix entailing sensorimotor-STN connectivity was determined, which is the main finding for the Ctx-STN state OFF medication.
We therefore discuss this issue now within the limitation section (page 20):
“Both motor impairment and motor improvement can cause movement during the resting state in PD. While such movement is a deviation from a resting state in healthy subjects, such movements are part of the disease and occur unwillingly. Therefore, such movements can arguably be considered part of the resting state of Parkinson’s disease. None of the patients in our cohort experienced hyperkinesia during the recording. All patients except for two were of the akinetic-rigid subtype. We verified that tremor movement is not driving our results. Recalculating the HMM states without these 2 subjects, even though it slightly changed some particular aspects of the HMM solution did not materially affect the conclusions.”
Figure R1: States obtained after removing one tremor dominant and one mixed type patient from analysis. Panel C shows the split OFF medication cortico-STN state. Most of the cortico-STN connectivity is captured by the state shown in the top row (Figure 1 C OFF). Only the motor-STN connectivity in the alpha and beta band (along with a medial frontal-STN connection in the alpha band) is captured separately by the states labeled “OFF Split” (Figure 1 C OFF SPLIT).
This reviewer was unclear on why increased "communication" in the medial OFC in delta and theta was interpreted as a pathological state indicating deteriorated frontal executive function. Given that the authors provide no evidence of poor executive function in the patients studied, the authors must at least provide evidence from other studies linking this feature with impaired executive function.
If we understand the comment correctly it refers to the statement in the abstract “Dopaminergic medication led to communication within the medial and orbitofrontal cortex in the delta/theta frequency range. This is in line with deteriorated frontal executive functioning as a side effect of dopamine treatment in Parkinson’s disease”
This statement is based on the dopamine overdose hypothesis reported in the Parkinson’s disease (PD) literature (Cools 2001; Kelly et al. 2009; MacDonald and Monchi 2011; Vaillancourt et al. 2013). We have elaborated upon the dopamine overdose hypothesis in the discussion on page 16. In short, dopaminergic neurons are primarily lost from the substantia nigra in PD, which causes a higher dopamine depletion in the dorsal striatal circuitry than within the ventral striatal circuits (Kelly et al. 2009; MacDonald and Monchi 2011). Thus, dopaminergic medication to treat the PD motor symptoms leads to increased dopamine levels in the ventral striatal circuits including frontal cortical activity, which can potentially explain the cognitive deficits observed in PD (Shohamy et al. 2005; George et al. 2013). We adjusted the abstract to read:
“Dopaminergic medication led to coherence within the medial and orbitofrontal cortex in the delta/theta frequency range. This is in line with known side effects of dopamine treatment such as deteriorated executive functions in Parkinson’s disease.”
In this article, authors repeatedly state their method allows them to delineate between pathological and physiological connectivity, but they don't explain how dynamical systems and discrete-state stochasticity support that goal.
To recapitulate, the HMM divides a continuous time series into discrete states. Each state is a time-delay embedded covariance matrix reflecting the underlying connectivity between brain regions as well as the specific temporal dynamics in the data when such state is active. See Packard et al., (1980) for details about how a time-delay embedding characterises a linear dynamical system.
Please note that the HMM was used as a data-driven, descriptive approach without explicitly assuming any a-priori relationship with pathological or physiological states. The relation between biology and the HMM states, thus, purely emerged from the data; i.e. is empirical. What we claim in this work is simply that the features captured by the HMM hold some relation with the physiology even though the estimation of the HMM was completely unsupervised (i.e. blind to the studied conditions). We have added this point also to the limitations of the study on page 19 and the following to the introduction to guide the reader more intuitively (page 4):
“To allow the system to dynamically evolve, we use time delay embedding. Theoretically, delay embedding can reveal the state space of the underlying dynamical system (Packard et al., 1980). Thus, by delay-embedding PD time series OFF and ON medication we uncover the differential effects of a neurotransmitter such as dopamine on underlying whole brain connectivity.”
Reviewer #2:
Sharma et al. investigated the effect of dopaminergic medication on brain networks in patients with Parkinson's disease combining local field potential recordings from the subthalamic nucleus and magnetencephalography during rest. They aim to characterize both physiological and pathological spectral connectivity.
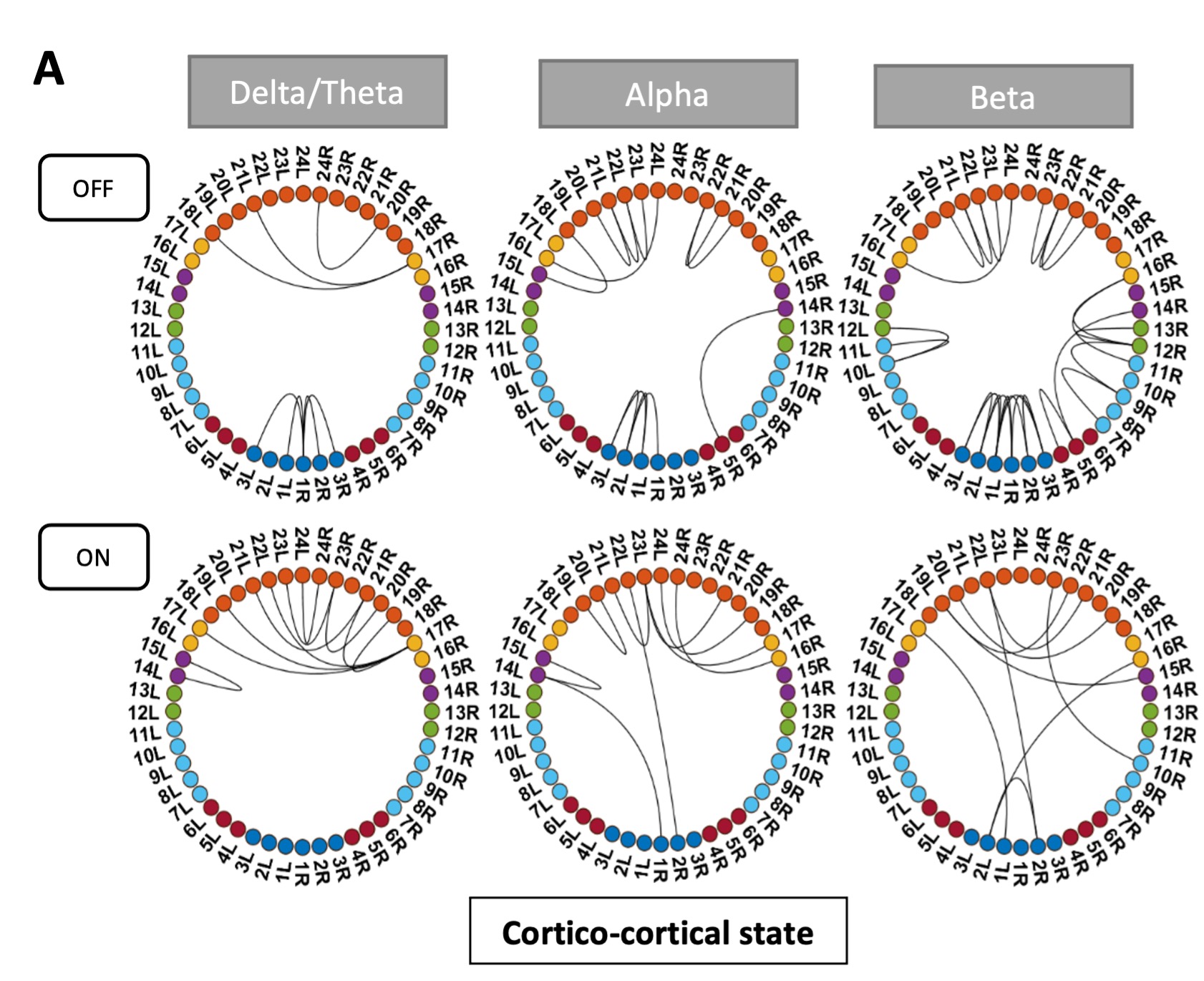
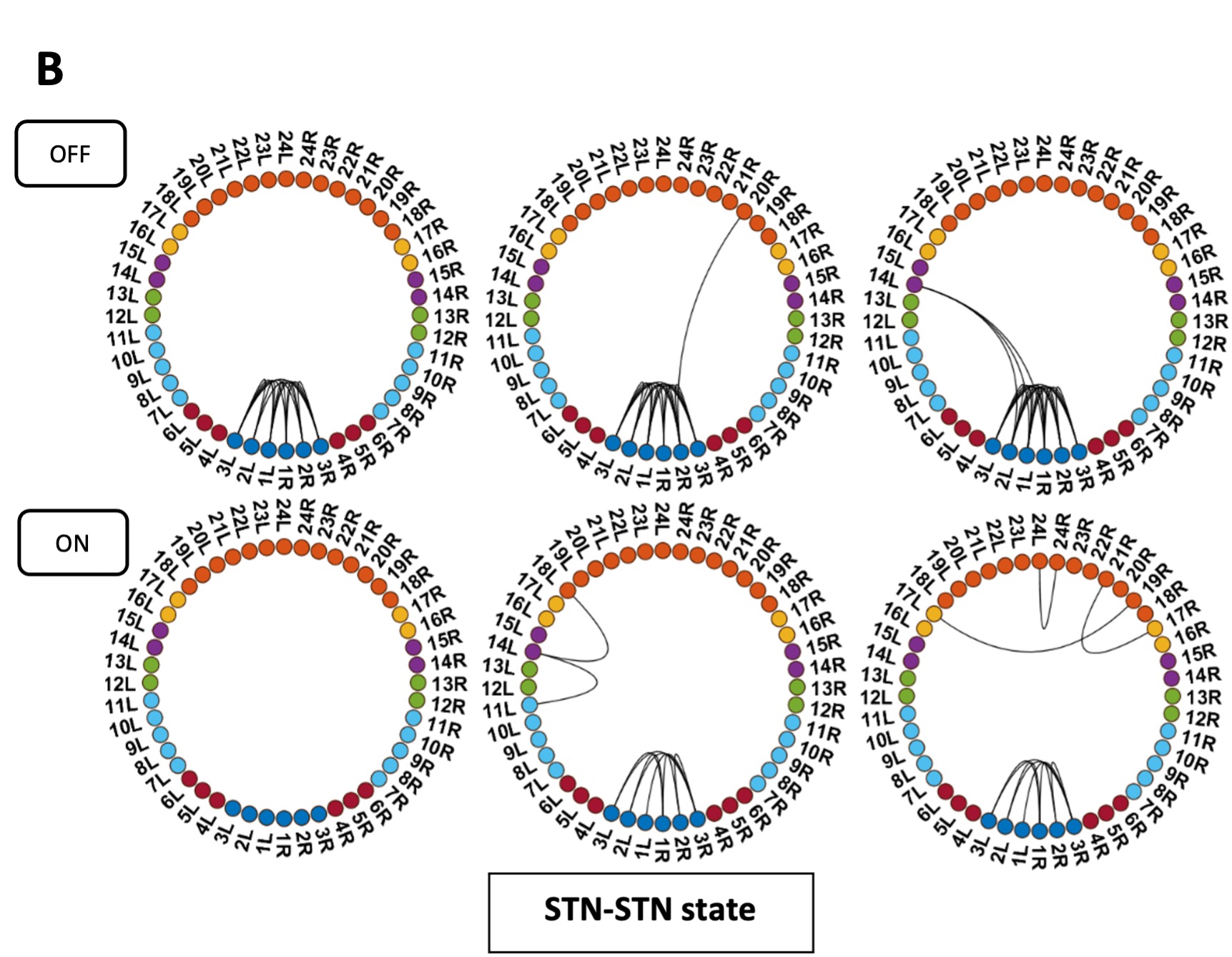
They identified three networks, or brain states, that are differentially affected by medication. Under medication, the first state (termed hyperdopaminergic state) is characterized by increased connectivity of frontal areas, supposedly responsible for deteriorated frontal executive function as a side effect of medical treatment. In the second state (communication state), dopaminergic treatment largely disrupts cortico-STN connectivity, leaving only selected pathways communicating. This is in line with current models that propose that alleviation of motor symptoms relates to the disruption of pathological pathways. The local state, characterized by STN-STN oscillatory activities, is less affected by dopaminergic treatment.
The authors utilize sophisticated methods with the potential to uncover the dynamics of activities within different brain network, which opens the avenue to investigate how the brain switches between different states, and how these states are characterized in terms of spectral, local, and temporal properties. The conclusions of this paper are mostly well supported by data, but some aspects, mainly about the presentation of the results, remain:
We would like to thank the reviewer for his succinct and clear understanding of our work.
- The presentation of the results is suboptimal and needs improvement to increase readers' comprehension. At some points this section seems rather unstructured, some results are presented multiple times, and some passages already include points rather suitable for the discussion, which adds too much information for the results section.
We have removed repetitions in the results sections and removed the rather lengthy introductory parts of each subsection. Moreover, we have now moved all parts, which were already an interpretation of our findings to the discussion.
- It is intriguing that the hyperdopaminergic state is not only identified under medication but also in the off-state. This is intriguing, especially with the results on the temporal properties of states showing that the time of the hyperdopaminergic state is unaffected by medication. When such a state can be identified even in the absence of levodopa, is it really optimal to call it "hyperdopaminergic"? Do the results not rather suggest that the identified network is active both off and on medication, while during the latter state its' activities are modulated in a way that could relate to side effects?
The reviewer’s interpretations of the results pertaining to the hyper-dopaminergic state are correct. The states had been named post-hoc as explained in the results section. The hyper-dopaminergic state’s name derived from it showing the overdosing effects of dopamine. Of course, these results are only visible on medication. But off medication, this state also exists without exhibiting the effects of excess dopamine. To avoid confusion or misinterpretation of the findings and also following the relevant comment by reviewer 1, we renamed all states to be more descriptive:
Hyperdopaminergic > Cortico-cortical state
Communication > Cortico-STN state
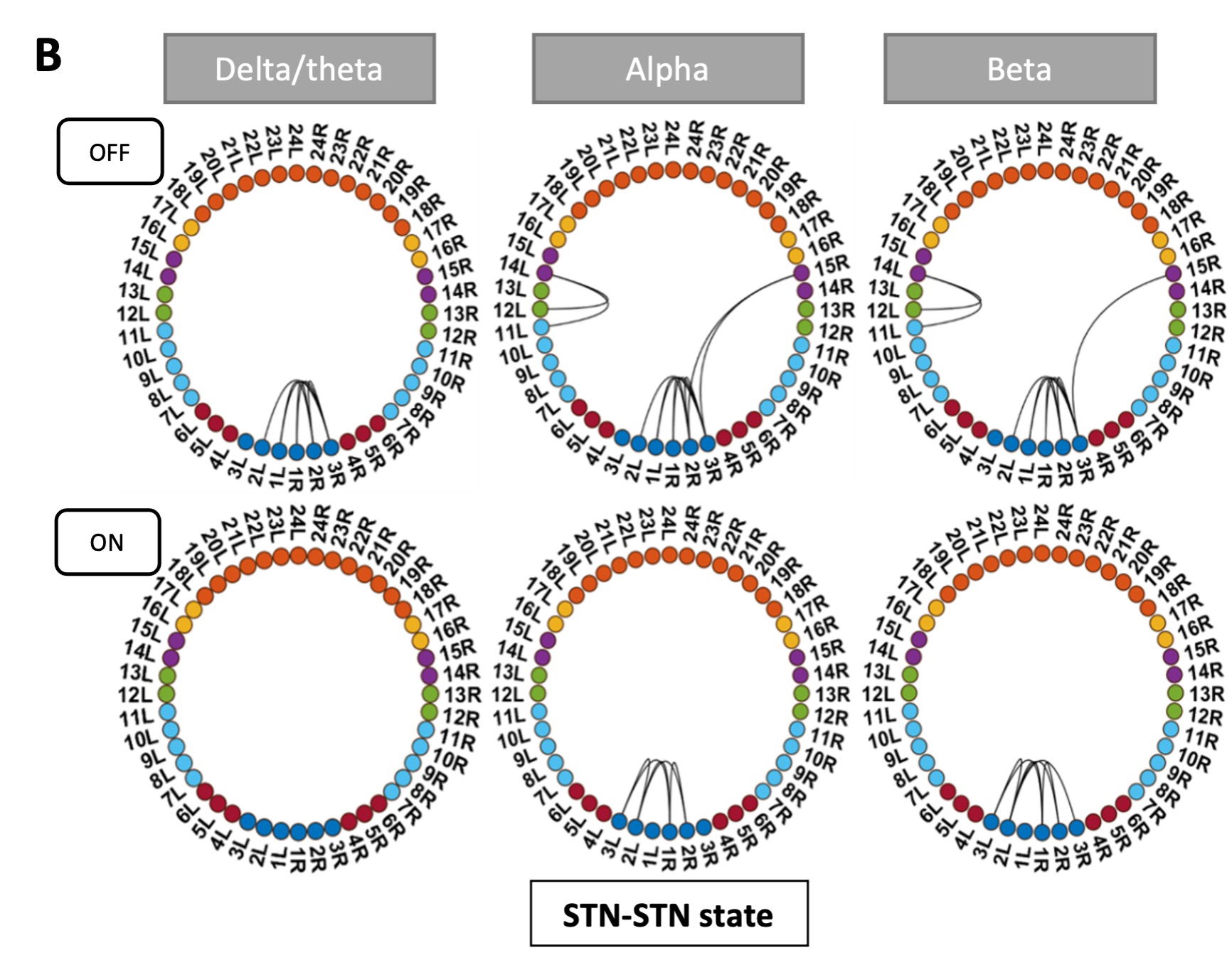
Local > STN-STN state.
- Some conclusions need to be improved/more elaborated. For example, the coherence of bilateral STN-STN did not change between medication off and on the state. Yet it is argued that a) "Since synchrony limits information transfer (Cruz et al. 2009; Cagnan, Duff, and Brown 2015; Holt et al. 2019) , local oscillations are a potential mechanism to prevent excessive communication with the cortex" (line 436) and b) "Another possibility is that a loss of cortical afferents causes local basal ganglia oscillations to become more pronounced" (line 438). Can these conclusions really be drawn if the local oscillations did not change in the first place?
We apologize for the unclear description. Our conclusion was based on the following results:
a) We state that STN-STN connectivity as measured by the magnitude of STN-STN coherence does not change OFF vs ON medication in the Cortico-STN state. This result is obtained using inter-medication analysis.
b) But ON medication, STN-STN coherence in the Cortico-STN state was significantly different from mean coherence within the ON condition. These results are obtained using intra-medication analysis.
Based on this, we conclude that in the Cortico-STN state, although OFF vs ON medication the magnitude of STN-STN coherence was unchanged, the STN-STN coherence was significantly different from mean coherence in the ON medication condition. The emergence of synchronous STN-STN activity may limit information exchange between STN and cortex ON medication.
An alternative explanation for these findings might be a mechanism preventing connectivity between cortex and the STN ON medication. This missing interaction between STN and cortex might cause STN-STN oscillations to increase compared to the mean coherence within the ON state. Unfortunately, we cannot test such causal influences with our analysis.
We have added the following discussion to the manuscript on page 17 in order to improve the exposition:
“Bilateral STN–STN coherence in the alpha and beta band did not change in the cortico-STN state ON versus OFF medication (InterMed analysis). However, STN-STN coherence was significantly higher than the mean level ON medication (IntraMed analysis). Since synchrony limits information transfer (Cruz et al. 2009; Cagnan, Duff, and Brown 2015; Holt et al. 2019), the high coherence within the STN ON medication could prevent communication with the cortex. A different explanation would be that a loss of cortical afferents leads to increased local STN coherence. The causal nature of the cortico-basal ganglia interaction is an endeavour for future research.”
Reviewer #3:
In PD, pathological neuronal activity along the cortico-basal ganglia network notably consists in the emergence of abnormal synchronized oscillatory activity. Nevertheless, synchronous oscillatory activity is not necessarily pathological and also serve crucial cognitive functions in the brain. Moreover, the effect of dopaminergic medication on oscillatory network connectivity occurring in PD are still poorly understood. To clarify these issues, Sharma and colleagues simultaneously-recorded MEG-STN LFP signals in PD patients and characterized the effect of dopamine (ON and OFF dopaminergic medication) on oscillatory whole-brain networks (including the STN) in a time-resolved manner. Here, they identified three physiologically interpretable spectral connectivity patterns and found that cortico-cortical, cortico-STN, and STN-STN networks were differentially modulated by dopaminergic medication.
Strengths:
- Both the methodological and experimental approaches used are thoughtful and rigorous.
a) The use of an innovative data-driven machine learning approach (by employing a hidden Markov model), rather than hand-crafted analyses, to identify physiologically interpretable spectral connectivity patterns (i.e., distinct networks/states) is undeniably an added value. In doing so, the results are not biased by the human expertise and subjectivity, which make them even more solid.
b) So far, the recurrent oscillatory patterns of transient network connectivity within and between the cortex and the STN reported in PD was evaluated/assessed to specific cortico-STN spectral connectivity. Conversely, whole-brain MEG studies in PD patients did not account for cortico-STN and STN-STN connectivity. Here, the authors studied, for the first time, the whole-brain connectivity including the STN (whole brain-STN approach) and therefore provide new evidence of the brain connectivity reported in PD, as well as new information regarding the effect of dopaminergic medication on the recurrent oscillatory patterns of transient network connectivity within and between the cortex and the STN reported in PD.
- Studying the temporal properties of the recurrent oscillatory patterns of transient network connectivity both ON and OFF medication is extremely important and provide interesting and crucial information in order to delineated pathological versus physiologically-relevant spectral brain connectivity in PD.
We would like to thank the reviewer for their valuable feedback and correct interpretation of our manuscript.
Weaknesses:
- In this study, the authors implied that the ON dopaminergic medication state correspond to a physiological state. However, as correctly mentioned in the limitations of the study, they did not have (for obvious reasons) a control/healthy group. Moreover, no one can exclude the emergence of compensatory and/or plasticity mechanisms in the brain of the PD patients related to the duration of the disease and/or the history of the chronic dopamine-replacement therapy (DRT). Duration of the disease and DRT history should be therefore considered when characterizing the recurrent oscillatory patterns of transient network connectivity within and between the cortex and the STN reported in PD, as well as when examining the effect of the dopaminergic medication on the functioning of these specific networks.
We would like to thank the reviewer for pointing this out. We regressed duration of disease (year of measurement – year of onset) on the temporal properties of the HMM states. We found no relationship between any of the temporal properties and disease duration. Similarly, we regressed levodopa equivalent dosage for each subject on the temporal properties and found no relationship. We now discuss this point in the manuscript (page 20):
“A further potential influencing factor might be the disease duration and the amount of dopamine patients are receiving. Both factors were not significantly related to the temporal properties of the states.”
- Here, the authors recorded LFPs in the STN activity. LFP represents sub-threshold (e.g., synaptic input) activity at best (Buzsaki et al., 2012; Logothetis, 2003). Recent studies demonstrated that mono-polar, but also bi-polar, BG LFPs are largely contaminated by volume conductance of cortical electroencephalogram (EEG) activity even when re-referenced (Lalla et al., 2017; Marmor et al., 2017). Therefore, it is likely that STN LFPs do not accurately reflect local cellular activity. In this study, the authors examined and measured coherence between cortical areas and STN. However, they cannot guarantee that STN signals were not contaminated by volume conducted signals from the cortex.
We appreciate this concern and thank the reviewer for bringing it up. Marmor et al. (2017) investigated this on humans and is therefore most closely related to our research. They find that re-referenced STN recordings are not contaminated by cortical signals. Furthermore, the data in Lalla et al. (2017) is based on recordings in rats, making a direct transfer to human STN recordings problematic due to the different brain sizes. Since we re-referenced our LFP signals as recommended in the Marmor paper, we think that contamination due to cortical signals is relatively minor; see Litvak et al. (2011), Hirschmann et al. (2013), and Neumann et al. (2016) for additional references supporting this. That being said, we now discuss this potential issue in the paper on page 20.
“Lastly, we recorded LFPs from within the STN –an established recording procedure during the implantation of DBS electrodes in various neurological and psychiatric diseases. Although for Parkinson patients results on beta and tremor activity within the STN have been reproduced by different groups (Reck et al. 2010, Litvak et al. 2011, Florin et al. 2013, Hirschmann et al. 2013, Neumann et al. 2016), it is still not fully clear whether these LFP signals are contaminated by volume-conducted cortical activity. However, while volume conduction seems to be a larger problem in rodents even after re-referencing the LFP signal (Lalla et al. 2017), the same was not found in humans (Marmor et al. 2017).”
- The methods and data processing are rigorous but also very sophisticated which make the perception of the results in terms of oscillatory activity and neural synchronization difficult.
To aid intuition on how to interpret the result in light of the methods used, one can compare the analysis pipeline to a windowing approach. In a more standard approach, windows of different time length can be defined for different epochs within the time series and for each window coherence and connectivity can be determined. The difference in our approach is that we used an unsupervised learning algorithm to select windows of varying length based on recurring patterns of whole brain network activity. Within those defined windows we then determine the oscillatory properties via coherence and power – which is the same as one would do in a classical analysis. We have added an explanation of the concept of “oscillatory activity” within our framework to the introduction (page 2 footnote):
“For the purpose of our paper, we refer to oscillatory activity or oscillations as recurrent, but transient frequency–specific patterns of network activity, even though the underlying patterns can be composed of either sustained rhythmic activity, neural bursting, or both (Quinn et al. 2019).”
Moreover, we provide a more intuitive explanation of the analysis within the first section of the results (page 4):
“Using an HMM, we identified recurrent patterns of transient network connectivity between the cortex and the STN, which we henceforth refer to as an ‘HMM state’. In comparison to classic sliding-window analysis, an HMM solution can be thought of as a data-driven estimation of time windows of variable length (within which a particular HMM state was active): once we know the time windows when a particular state is active, we compute coherence between different pairs of regions for each of these recurrent states.”
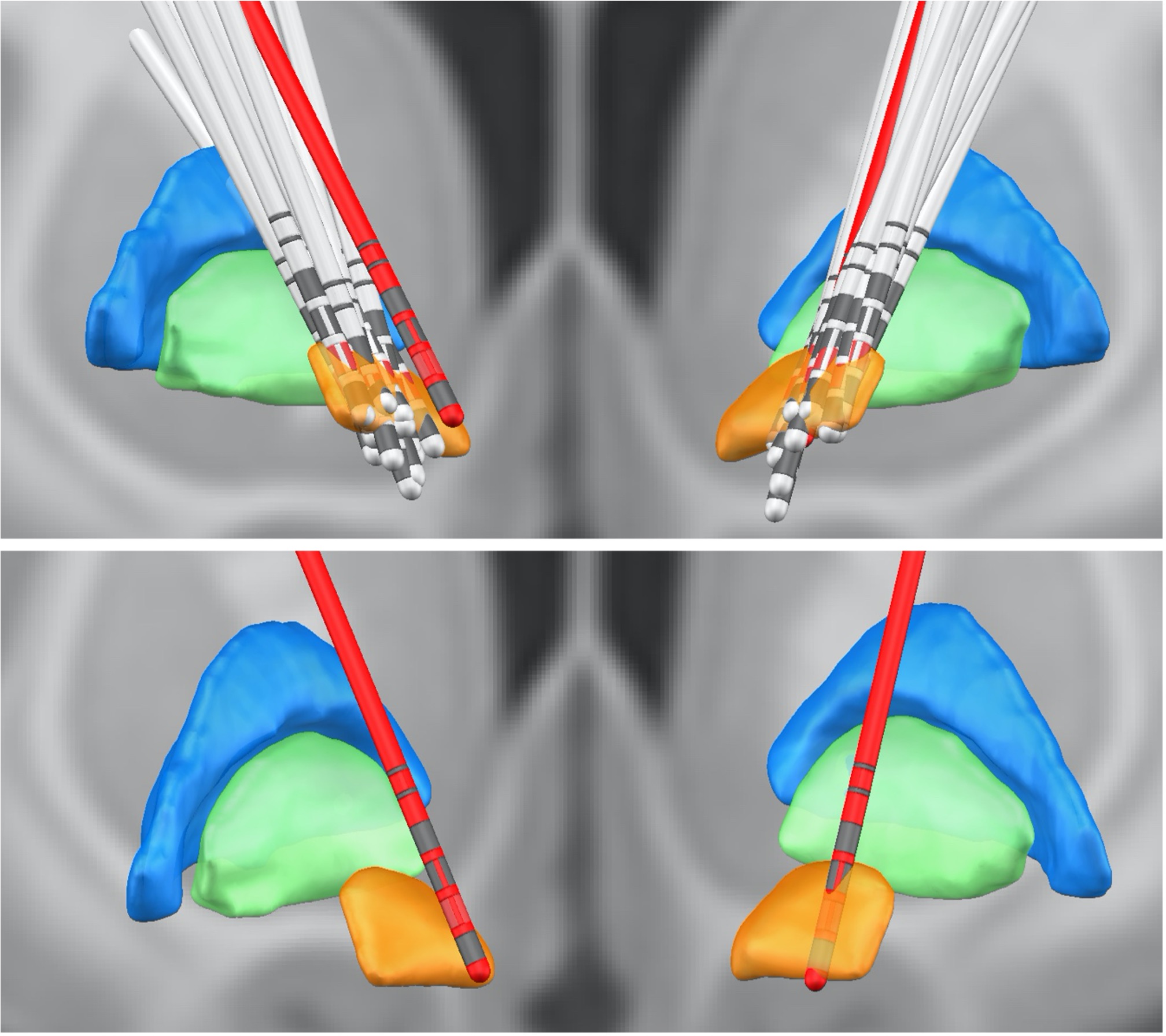
- Previous studies have shown that abnormal oscillations within the STN of PD patients are limited to its dorsolateral/motor region, thus dividing the STN into a dorsolateral oscillatory/motor region and ventromedial non-oscillatory/non-motor region (Kuhn et al. 2005; Moran et al. 2008; Zaidel et al. 2009, 2010; Seifreid et al. 2012; Lourens et al. 2013, Deffains et al., 2014). However, the authors do not provide clear information about the location of the LFP recordings within the STN.
We selected the electrode contacts based on intraoperative microelectrode recordings (for details, see page 23). The first directional recording height after the entry into the STN was selected to obtain the three directional LFP recordings from the respective hemisphere. This practice has been proven to improve target location (Kochanski et al., 2019; Krauss et al., 2021). The common target area for DBS surgery is the dorsolateral STN. To confirm that the electrodes were actually located within this part of the STN, we now reconstructed the DBS location with Lead-DBS (Horn et al. 2019). All electrodes – except for one – were located within the dorsolateral STN (see figure 7 of the manuscript). To exclude that our results were driven by outlier, we reanalysed our data without this patient. No change in the overall connectivity pattern was observed (see figure R3 of the response letter).
Figure R2: Lead DBS reconstruction of the location of electrodes in the STN for different subjects. The red electrodes have not been placed properly in the STN. The contacts marked in red represent the directional contacts from which the data was used for analysis.
Figure R3: HMM states obtained after running the analysis without the subject with the electrode outside the STN.
References:
Buzsáki G, Anastassiou CA, Koch C. The origin of extracellular fields and currents-EEG, ECoG, LFP and spikes. Nat Rev Neurosci 2012; 13: 407–20.
Cagnan H, Duff EP, Brown P. The relative phases of basal ganglia activities dynamically shape effective connectivity in Parkinson’s disease. Brain 2015; 138: 1667–78.
Cools R. Enhanced or impaired cognitive function in Parkinson’s disease as a function of dopaminergic medication and task demands. Cereb Cortex 2001; 11: 1136–43.
Cruz A V., Mallet N, Magill PJ, Brown P, Averbeck BB. Effects of dopamine depletion on network entropy in the external globus pallidus. J Neurophysiol 2009; 102: 1092–102.
Florin E, Erasmi R, Reck C, Maarouf M, Schnitzler A, Fink GR, et al. Does increased gamma activity in patients suffering from Parkinson’s disease counteract the movement inhibiting beta activity? Neuroscience 2013; 237: 42–50.
George JS, Strunk J, Mak-Mccully R, Houser M, Poizner H, Aron AR. Dopaminergic therapy in Parkinson’s disease decreases cortical beta band coherence in the resting state and increases cortical beta band power during executive control. NeuroImage Clin 2013; 3: 261–70.
Hirschmann J, Özkurt TE, Butz M, Homburger M, Elben S, Hartmann CJ, et al. Differential modulation of STN-cortical and cortico-muscular coherence by movement and levodopa in Parkinson’s disease. Neuroimage 2013; 68: 203–13.
Holt AB, Kormann E, Gulberti A, Pötter-Nerger M, McNamara CG, Cagnan H, et al. Phase-dependent suppression of beta oscillations in parkinson’s disease patients. J Neurosci 2019; 39: 1119–34.
Horn A, Li N, Dembek TA, Kappel A, Boulay C, Ewert S, et al. Lead-DBS v2: Towards a comprehensive pipeline for deep brain stimulation imaging. Neuroimage 2019; 184: 293–316.
Kelly C, De Zubicaray G, Di Martino A, Copland DA, Reiss PT, Klein DF, et al. L-dopa modulates functional connectivity in striatal cognitive and motor networks: A double-blind placebo-controlled study. J Neurosci 2009; 29: 7364–78.
Kochanski RB, Bus S, Brahimaj B, Borghei A, Kraimer KL, Keppetipola KM, et al. The impact of microelectrode recording on lead location in deep brain stimulation for the treatment of movement disorders. World Neurosurg 2019; 132: e487–95.
Krauss P, Oertel MF, Baumann-Vogel H, Imbach L, Baumann CR, Sarnthein J, et al. Intraoperative neurophysiologic assessment in deep brain stimulation surgery and its impact on lead placement. J Neurol Surgery, Part A Cent Eur Neurosurg 2021; 82: 18–26.
Lalla L, Rueda Orozco PE, Jurado-Parras MT, Brovelli A, Robbe D. Local or not local: Investigating the nature of striatal theta oscillations in behaving rats. eNeuro 2017; 4: 128–45.
Litvak V, Jha A, Eusebio A, Oostenveld R, Foltynie T, Limousin P, et al. Resting oscillatory cortico-subthalamic connectivity in patients with Parkinson’s disease. Brain 2011; 134: 359–74.
MacDonald PA, MacDonald AA, Seergobin KN, Tamjeedi R, Ganjavi H, Provost JS, et al. The effect of dopamine therapy on ventral and dorsal striatum-mediated cognition in Parkinson’s disease: Support from functional MRI. Brain 2011; 134: 1447–63.
MacDonald PA, Monchi O. Differential effects of dopaminergic therapies on dorsal and ventral striatum in Parkinson’s disease: Implications for cognitive function. Parkinsons Dis 2011; 2011: 1–18.
Marmor O, Valsky D, Joshua M, Bick AS, Arkadir D, Tamir I, et al. Local vs. volume conductance activity of field potentials in the human subthalamic nucleus. J Neurophysiol 2017; 117: 2140–51.
Neumann WJ, Degen K, Schneider GH, Brücke C, Huebl J, Brown P, et al. Subthalamic synchronized oscillatory activity correlates with motor impairment in patients with Parkinson’s disease. Mov Disord 2016; 31: 1748–51.
Packard NH, Crutchfield JP, Farmer JD, Shaw RS. Geometry from a time series. Phys Rev Lett 1980; 45: 712–6.
Quinn AJ, van Ede F, Brookes MJ, Heideman SG, Nowak M, Seedat ZA, et al. Unpacking Transient Event Dynamics in Electrophysiological Power Spectra. Brain Topogr 2019; 32: 1020–34.
Reck C, Himmel M, Florin E, Maarouf M, Sturm V, Wojtecki L, et al. Coherence analysis of local field potentials in the subthalamic nucleus: Differences in parkinsonian rest and postural tremor. Eur J Neurosci 2010; 32: 1202–14.
Shohamy D, Myers CE, Grossman S, Sage J, Gluck MA. The role of dopamine in cognitive sequence learning: Evidence from Parkinson’s disease. Behav Brain Res 2005; 156: 191–9.
Spiegel J, Hellwig D, Samnick S, Jost W, Möllers MO, Fassbender K, et al. Striatal FP-CIT uptake differs in the subtypes of early Parkinson’s disease. J Neural Transm 2007; 114: 331–5.
Vaillancourt DE, Schonfeld D, Kwak Y, Bohnen NI, Seidler R. Dopamine overdose hypothesis: Evidence and clinical implications. Mov Disord 2013; 28: 1920–9.
-

Reviewer #3 (Public Review):
In PD, pathological neuronal activity along the cortico-basal ganglia network notably consists in the emergence of abnormal synchronized oscillatory activity. Nevertheless, synchronous oscillatory activity is not necessarily pathological and also serve crucial cognitive functions in the brain. Moreover, the effect of dopaminergic medication on oscillatory network connectivity occurring in PD are still poorly understood. To clarify these issues, Sharma and colleagues simultaneously-recorded MEG-STN LFP signals in PD patients and characterized the effect of dopamine (ON and OFF dopaminergic medication) on oscillatory whole-brain networks (including the STN) in a time-resolved manner. Here, they identified three physiologically interpretable spectral connectivity patterns and found that cortico-cortical, …
Reviewer #3 (Public Review):
In PD, pathological neuronal activity along the cortico-basal ganglia network notably consists in the emergence of abnormal synchronized oscillatory activity. Nevertheless, synchronous oscillatory activity is not necessarily pathological and also serve crucial cognitive functions in the brain. Moreover, the effect of dopaminergic medication on oscillatory network connectivity occurring in PD are still poorly understood. To clarify these issues, Sharma and colleagues simultaneously-recorded MEG-STN LFP signals in PD patients and characterized the effect of dopamine (ON and OFF dopaminergic medication) on oscillatory whole-brain networks (including the STN) in a time-resolved manner. Here, they identified three physiologically interpretable spectral connectivity patterns and found that cortico-cortical, cortico-STN, and STN-STN networks were differentially modulated by dopaminergic medication.
Strengths:
- Both the methodological and experimental approaches used are thoughtful and rigorous.
a) The use of an innovative data-driven machine learning approach (by employing a hidden Markov model), rather than hand-crafted analyses, to identify physiologically interpretable spectral connectivity patterns (i.e., distinct networks/states) is undeniably an added value. In doing so, the results are not biased by the human expertise and subjectivity, which make them even more solid.
b) So far, the recurrent oscillatory patterns of transient network connectivity within and between the cortex and the STN reported in PD was evaluated/assessed to specific cortico-STN spectral connectivity. Conversely, whole-brain MEG studies in PD patients did not account for cortico-STN and STN-STN connectivity. Here, the authors studied, for the first time, the whole-brain connectivity including the STN (whole brain-STN approach) and therefore provide new evidence of the brain connectivity reported in PD, as well as new information regarding the effect of dopaminergic medication on the recurrent oscillatory patterns of transient network connectivity within and between the cortex and the STN reported in PD.
- Studying the temporal properties of the recurrent oscillatory patterns of transient network connectivity both ON and OFF medication is extremely important and provide interesting and crucial information in order to delineated pathological versus physiologically-relevant spectral brain connectivity in PD.
Weaknesses:
In this study, the authors implied that the ON dopaminergic medication state correspond to a physiological state. However, as correctly mentioned in the limitations of the study, they did not have (for obvious reasons) a control/healthy group. Moreover, no one can exclude the emergence of compensatory and/or plasticity mechanisms in the brain of the PD patients related to the duration of the disease and/or the history of the chronic dopamine-replacement therapy (DRT). Duration of the disease and DRT history should be therefore considered when characterizing the recurrent oscillatory patterns of transient network connectivity within and between the cortex and the STN reported in PD, as well as when examining the effect of the dopaminergic medication on the functioning of these specific networks.
Here, the authors recorded LFPs in the STN activity. LFP represents sub-threshold (e.g., synaptic input) activity at best (Buzsaki et al., 2012; Logothetis, 2003). Recent studies demonstrated that mono-polar, but also bi-polar, BG LFPs are largely contaminated by volume conductance of cortical electroencephalogram (EEG) activity even when re-referenced (Lalla et al., 2017; Marmor et al., 2017). Therefore, it is likely that STN LFPs do not accurately reflect local cellular activity. In this study, the authors examined and measured coherence between cortical areas and STN. However, they cannot guarantee that STN signals were not contaminated by volume conducted signals from the cortex.
The methods and data processing are rigorous but also very sophisticated which make the perception of the results in terms of oscillatory activity and neural synchronization difficult.
Previous studies have shown that abnormal oscillations within the STN of PD patients are limited to its dorsolateral/motor region, thus dividing the STN into a dorsolateral oscillatory/motor region and ventromedial non-oscillatory/non-motor region (Kuhn et al. 2005; Moran et al. 2008; Zaidel et al. 2009, 2010; Seifreid et al. 2012; Lourens et al. 2013, Deffains et al., 2014). However, the authors do not provide clear information about the location of the LFP recordings within the STN.
Overall, the methods and analysis strategy are innovative and rigorously conducted. However, there are still defects/flaws in the methodological approach which should be corrected in order to guarantee that the results reported in this study support authors' claims and conclusions. The paper will be of particular interest for neuroscientists and clinical community interesting in the PD pathophysiology and the development of new therapeutic approaches aiming at restoring normal cortico-basal ganglia activity.
-

Reviewer #2 (Public Review):
Sharma et al. investigated the effect of dopaminergic medication on brain networks in patients with Parkinson's disease combining local field potential recordings from the subthalamic nucleus and magnetencephalography during rest. They aim to characterize both physiological and pathological spectral connectivity.
They identified three networks, or brain states, that are differentially affected by medication. Under medication, the first state (termed hyperdopaminergic state) is characterized by increased connectivity of frontal areas, supposedly responsible for deteriorated frontal executive function as a side effect of medical treatment. In the second state (communication state), dopaminergic treatment largely disrupts cortico-STN connectivity, leaving only selected pathways communicating. This is in line …
Reviewer #2 (Public Review):
Sharma et al. investigated the effect of dopaminergic medication on brain networks in patients with Parkinson's disease combining local field potential recordings from the subthalamic nucleus and magnetencephalography during rest. They aim to characterize both physiological and pathological spectral connectivity.
They identified three networks, or brain states, that are differentially affected by medication. Under medication, the first state (termed hyperdopaminergic state) is characterized by increased connectivity of frontal areas, supposedly responsible for deteriorated frontal executive function as a side effect of medical treatment. In the second state (communication state), dopaminergic treatment largely disrupts cortico-STN connectivity, leaving only selected pathways communicating. This is in line with current models that propose that alleviation of motor symptoms relates to the disruption of pathological pathways. The local state, characterized by STN-STN oscillatory activities, is less affected by dopaminergic treatment.
The authors utilize sophisticated methods with the potential to uncover the dynamics of activities within different brain network, which opens the avenue to investigate how the brain switches between different states, and how these states are characterized in terms of spectral, local, and temporal properties. The conclusions of this paper are mostly well supported by data, but some aspects, mainly about the presentation of the results, remain:
The presentation of the results is suboptimal and needs improvement to increase readers' comprehension. At some points this section seems rather unstructured, some results are presented multiple times, and some passages already include points rather suitable for the discussion, which adds too much information for the results section.
It is intriguing that the hyperdopaminergic state is not only identified under medication but also in the off-state. This is intriguing, especially with the results on the temporal properties of states showing that the time of the hyperdopaminergic state is unaffected by medication. When such a state can be identified even in the absence of levodopa, is it really optimal to call it "hyperdopaminergic"? Do the results not rather suggest that the identified network is active both off and on medication, while during the latter state its' activities are modulated in a way that could relate to side effects?
Some conclusions need to be improved/more elaborated. For example, the coherence of bilateral STN-STN did not change between medication off and on the state. Yet it is argued that a) "Since synchrony limits information transfer (Cruz et al. 2009; Cagnan, Duff, and Brown 2015; Holt et al. 2019) , local oscillations are a potential mechanism to prevent excessive communication with the cortex" (line 436) and b) "Another possibility is that a loss of cortical afferents causes local basal ganglia oscillations to become more pronounced" (line 438). Can these conclusions really be drawn if the local oscillations did not change in the first place?
-

Reviewer #1 (Public Review):
The largest concern with the manuscript is its use of resting-state recordings in Parkinson's Disease patients on and off levodopa, which the authors interpret as indicative of changes in dopamine levels in the brain but not indicative of altered movement and other neural functions. For example, when patients are off medication, their UPDRS scores are elevated, indicating they likely have spontaneous movements or motor abnormalities that will likely produce changed activations in MEG and LFP during "rest". Authors must address whether it is possible to study a true "resting state" in unmedicated patients with severe PD. At minimum this concern must be discussed in the manuscript.
This reviewer was unclear on why increased "communication" in the medial OFC in delta and theta was interpreted as a pathological …
Reviewer #1 (Public Review):
The largest concern with the manuscript is its use of resting-state recordings in Parkinson's Disease patients on and off levodopa, which the authors interpret as indicative of changes in dopamine levels in the brain but not indicative of altered movement and other neural functions. For example, when patients are off medication, their UPDRS scores are elevated, indicating they likely have spontaneous movements or motor abnormalities that will likely produce changed activations in MEG and LFP during "rest". Authors must address whether it is possible to study a true "resting state" in unmedicated patients with severe PD. At minimum this concern must be discussed in the manuscript.
This reviewer was unclear on why increased "communication" in the medial OFC in delta and theta was interpreted as a pathological state indicating deteriorated frontal executive function. Given that the authors provide no evidence of poor executive function in the patients studied, the authors must at least provide evidence from other studies linking this feature with impaired executive function.
In this article, authors repeatedly state their method allows them to delineate between pathological and physiological connectivity, but they don't explain how dynamical systems and discrete-state stochasticity support that goal.
-

Evaluation Summary:
This manuscript is of broad interest to readers in the field of clinical neuroscience and movement disorders. The identification and characterization of dynamic networks that are differentially affected by dopaminergic medication in patients with Parkinson's disease is an important contribution to our understanding of physiologic and pathologic brain activities. The used methods provide the potential to uncover spectral, local, and temporal properties of dynamic neural systems. Overall, the data are properly analyzed, although many aspects of reporting the results could be worked out better.
(This preprint has been reviewed by eLife. We include the public reviews from the reviewers here; the authors also receive private feedback with suggested changes to the manuscript. Reviewer #1 and Reviewer #2 agreed to share …
Evaluation Summary:
This manuscript is of broad interest to readers in the field of clinical neuroscience and movement disorders. The identification and characterization of dynamic networks that are differentially affected by dopaminergic medication in patients with Parkinson's disease is an important contribution to our understanding of physiologic and pathologic brain activities. The used methods provide the potential to uncover spectral, local, and temporal properties of dynamic neural systems. Overall, the data are properly analyzed, although many aspects of reporting the results could be worked out better.
(This preprint has been reviewed by eLife. We include the public reviews from the reviewers here; the authors also receive private feedback with suggested changes to the manuscript. Reviewer #1 and Reviewer #2 agreed to share their names with the authors.)
-